Abbott Unveils Post-Approval Study Data Demonstrating Potential of Esprit BTK to Treat Significantly Sicker Patients with CLTI and Complex Lesions
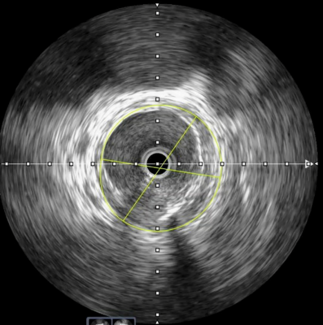
Initial data from the Esprit™ BTK Post-Approval Study (PAS) presented at the Charing Cross Symposium in London, UK, revealed promising 30-day results that showed Abbott’s Esprit™ BTK Everolimus Eluting Resorbable Scaffold System is a safe and effective approach in patients with very challenging critical limb-threatening ischemia (CLTI) and complex lesions.
Interim findings are consistent with outcomes from the pivotal LIFE-BTK study – previously published in the New England Journal of Medicine and presented at the 2025 Transcatheter Cardiovascular Therapeutics meeting – despite a range of challenging patient types that were not part of the original LIFE-BTK study, including people with end-stage renal disease and those classified as Rutherford-Becker 6, an advanced stage of CLTI in which ulcers or gangrene are already present.
At 30-days post treatment, the interim data from the PAS study (n=200) demonstrated promising early results, including:
- All patients (100%) were free from reintervention (defined as clinically-driven target lesion revascularization).
- A high safety profile comparable to the LIFE-BTK study, with 2.6% for the composite endpoint of major adverse limb events and peri-operative death, including 1% above-ankle amputation, which was not related to the device.
- Physicians successfully treated longer target lesions with an average length of 69.6 ± 61.4 mm, as reported by investigators (range: 5.0 mm to 400 mm).
- Lesions in the LIFE-BTK trial were an average length of 52.8 ± 41.9 mm (range: 2.0 mm to 170 mm). - Physicians were also able to treat more advanced calcifications with the sites reporting 39.7% of lesions were categorized as moderate and 19.2% considered severe.
- In the LIFE-BTK trial, sites categorized 27.7% of lesions to be moderate and only 3.0% considered severe.
"The initial 30-day results from the Esprit BTK Post-Approval Study demonstrate how beneficial the Esprit BTK System could be to help reduce amputations and improve the lives of those most at risk," stated Ramon Varcoe, Professor of Surgery and Academic Lead at University of New South Wales. “We’re seeing early evidence that Esprit BTK can deliver the same robust safety and efficacy observed in LIFE-BTK, even when tackling sicker patients with longer, more complex calcified lesions. While these are still early results, it opens the possibility that the Esprit BTK System could be beneficial for those with more severe wounds, end-stage renal disease, and diverse populations who are at risk of amputation.”
“The initial results from the Esprit BTK Post-Approval Study are highly encouraging, further validating our technology in the real world,” said Ethan Korngold, M.D., Chief Medical Officer at Abbott. “These data, especially the consistent performance observed in challenging patients with more advanced disease and complex lesions, reinforce our belief that Esprit BTK is a critical tool in the fight against CLTI. Esprit BTK performs well in the real world: in calcified lesions, long lesions, chronic total occlusions, and in conjunction with advanced vessel prep. It’s an important step toward expanding options for those most at risk of amputation and improving lives.”
About the Esprit BTK Post Approval Study:
The Esprit BTK Post-Approval Study (PAS) is a prospective, multicenter, single-arm, observational study assessing the long-term safety and effectiveness of the Esprit BTK System. Following the positive LIFE-BTK trial, this PAS monitors patients with CLTI treated with the dissolvable scaffold to prevent artery re-closure. Interim findings from this study are consistent with LIFE-BTK and support the real-world performance of the Esprit BTK scaffold.
About the LIFE-BTK Trial:
The pivotal LIFE-BTK trial established a strong foundation for the resorbable Esprit BTK System, demonstrating superiority of the technology over standard of care for treatment of below-the-knee (BTK) disease in CLTI patients.i Results from LIFE-BTK showed favorable rates of restoring blood flow and high, sustained limb salvage rates, alongside a low incidence of adverse events, for its enrolled patient population.ii These positive outcomes set a crucial benchmark for the Esprit BTK System, paving the way for new possibilities in reducing amputation and mortality rates in patients.
About CLTI:
CLTI is the most advanced stage of peripheral artery disease caused by severe blockages in arteries that significantly restrict blood flow to the legs or feet. The condition can lead to persistent pain, ulcers, and gangrene if left untreated. As many as 40% of people with CLTI will undergo an amputation.iii
i. Varcoe, RL., et al. Drug-Eluting Resorbable Scaffold versus Angioplasty for Infrapopliteal Artery Disease. N Eng J Med 2024;390:9-19.
ii. Parikh, S., Three-Year Outcomes of the LIFE-BTK Randomized Controlled Trial Evaluating the Esprit™ BTK Drug-Eluting Resorbable Scaffold for Treatment of Infrapopliteal Lesions, Presented at TCT 2025.
iii. Uccioli L, Meloni M, Izzo V, et al. Critical limb ischemia: current challenges and future prospects. Vasc Health Risk Manag. 2018;14:63-74.
Find More:
Renal Denervation Topic Center
Cardiovascular Ambulatory Surgery Centers (ASCs) Topic Center
Grand Rounds With Morton Kern, MD
Peripheral Artery Disease Topic Center
Go to Cath Lab Digest's Current Issue
Go to the Journal of Invasive Cardiology Issue