Cycling Through the Same ADHD Treatments Without Finding the Right Fit?
Take a different approach1-6
Qelbree (viloxazine extended-release capsules)—
The only ADHD treatment with serotonin (5-HT2c) pharmacodynamics approved in FDA product labeling1,7
INDICATION
Qelbree is indicated for the treatment of ADHD in adults and pediatric patients 6 years and older.

Please see full Important Safety Information to the top left.
Rx Qelbree for multimodal pharmacodynamics1
Qelbree is the first and only ADHD nonstimulant with a multimodal pharmacodynamic profile1-4
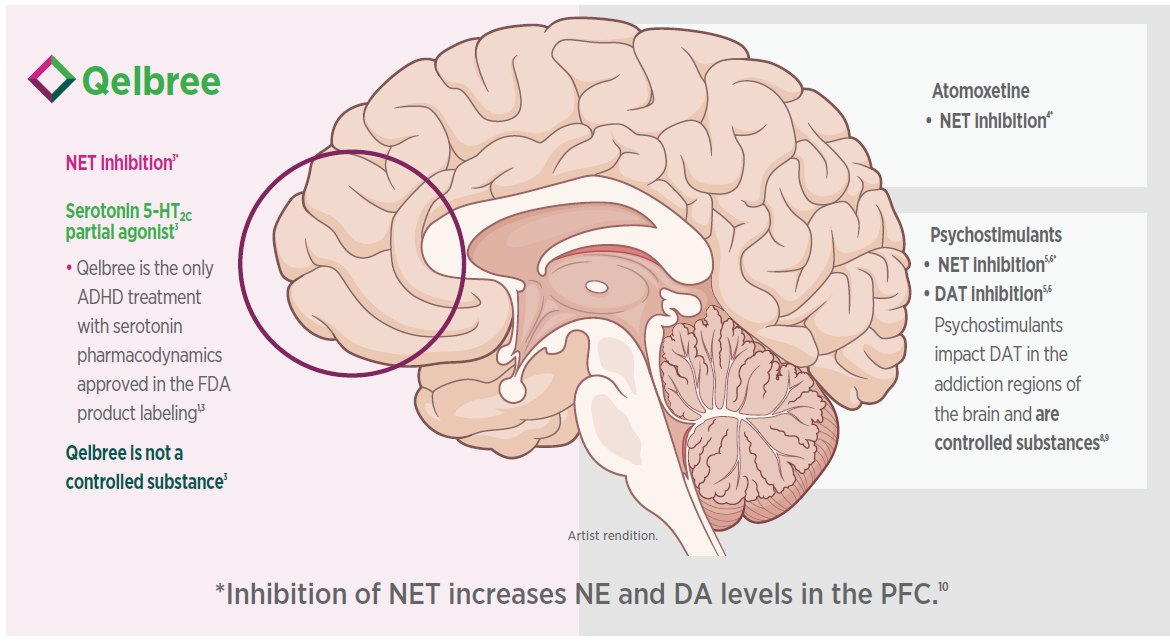
The pharmacodynamic activity of viloxazine is based on non-clinical studies and the clinical significance of the data is unknown. The mechanism of action of viloxazine in the treatment of ADHD is unclear; however, it is thought to be through inhibiting the reuptake of NE.1
IMPORTANT SAFETY INFORMATION (CONT'D)
CONTRAINDICATIONS
- Concomitant administration of a monoamine oxidase inhibitor (MAOI), or dosing within 14 days after discontinuing an MAOI, because of an increased risk of hypertensive crisis
- Concomitant administration of sensitive CYP1A2 substrates or CYP1A2 substrates with a narrow therapeutic range
Please see full Important Safety Information to the top left.
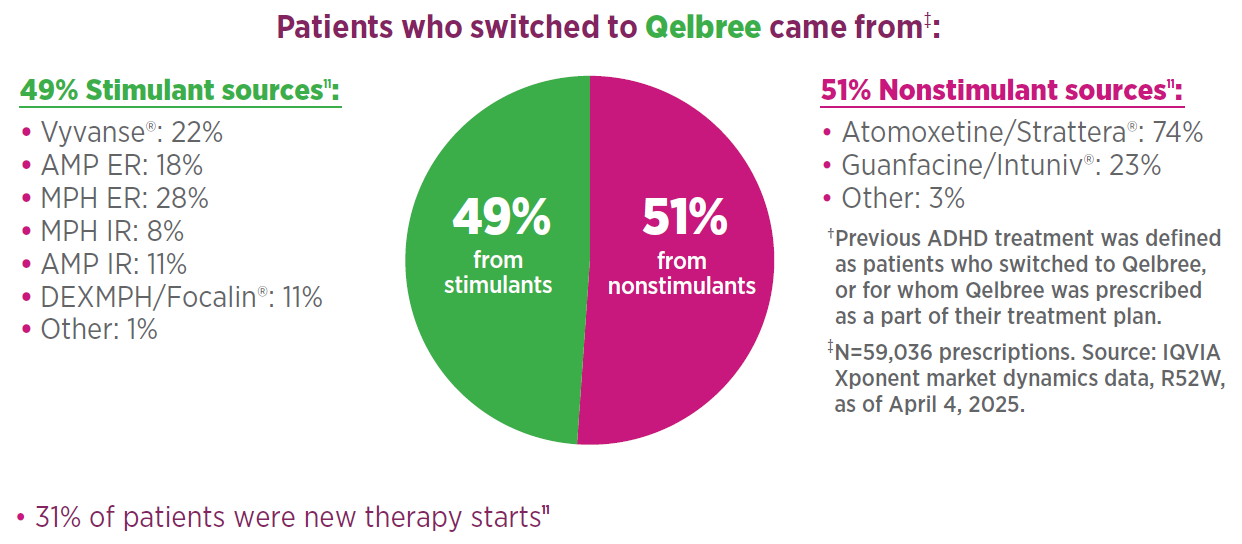
Rx Qelbree for coverage when a treatment change is needed1,11
Market data show that 69% of patients were prescribed Qelbree because a change in their previous ADHD treatment† was needed11
Qelbree is covered across ~75% of commercial lives.11
Abbreviations: ADHD, attention-deficit/hyperactivity disorder; AMP, mixed amphetamine salts; DEXMPH, dexmethylphenidate; ER, extended release; IR, immediate release; MPH, methylphenidate.
IMPORTANT SAFETY INFORMATION (CONT'D)
- Suicidal thoughts and behaviors: Closely monitor all Qelbree-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes
Please see full Important Safety Information to the top left.
Rx Qelbree for treatment convenience1
Transition patients to once-daily Qelbree
✓ Qelbree is prescribed once daily (AM or PM) for full 24-hour exposure1,11
✓ Qelbree can be conveniently prescribed and refilled without a new prescription every month1,11
✓ Qelbree has no known addiction potential or evidence of abuse1,12,13
✓ Qelbree—Up to 90 days of treatment in 1 Rx! Patients pay as little as $20* per Rx.
✓ Qelbree is available at pharmacies nationwide.11
Rx Qelbree for resources that support your patients and your practice!
Speak with your representative about resources we provide to help you start the transition!

IMPORTANT SAFETY INFORMATION (CONT'D)
- Heart rate, blood pressure increases: Qelbree can cause an increase in diastolic blood pressure and heart rate. Assess these measures prior to starting therapy, following increases in dosage, and periodically during therapy
Please see full Important Safety Information to the top left.
Please see full Prescribing Information, including Boxed Warning.
Qelbree (viloxazine extended-release capsules) is available in 100 mg, 150 mg, and 200 mg capsule strengths. Learn more about Qelbree, an extended-release, nonstimulant medication for ADHD: https://www.QelbreeHCP.com/
References:
- Qelbree [package insert]. Rockville, MD: Supernus Pharmaceuticals, Inc.
- Strattera [package insert]. Indianapolis, IN: Lilly USA, LLC.
- Kapvay [package insert]. Atlanta, GA: Shionogi Pharma, Inc.
- Intuniv [package insert]. Lexington, MA: Takeda Pharmaceuticals U.S.A.
- Ritalin [package insert]. East Hanover, NJ: Novartis Pharmaceuticals Corporation. doi:10.1016/j.jpeds.2009.01.018
- Adderall XR [package insert]. Horsham, PA. Teva Pharmaceuticals USA.
- Arnsten AFT. The emerging neurobiology of attention deficit hyperactivity disorder: the key role of the prefrontal association cortex. J Pediatr. 2009;154(5):I-S43. doi:10.1016/j.jpeds.2009.01.018
- Verghese C, Patel P, Abdijadid S. Methylphenidate. In: StatPearls. StatPearls Publishing; 2024. January 13, 2025. https://www.ncbi.nlm.nih.gov/books/NBK482451/
- Martin D, Le JK. Amphetamine. In: StatPearls. StatPearls Publishing; 2023. Accessed January 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK556103/
- Stahl SM. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications. 5th ed. Cambridge University Press; 2021.
- Data on file, Supernus Pharmaceuticals.
- Yanagita T, Wakasa Y, Kiyohara H. Drug dependence potential of viloxazine hydrochloride tested in rhesus monkeys. Pharmacol Biochem Behav. 1980;12:155-161.
- Food and Drug Administration. Table of Prescription Stimulant Label Changes. August 26, 2025. https://www.fda.gov/media/168050/download
QBE.2026-0035










