Palmar Filet Flap for Soft Tissue Reconstruction and Targeted Muscle Reinnervation for Transradial Amputation
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of ePlasty or HMP Global, their employees, and affiliates.
Abstract
Background. Preserving skeletal length in transradial amputation is critical to maintain functional movement and prosthetic fitting. In traumatic or oncologic cases where soft tissue coverage is insufficient, free tissue transfer may be necessary to maximize skeletal length. For patients with segmental disease restricted to the forearm, the hand that would otherwise be discarded can serve as a “spare-parts” free flap. This report describes the use of a palmar filet free flap for coverage and targeted reinnervation in the setting of transradial amputation.
Methods. A 63-year-old woman with multiply recurrent dermal pleomorphic sarcoma of the right forearm underwent transradial amputation for definitive oncologic control. As the hand was not involved by tumor, it was repurposed as a palmar filet free flap to reconstruct the amputation stump without compromising forearm length and achieve targeted muscle reinnervation.
Results. The patient underwent successful oncologic resection with negative margins and immediate reconstruction using a palmar free flap. At the 6-month follow-up, she demonstrated protective sensation in the flap without neuroma pain and was successfully fitted with a myoelectric prosthesis.
Conclusions. In select patients, a palmar free flap offers a unique reconstructive option following transradial amputation. This approach provides durable soft tissue coverage without added donor site morbidity, enables sensory restoration through neurotization, and reduces the risk of neuroma-related pain via targeted muscle reinnervation.
Introduction
Transradial amputation is the most common form of major limb amputation in the upper extremity.1 Preservation of adequate forearm length maintains pronation-supination and flexion-extension movement and allows for improved prosthetic fit, which improves functional outcomes for patients.2 The primary method of preserving length relies on native soft tissue to provide coverage of bone and muscle. However, surgeons can be faced with poor quality or inadequate soft tissue to maximize the length of transradial amputations in cases such as trauma or oncologic ablation where wide surgical margins are required. This may necessitate the use of skin grafts, regional flaps, or free flaps to provide adequate coverage of the residual limb without skeletal shortening, which would compromise function.
In situations of amputation for disease restricted to the forearm rather than the hand, it is not uncommon that a short transradial amputation at the borderline of sufficient or insufficient bony length may be indicated. In these cases, there exists an opportunity to augment amputation stump coverage and preserve critical length by utilizing the hand as a free flap donor site, which would otherwise be discarded with the specimen.3 There are numerous potential advantages of this “spare-parts” fillet flap technique including (1) the accomplishment of free tissue transfer without additional donor site morbidity, (2) the provision of tough glabrous skin at the limb-prosthesis interface, and (3) the achievement of flap neurotization, which offers a sensate stump and may mitigate postoperative neuropathic pain.4-6 In this article, we describe a patient who was indicated for a short transradial amputation because of a recurrent sarcoma of the forearm. We describe our surgical technique for preservation of the below-elbow level with sufficient length to maintain flexion-extension in a prosthesis, as well as nerve management using a “spare-parts” palmar filet free flap.
Surgical anatomy
Transradial amputation involves removal of the upper extremity through the radius and ulna with preservation of the elbow joint. Functional outcomes of transradial amputation depend on how much bony length is maintained. The degree of pronation-supination range of motion corresponds to the residual length of the limb and whether the pronator teres muscle is preserved.2 At least 8 cm of residual length is required for some maintenance of forearm rotation.2 Even without forearm rotation, a minimum ulna length of 5 cm is recommended for successful prosthetic fitting with maintenance of functional flexion-extension.2,7 Although some surgeons recommend reinserting the biceps tendon from the radial tuberosity to the residual ulna to facilitate elbow flexion in short transradial amputations, the relative benefit of this is unclear when the brachialis is functional.7-9
Depending on the level of transradial amputation, most of the musculature of the anterior compartment, posterior compartment, and mobile wad may be removed. Vascular control of major arteries (radial and ulnar) and veins (cephalic and basilic) is performed. Similarly, major nerves including the median, ulnar, radial sensory, and lateral antebrachial cutaneous (LABC) nerves are identified and transected with a plan for targeted muscle reinnervation (TMR), regenerative peripheral nerve interface (RPNI), or other forms of primary nerve stump management.7 When a free flap for soft tissue coverage and TMR is anticipated, vessels and nerves are ligated as distally as appropriate for the given context (eg, respecting oncologic margins).
The palmar filet flap can be raised as a composite flap depending on the reconstructive needs of the defect. For soft tissue coverage, it can be raised as a myocutaneous flap incorporating the intrinsic muscles of the hand and using the ulnar artery as the vascular pedicle. The cephalic vein and other superficial veins can also be harvested as additional venous drainage options. The median and ulnar nerves, along with terminal cutaneous branches such as the palmar cutaneous nerve and LABC, are elevated with the flap as recipients for TMR and/or sensory nerve transfer. Distally at the metacarpophalangeal joint level, the digital nerves and arteries can be ligated; alternatively, the digits can be fileted with dorsal incisions to increase the reach and coverage area of the flap.
Methods
History and presentation
A 63-year-old woman with a history of right forearm dermal pleomorphic sarcoma had previously undergone multiple resections because of recurrence of disease. Along with 4 previous surgical resections, she had also received adjuvant radiation and brachytherapy. In initial efforts to pursue limb salvage, the oncologic defects were reconstructed with skin grafts, a lateral arm flap, and a medial sural artery perforator flap at different stages of recurrence. She presented again with a palpable mass on the dorsal forearm, which was biopsy-proven positive for another recurrence within a year of her most recent resection. Metastatic workup was negative. The refractory and recurrent nature of her cancer, in addition to declining limb function related to a pathologic ulnar fracture, informed the patient’s decision to proceed with a transradial amputation.
The objective of the case was to achieve oncologic clearance and reconstruct a maximally functional and minimally painful residual upper extremity. In order to resect all previously operated tissue and new recurrence just proximal to the midpoint of the forearm (Figure 1), the transradial amputation would be performed fairly proximally to achieve 2-cm surgical margins. It was clear preoperatively that local soft tissues would be inadequate to close the wound while maintaining enough bony length for optimal function and prosthetic fitting. As the palm of the hand was unaffected by cancer, we recommended a free palmar filet flap to maintain limb length and for TMR, aiming to maximize myoelectric signals for future prosthetic use and reduce the risk of neuroma formation.
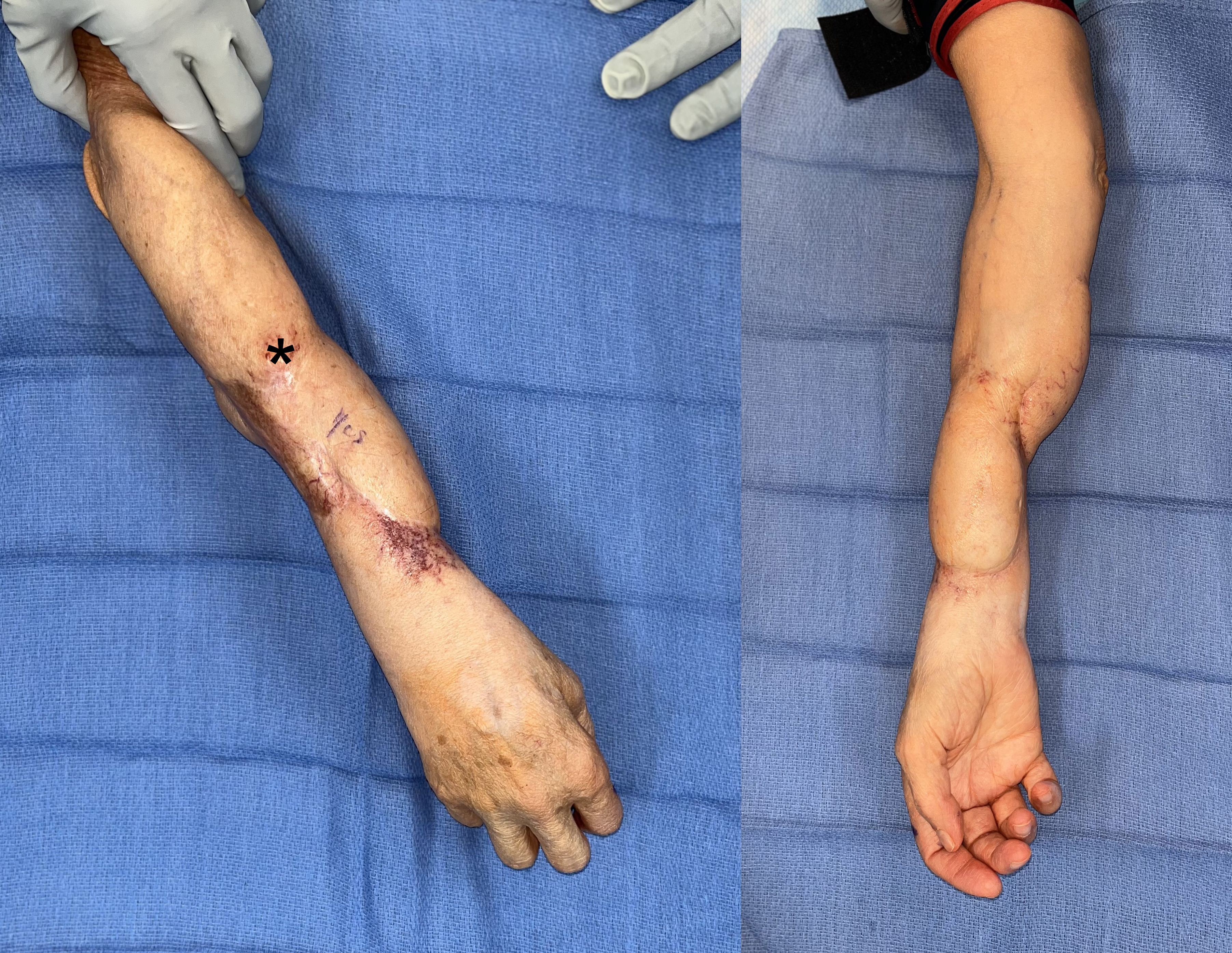
Figure 1: The right arm was previously reconstructed with skin grafting, a lateral arm, and medial sural artery perforator free flaps. The asterisk (*) notes the site of recurrence.
Surgical technique
The palmar filet flap was elevated prior to the transradial amputation to confirm adequate perfusion to the flap and minimize ischemia time. Under tourniquet control, a palmar flap was designed based on the ulnar artery as the vascular pedicle. The radial incision bordered the margin of the thenar eminence; the ulnar incision was extended to include some dorsal skin and a superficial vein, and the distal incision was at the palmar digital crease (Figure 2). Proximally, the ulnar neurovascular bundle, the median nerve including its palmar cutaneous branch, and the LABC nerve were identified and protected. The radial artery was ligated.
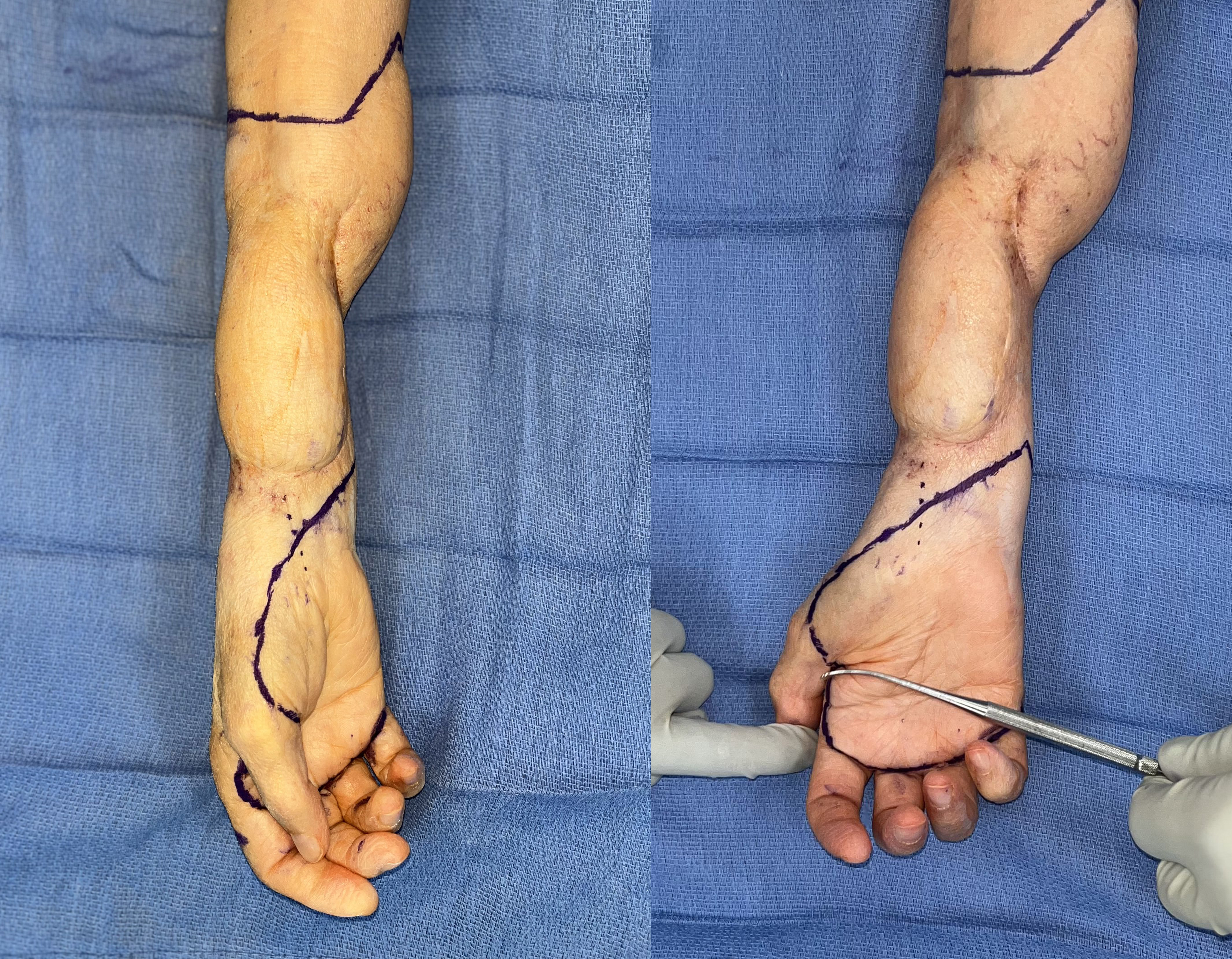
Figure 2: Surgical markings for the palmar free filet flap.
The flap was then elevated in a submuscular plane starting radially at the thenar eminence. The entirety of the intrinsic hand musculature, including the thenar eminence, the hypothenar eminence, and the volar and dorsal interossei, was raised as a myocutaneous flap directly off the metacarpals using monopolar cautery (Figure 3). At each digit, the flexor tendons, lumbricals, and proper digital arteries were divided. The proper digital nerves were divided distally using a crush and cauterize technique to minimize the risk of neuroma formation at the digit following reinnervation by the median and ulnar nerves via TMR.
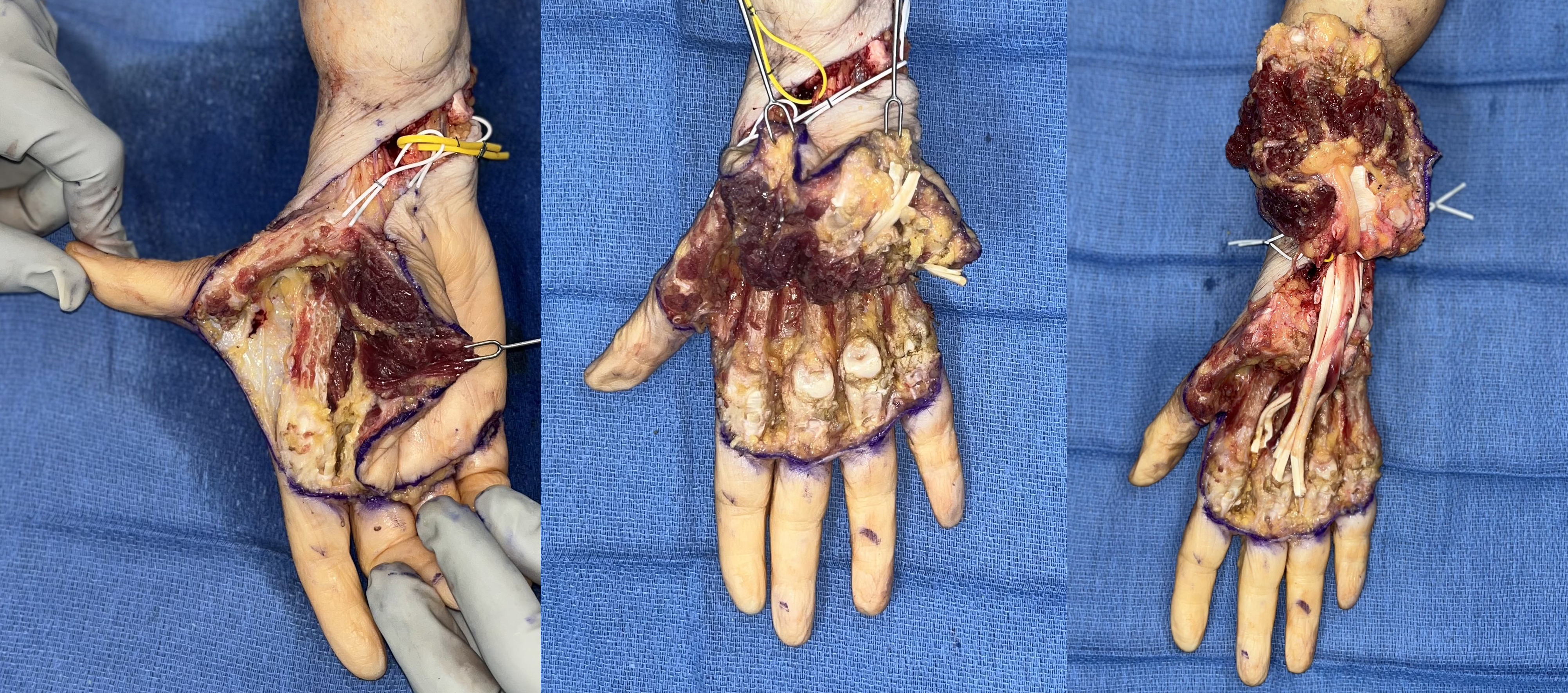
Figure 3: Elevation of the myocutaneous palmar free filet flap with harvest of the thenar eminence, hypothenar eminence, and intrinsic muscles of the hand.
The carpal tunnel was entered by dividing the radial and ulnar attachments of the transverse carpal ligament. The median nerve was then elevated together with the flap while leaving behind the flexor tendons. Once the flap was fully elevated, the tourniquet was deflated to confirm perfusion and to achieve hemostasis (Figure 4).
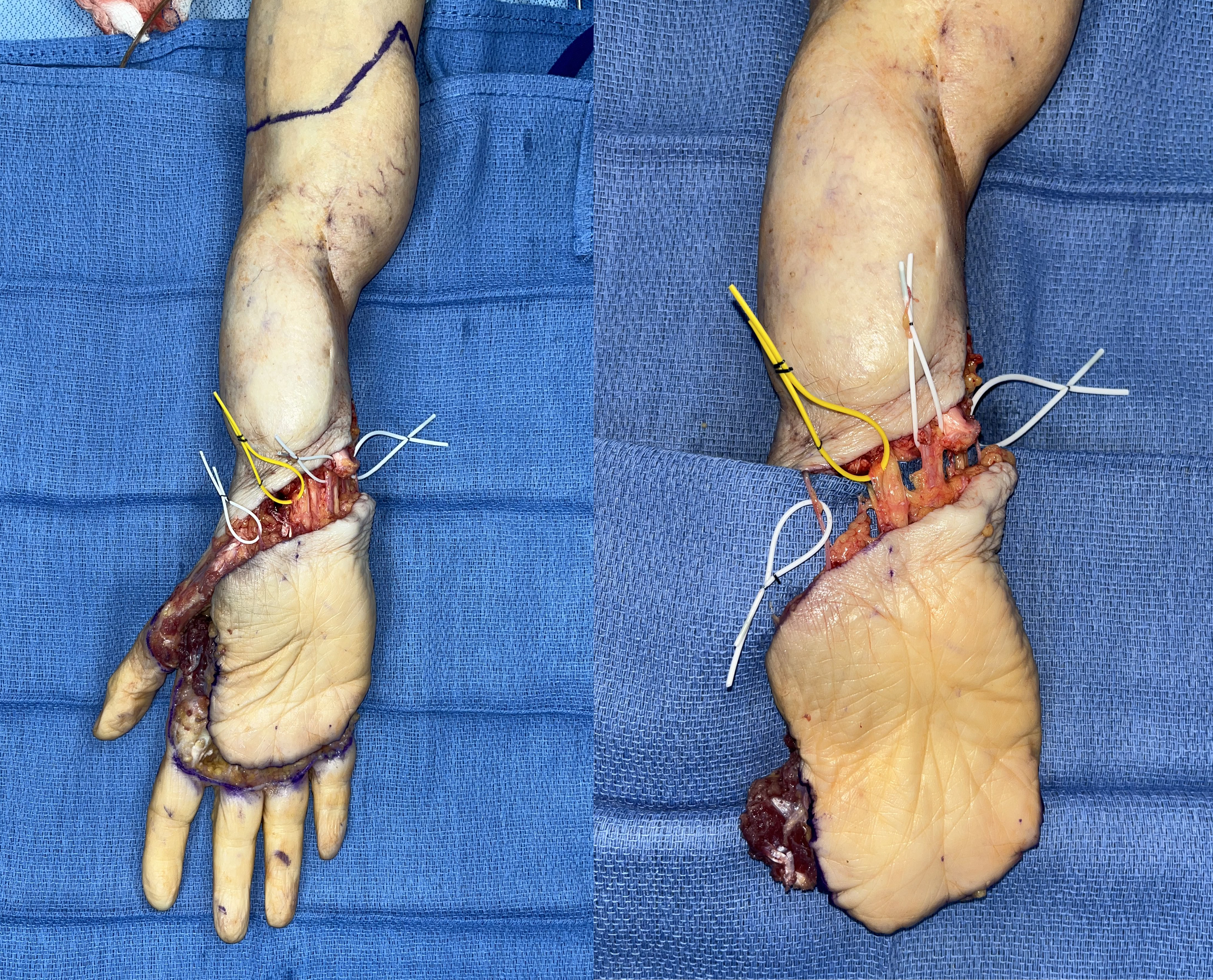
Figure 4: Completed elevation of the flap. The structures were identified with vessel loops from radial to ulnar: lateral antebrachial cutaneous nerve (white loop), palmar cutaneous branch of the median nerve (yellow loop), ulnar neurovascular bundle (white loop), and superficial dorsal vein (white loop).
The tourniquet was then reinflated after 20 minutes of reperfusion to the flap to proceed with transradial amputation. A 2-cm margin was marked proximally to the recurrent tumor. Critical structures, including the radial sensory nerve, LABC, median nerve, ulnar nerve, and radial artery, were identified and divided as distally as possible while respecting the surgical margins. The ulnar artery was kept intact so that perfusion to the flap could be checked again. The radial sensory nerve was dissected proximally to identify the posterior interosseous nerve (PIN), the supinator branch, and the extensor carpi radialis brevis (ECRB) branch. Motor branches were confirmed with use of a nerve stimulator. The forearm musculature was all divided while protecting the critical structures and leaving the radius and ulna intact.
The tourniquet was then deflated to achieve hemostasis. The flap remained perfused via the ulnar artery, which traversed the eventual surgical specimen. The flap pedicle was then divided distally, flushed with heparinized saline, and placed on a back table on ice. The transradial amputation was then completed with division of the radius and ulna, preserving roughly 6 cm of proximal skeletal length. Primary closure at this point would have required further shortening of the radius and ulna.
The palmar free flap was then brought back into the surgical field. Vascular anastomosis was completed using 9-0 nylon under the microscope from the proximal radial artery to the ulnar artery flap pedicle, along with 2 venae comitans and 2 superficial veins. Traditional TMR was then performed by coapting the radial sensory nerve to the supinator branch for neuroma control. The PIN was transferred to one of the 2 working ECRB branches as an additional proximal motor signal for myoelectric control. The proximal LABC, median, and ulnar nerves were transferred to their own distal targets in the flap, with attention to include the distal palmar cutaneous branch in the median nerve coaptation (Figure 5). The flap was inset over a Penrose drain (Figure 6).
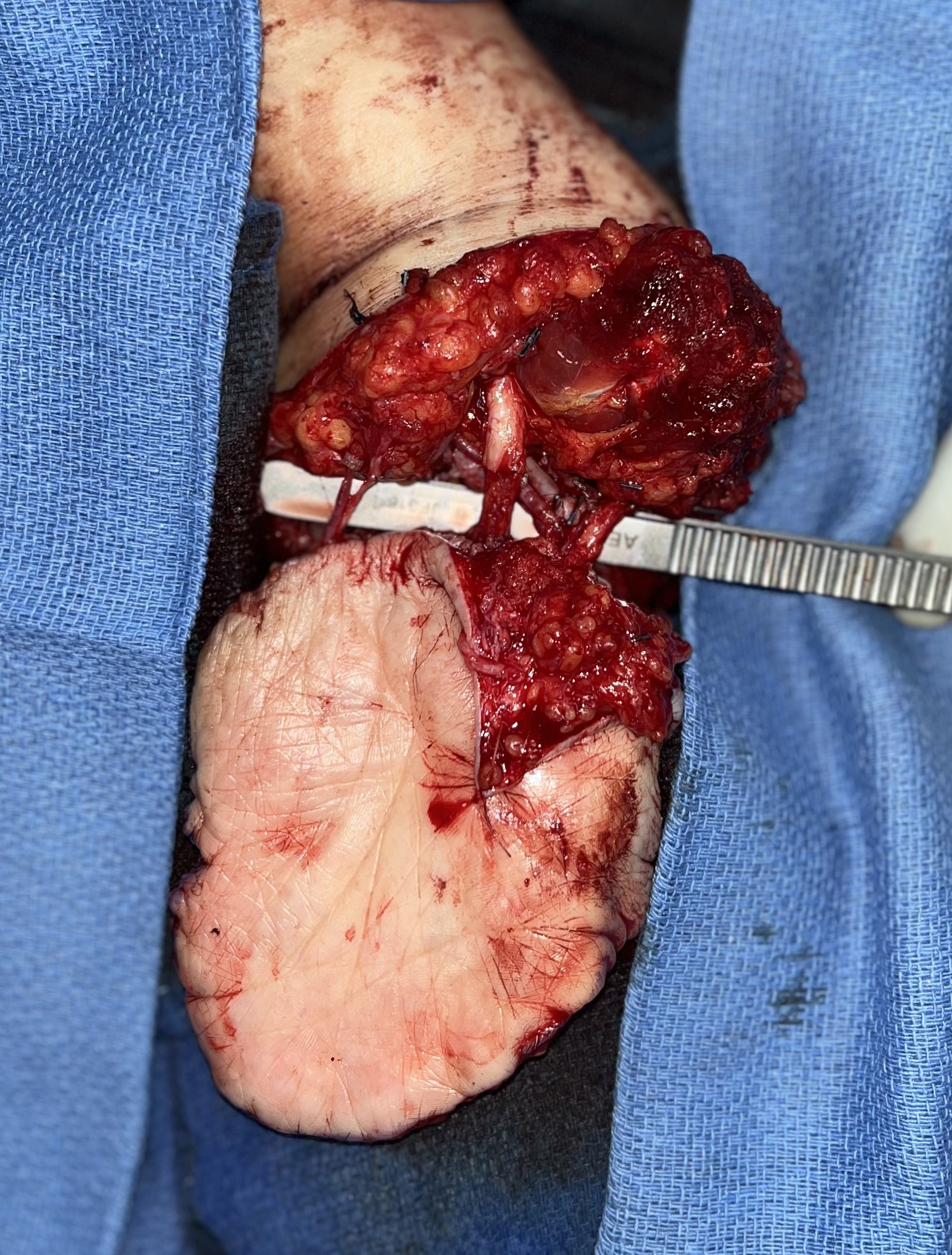
Figure 5: The ulnar artery pedicle of the flap is anastomosed to the radial artery proximally, demonstrating excellent perfusion. The proximal median and ulnar nerves are coapted to their respective distal targets in the flap.
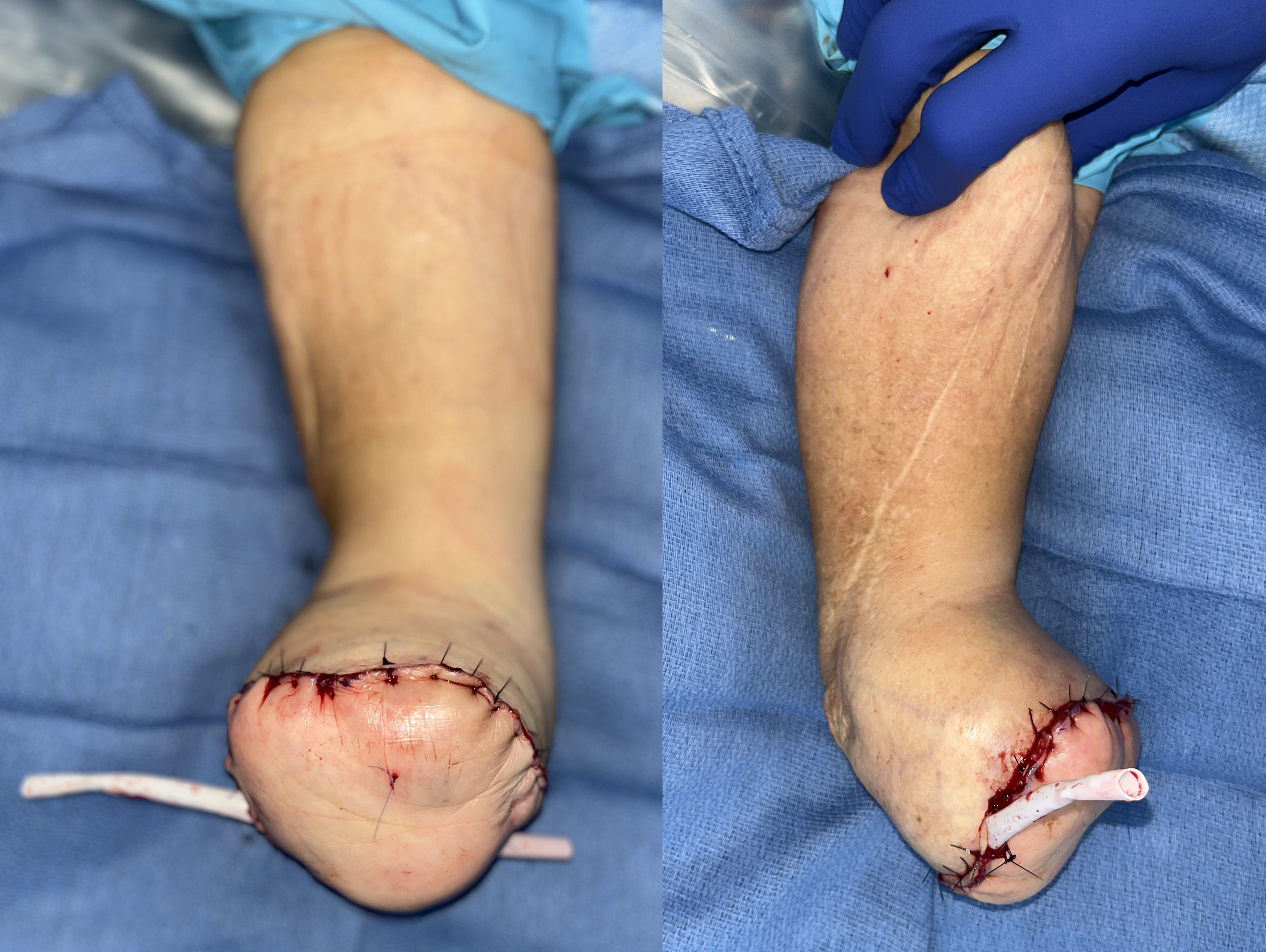
Figure 6: Closure and inset of the flap over a Penrose drain.
Results
This patient had an uneventful postoperative course and recovery. All margins were negative on permanent pathology. The patient did report early symptoms of phantom limb pain, which was managed with neuropathic pain medications and graded motor imagery. At the 3-month postoperative follow-up, she was evaluated and fitted for myoelectric prosthesis (Figure 7). At 6 months, the patient reported protective sensation in the flap without neuroma pain. She has gone on to successfully adopt a dual signal myoelectric prosthesis (Figure 8, Supplementary Video). Additional signals are available, and efforts to secure a more advanced prosthesis are ongoing.

Figure 7: At 3 months postoperative, the patient demonstrated a well-healed and padded amputation stump fitted with a myoelectric prosthesis.

Figure 8: Patient demonstrating use of her myoelectric prosthesis.
Discussion
The free palmar filet flap was an optimal choice that achieved many surgical objectives. It was used to preserve bony length in a relatively proximal transradial amputation, providing robust glabrous tissue coverage and padding as a thick myocutaneous flap.
With respect to nerve management, the combined TMR and sensory neurotization of the palmar filet flap provided a unified solution to address neuroma pain and generate signals for a myoelectric prosthesis. In this case, usable myoelectric signals could be anticipated from the natively innervated extensor carpi radialis longus (ECRL) and the partially innervated ECRB by TMR of the PIN, in addition to the intrinsic hand innervation by the median and ulnar nerve motor components. The benefits of TMR on neuropathic and phantom limb pain may take up to 12 months to be observed.10 This technique also provided excellent size match between the proximal and distal nerves for TMR, as larger nerves such as the median and ulnar nerves were able to be repaired to their own distal target. Size mismatch between a larger proximal nerve and a smaller distal nerve is a common issue in TMR, which may result in neuroma formation from axons that are not directed through endoneurial tubules.11 Including nerve transfer to the median nerve (and its palmar cutaneous branch) and the ulnar nerve may allow for the recovery of protective sensory perception within the flap.
Transradial amputation is associated with upper extremity dysfunction and postoperative neuropathic pain. While a free palmer filet flap can help mitigate neuroma formation and improve upper extremity function for amputees, it needs to be carefully applied for the correct patient. As a free flap, it carries risk of microvascular complications, which should be weighed against its benefits, particularly in comorbid patients with peripheral vascular disease, coagulopathies, and smoking history. There is also the risk of occult residual disease in the flap itself in oncologic cases, such as this patient. It is critical to have a complete oncologic history of recurrent disease, treatment course, and reconstructions to guide surgical decision-making. The objective of preserving pedicle and nerve length should always be secondary to obtaining an adequate oncologic margin.
Conclusions
In high transradial amputation approaching the elbow joint, the maintenance of maximal skeletal length is critical to maximize options for prosthetic rehabilitation. In cases of transradial amputation for the indication of forearm malignancy where the hand is unaffected, a “spare-parts” palmar free filet flap can be used to maximize limb length. This technique not only provides tough, glabrous, and potentially sensate tissue to cap the amputation but also preserves a few critical centimeters of skeleton. Length preservation is important for maintaining pronation-supination (approximately 8 cm required) and/or elbow flexion in a prosthesis (approximately 5 cm required). The flap also provides large nerve targets for TMR and sensory neurotization, which likely mitigate painful neuroma and provide additional myoelectric signals via the intrinsic hand musculature.
Acknowledgments
Authors: Brian Chin, MD, MSc1; Rosemarie Rinfret-Paquet, MD2; Mitchell A. Pet, MD3
Affiliations: 1Division of Plastic Surgery, McMaster University, Hamilton, Ontario, Canada; 2Division of Plastic Surgery, Laval University, Quebec City, Quebec, Canada; 3Division of Plastic and Reconstructive Surgery, Washington University School of Medicine, St. Louis, Missouri
Correspondence: Mitchell A. Pet, MD; mpet@wustl.edu
Ethics: The patient provided written informed consent for publication of this case report and all accompanying clinical details and images.
Disclosure: The authors disclose no financial or other conflicts of interest.
References
1. Tennent DJ, Wenke JC, Rivera JC, Krueger CA. Characterisation and outcomes of upper extremity amputations. Injury. 2014;45(6):965-969. doi:10.1016/j.injury.2014.02.009
2. Fitzgibbons P, Medvedev G. Functional and clinical outcomes of upper extremity amputation. J Am Acad Orthop Surg. 2015;23(12):751-760. doi:10.5435/JAAOS-D-14-00302
3. Cavadas PC, Raimondi P. Free fillet flap of the hand for elbow preservation in nonreplantable forearm amputation. J Reconstr Microsurg. 2004;20(5):363-366. doi:10.1055/s-2004-829999
4. Pet MA, Ko JH, Friedly JL, Mourad PD, Smith DG. Does targeted nerve implantation reduce neuroma pain in amputees? Clin Orthop Relat Res. 2014;472(10):2991-3001. doi:10.1007/s11999-014-3602-1
5. Souza JM, Cheesborough JE, Ko JH, Cho MS, Kuiken TA, Dumanian GA. Targeted muscle reinnervation: a novel approach to postamputation neuroma pain. Clin Orthop Relat Res. 2014;472(10):2984-2990. doi:10.1007/s11999-014-3528-7
6. Dumanian GA, Potter BK, Mioton LM, et al. Targeted muscle reinnervation treats neuroma and phantom pain in major limb amputees: a randomized clinical trial. Ann Surg. 2019;270(2):238-246. doi:10.1097/SLA.0000000000003088
7. Morgan EN, Kyle Potter B, Souza JM, Tintle SM, Nanos GP III. Targeted muscle reinnervation for transradial amputation: description of operative technique. Tech Hand Up Extrem Surg. 2016;20(4):166-171. doi:10.1097/BTH.0000000000000141
8. Chow I, Gaston RG. Transradial amputation and wrist disarticulation. Operative Techniques in Orthopaedics. 2023;33(3):101058. doi:10.1016/j.oto.2023.101058
9. Ilayperuma I, Uluwitiya SM, Nanayakkara BG, Palahepitiya KN. Re-visiting the brachialis muscle: morphology, morphometry, gender diversity, and innervation. Surg Radiol Anat. 2019;41(4):393-400. doi:10.1007/s00276-019-02182-2
10. Alexander JH, Jordan SW, West JM, et al. Targeted muscle reinnervation in oncologic amputees: Early experience of a novel institutional protocol. J Surg Oncol. 2019;120(3):348-358. doi:10.1002/jso.25586
11. Valerio I, Schulz SA, West J, Westenberg RF, Eberlin KR. Targeted muscle reinnervation combined with a vascularized pedicled regenerative peripheral nerve interface. Plast Reconstr Surg Glob Open. 2020;8(3):e2689. doi:10.1097/GOX.0000000000002689