Cutaneous Safety Evaluation of Pure Hypochlorous Acid Solution in Preterm Infants and Neonates With Complex Wounds
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Preterm infants are at heightened risk of skin injury and infection due to the immaturity of their epidermal barrier and immune defenses. Safe wound cleansing options remain limited in this population. Objective. To evaluate cutaneous safety and tolerability associated with use of a pure hypochlorous acid-preserved cleanser (pHA) in preterm and neonatal patients with complex wounds. Materials and Methods. This retrospective study evaluated the cutaneous safety of a pHA in 100 preterm and neonatal patients with complex wounds admitted to a tertiary care facility between January 2023 and July 2025. Demographic, clinical, and wound data were extracted from the electronic medical record. pHA was applied at each wound care encounter and was continued until wound closure, discharge, or transfer of care. Results. Patients received a mean of 7.6 pHA applications over a mean treatment duration of 18.7 days. The youngest treated infant was born at 21 weeks’ gestation. Across 766 cumulative applications, no cutaneous adverse effects or wound-related complications were observed, including contact dermatitis, erythema, chemical burns, infection, or secondary breakdown. In patients receiving pHA concurrently with other advanced wound therapies, no cutaneous adverse effects attributable to combined use were identified. Conclusion. This retrospective series represents the largest reported evaluation to date of cutaneous safety and tolerability associated with pHA use in neonatal patients. Future prospective multicenter studies are warranted to further characterize cutaneous safety of pHA use and to evaluate efficacy outcomes in neonatal wound care.
Preterm infants are highly vulnerable to skin injury and subsequent sepsis because of the immaturity of their cutaneous barrier and their immune system.1 Because of the combination of a thin epidermis, immature lipid composition, neutral-to-alkaline skin pH, and limited antimicrobial defenses, neonatal skin—particularly in preterm infants—has delayed barrier function, is prone to transepidermal water loss, and is more susceptible to infection and injury than adult skin.2,3 Skin injury is also common in the neonatal intensive care unit (NICU), where intrinsic fragility, invasive procedures, and frequent use of medical devices further amplify the risk of breakdown and contamination.4,5 These wounds significantly increase susceptibility to infection in the NICU setting, contributing to heightened morbidity, mortality, and prolonged hospitalization.6,7 Consequently, prevention and timely management of wound-related complications are essential components of neonatal intensive care.
Complex wounds in this population frequently arise from the structural fragility and immaturity of the epidermis and dermis.8 Whereas minor wounds such as mild diaper dermatitis or heel-stick injuries may heal with conservative measures, more complex wounds require repeated evaluation and specialized interventions such as selective debridement, negative pressure wound therapy (NPWT), or advanced dressings. In the present study, the term “complex wounds” refers to neonatal wounds requiring active management and repeated assessments, as defined in the “Materials and Methods” section.
Many commonly used wound cleansers pose risks of localized or systemic adverse effects in preterm neonates, including tissue irritation and inflammation, delayed healing, and allergic dermatitis.9 Clinical practice has also been inconsistent: Sathiyamurthy et al10 highlighted marked variation in cleansing and antiseptic use across NICUs, underscoring the lack of consensus and limited evidence base for neonatal wound care. Similarly, a survey of Canadian NICU clinicians by McCord et al11 found that only 43% were fully satisfied with currently available wound cleansing agents, further highlighting the need for safer, more effective solutions in this specific population.
Pure hypochlorous acid–preserved cleanser (pHA) has demonstrated favorable safety and utility in adult wound care populations for multiple acute and chronic wounds.12-17 As a wound cleansing agent, pHA exhibits broad-spectrum antimicrobial activity in vitro and aids in the removal of microorganisms from the wound surface.14,15 Its acidic profile (pH 3.5–5.5) approximates the physiologic pH of skin and the wound environment and may support conditions favorable for wound healing.16,17 Limited pediatric case reports18-21 suggest similar benefit, but a systematic evaluation in preterm and neonatal populations is lacking.
Building on the positive clinical experience of the authors of the present study, this retrospective analysis describes outcomes in 100 preterm and neonatal patients consecutively treated with pHA, with the primary objective of evaluating cutaneous safety in this fragile population. Safety monitoring focused on the presence or absence of cutaneous adverse effects, including contact dermatitis, wound infection, chemical burns, erosive skin injury, irritation, erythema, and secondary skin breakdown after repeated applications.
Materials and Methods
Study design
This retrospective, descriptive study evaluated the cutaneous safety of pHA (Vashe wound solution; Urgo Medical North America) in the management of complex wounds in preterm infants and neonates requiring advanced wound care interventions. The study followed a nonmasked, noncomparative design intended to reflect real-world clinical use. A total of 100 consecutively treated patients were included, with assessments conducted between January 2023 and July 2025 in a single tertiary care facility in Houston, Texas. The primary aim of the study was to document cutaneous safety and tolerability following topical pHA use; the study was not designed to evaluate wound healing efficacy or antimicrobial outcomes. Additional observations related to wound healing were descriptive in nature. Patients were followed until wound closure, discharge home (with wound care subsequently managed by parents or caregivers), or transfer to another facility for non–wound-related care needs.
Study population and eligibility criteria
Patients were identified from consultations requested for wound care management in the NICU and the pediatric intensive care unit (PICU).
Eligibility criteria included infants younger than 12 months admitted to the NICU or PICU with wounds classified as complex, defined as open wounds requiring active management and repeated assessments beyond routine neonatal skin care. The frequency of bedside assessment and treatment varied depending on the nature and severity of the wound. Some patients required daily assessment and thus, daily application of pHA, while others required less frequent interventions.
Complex wounds were categorized as follows: intravenous extravasation injuries, surgical wounds, skin tears, pressure injuries, skin and soft tissue infections, diaper dermatitis with skin breakdown, congenital skin anomalies, and miscellaneous wounds (eg, traumatic delivery-
related injuries and contact dermatitis).
Only patients who underwent more than 1 bedside wound care encounter and received pHA application on more than 1 occasion were included.
Exclusion criteria included age greater than 12 months, wounds not meeting the definition of “complex,” receipt of only a single pHA application, incomplete medical records, or loss to follow-up before a second wound care assessment. No exclusions were applied based on comorbidities or concurrent therapies.
Masking and bias considerations
Masking was not feasible because (1) the intervention required intentional application of pHA by the treating provider and (2) parents and guardians were necessarily aware of bedside interventions. To mitigate bias, all wound care interventions and clinical assessments were performed exclusively by 2 dedicated wound care specialists (R.A. and E.H.), ensuring consistency in technique and documentation.
Data collection
Demographic and clinical data were extracted from the electronic medical record, including gestational age at birth, chronological age at first pHA application, the number of applications per patient, duration of pHA use per patient, sex, weight, wound type, wound location, and wound size. Wound size was approximated in square centimeters by multiplying the greatest length by the greatest width, measured with a disposable bedside guide.
Data on concurrent NICU therapies, such as phototherapy, incubator or crib type, and environmental humidification, were extracted given their potential relevance to wound healing or interaction with cleansing practices. Concurrent wound care modalities were also recorded: medical-grade honey, extracellular matrix dressings, NPWT, skin grafts, and cyanoacrylate skin barrier.
All procedures, assessments, and documentation were performed by only 2 providers (R.A. and E.H.), ensuring consistency and standardization of care and evaluations. Clinical documentation was based on contemporaneous progress notes authored by the treating providers, further minimizing variability in terminology and outcome reporting.
pHA treatment protocol
Application of pHA was standardized. For each treatment, 10 mL to 20 mL of solution was drawn into a sterile syringe immediately before use. The solution was applied to sterile gauze and placed directly on the wound bed for 1 minute without mechanical force. For cavitary or recessed wounds, direct irrigation was performed using gentle syringe instillation. The 1-minute application time was selected to balance wound contact with minimizing procedural stress in preterm and neonatal patients.
Application of pHA was initiated at the first wound care assessment and repeated with each subsequent hospital-based wound assessment and dressing change. The frequency of wound assessments and thus, pHA application, varied according to the clinical need, and not all patients underwent daily wound evaluations. In extremely preterm infants, direct hands-on care was limited to reduce stress and maintain safety. In other cases, the wound management approach (eg, NPWT) did not require daily dressing changes. Consequently, the mean number of pHA applications per patient differed from the mean overall duration of pHA use, reflecting variation in wound type, patient fragility, and frequency of dressing changes. Application continued until wound closure, discharge home (with wound care managed by parents or caregivers), or transfer to another facility for non–wound-related care needs. All hospital-based wound assessments and pHA applications were performed by the 2 study investigators, ensuring consistency in technique and documentation.
Ethical considerations
Approval was obtained from the University of Houston Institutional Review Board (IRB number STUDY00004982) prior to initiation of data collection. The retrospective design qualified for a waiver of informed consent, because only previously documented clinical data were analyzed and no interventions outside standard care were introduced. The study was conducted in accordance with the Declaration of Helsinki and applicable local regulatory requirements.
Statistical analysis
Given the retrospective, descriptive design, no formal hypothesis testing was performed. Data were summarized in terms of frequencies, distributions, and clinical observations. Continuous variables (eg, wound size, weight) were reported as means and ranges, and categorical variables were summarized as counts and percentages. Analyses were exploratory and descriptive only, considering the noncomparative design of this study.
Results
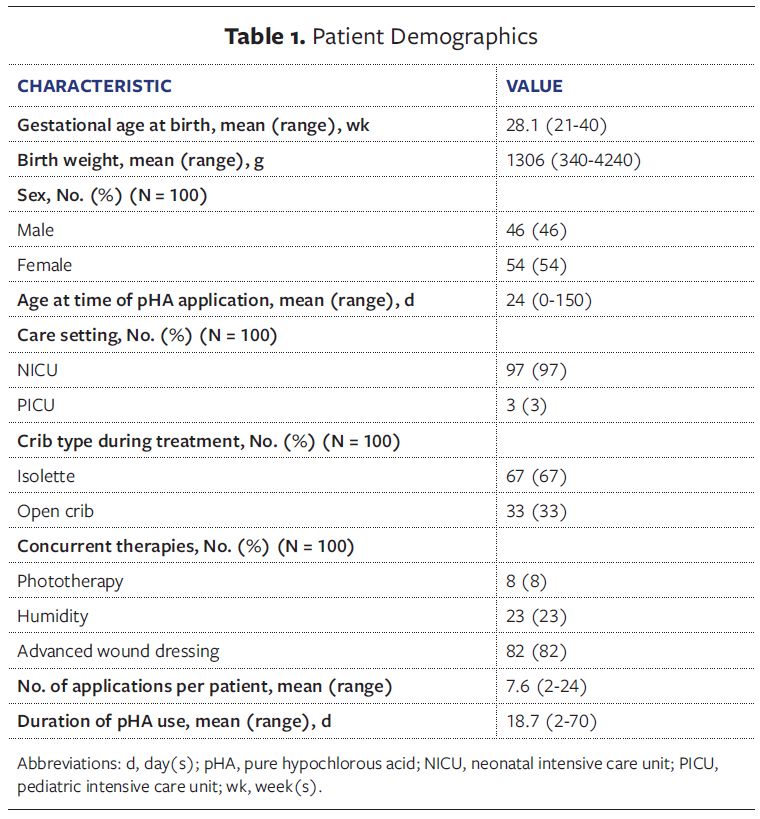
A total of 100 consecutive patients met the inclusion criteria, and the study population was well balanced between male and female patients (46 [46%] and 54 [54%], respectively). The mean gestational age at birth was 28.1 weeks (range, 21 weeks–40 weeks), with a mean birth weight of 1 306 g (range, 340 g–4 240 g). The youngest treated infant was born at 21 weeks’ gestation. The mean chronological age at treatment initiation was 24 days (range, 0 days–150 days). Most patients (97%) were admitted to the NICU, with 3% admitted to the PICU. Two-thirds of patients (67%) were managed in isolettes, and one-third (33%) were managed in open cribs. The mean duration of treatment was 18.7 days (range, 2 days-70 days) (Table 1, Figure 1).
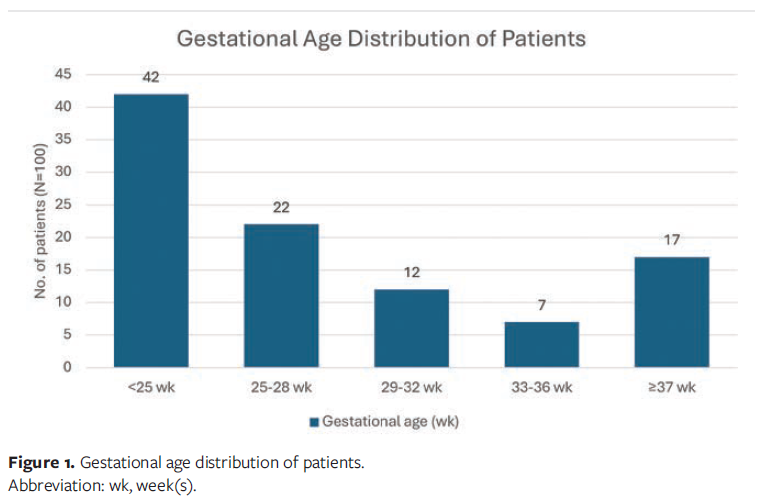
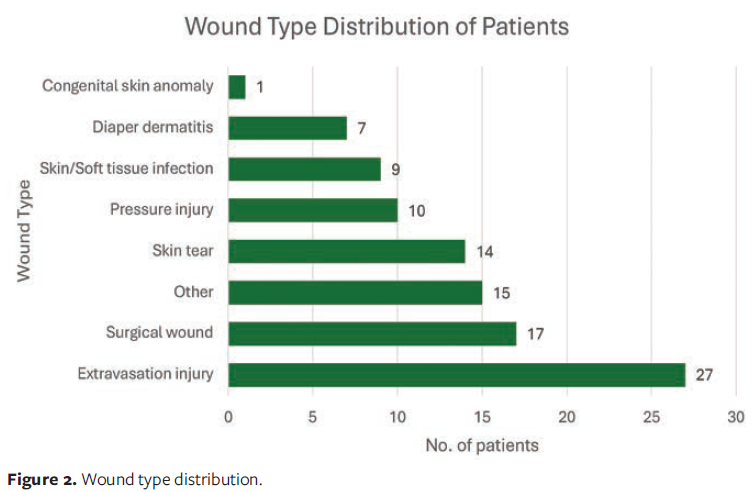
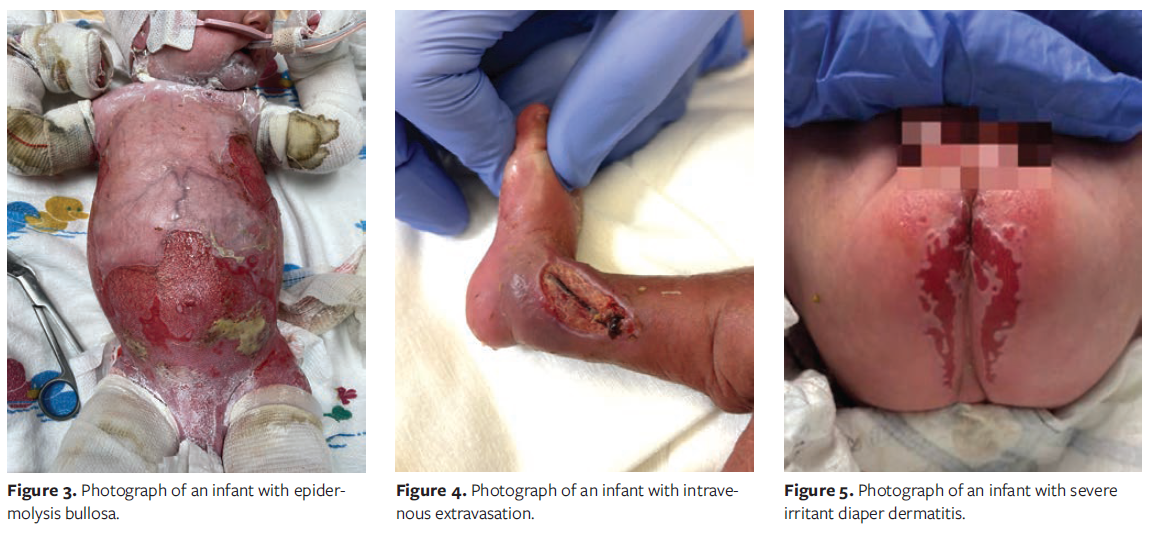
The mean wound size (length × width) at baseline was 5.25 cm² (range, 0.04 cm²–80 cm²). The most common wound etiologies were extravasation injury (27%) and surgical wounds (17%). The least common etiologies included diaper dermatitis with skin breakdown (7%) and congenital skin anomalies (1%). Additional etiologies included skin tears, pressure injuries, and skin and soft tissue infections, as well as miscellaneous wounds such as traumatic delivery–related injuries and contact dermatitis (Figure 2). Representative examples of these complex wounds are shown in Figures 3–5.
Primary objective
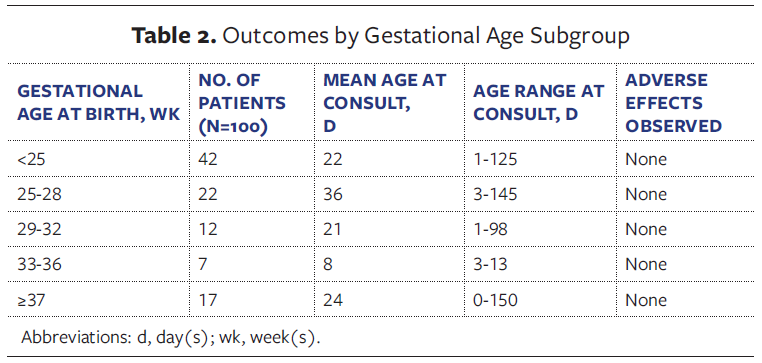
Across 766 cumulative applications of pHA (mean 7.6 applications per patient), no cutaneous adverse effects or wound-related complications were identified. Patients who received only a single treatment were excluded to ensure tolerance was evaluated under repeated exposure. Specifically, no cases of contact dermatitis, wound infection, chemical burns, erythema, or secondary breakdown were observed across all wound types and gestational age categories. The largest subgroup of treated patients consisted of infants born at less than 25 weeks’ gestation (n = 42 [42%]), underscoring that even the most preterm neonates tolerated repeated pHA exposure without incident (Table 2).
pHA was also used in combination with other advanced wound therapies, including medical-grade honey (66 patients), extracellular matrix dressings (32 patients), NPWT (17 patients), skin grafts (9 patients), and cyanoacrylate skin barrier (3 patients). In these cases, wounds progressed toward closure during the course of care, and no cutaneous adverse effects attributable to the concurrent use of pHA with these modalities were observed.
Discussion
This retrospective study represents the largest documented evaluation to date of cutaneous safety associated with pHA use in preterm infants and neonates with complex wounds. Across 100 patients and 766 cumulative applications, cutaneous tolerability of pHA was observed for all wound types, including in infants born as early as 21 weeks’ gestation, with no cutaneous adverse effects or wound-related complications identified (Table 2). Notably, the largest subgroup consisted of infants born at the threshold of viability (<25 weeks’ gestation), further emphasizing the relevance of these findings in the most preterm patient population. Collectively, these observations suggest that pHA use was not associated with cutaneous adverse effects in this cohort, supporting its tolerability when applied to immature and compromised neonatal skin.
Managing wounds in preterm infants is particularly challenging due to the immaturity of both the skin barrier and immune defenses. Current cleansing options are limited; although normal saline is widely used for its perceived cutaneous safety, it provides little evidence-based cleansing or bioburden-reducing effect.22,23 Conversely, povidone-iodine–, chlorhexidine-, and sodium hypochlorite–based solutions are potent surface disinfectants but have demonstrated cytotoxicity in open wounds.24 Clinically, their use has been associated with adverse outcomes, including delayed healing, chemical burns, thyroid suppression with iodine, and skin necrosis with chlorhexidine in preterm infants.18,19 Within this context, the absence of cutaneous adverse effects in the cohort in the present study suggests that pHA may represent a clinically meaningful alternative, consistent with reports in adult populations describing cutaneous tolerability across a range of wound types.12-17 By extending these safety findings to preterm and neonatal patients, whose skin barrier and immune function differ substantially, the present series helps fill an important evidence gap. Based on the favorable clinical experience of the authors of the present study, pHA was adopted as the standard of care for the management of complex wounds in the NICU and PICU at the study institution.
Importantly, these results align with prior pediatric case reports describing cutaneous safety and tolerability of pHA in children with burns and complex wounds.18-21 In vitro and clinical data also support its favorable cytotoxicity profile compared with traditional wound cleansing solutions with antimicrobial ingredients,25,26 a finding initially emphasized by Schultz et al,25 who highlighted pHA’s improved cutaneous safety profile relative to other agents. While the current study was not designed to assess efficacy, no cutaneous adverse effects were observed in patients receiving pHA concurrently with other advanced wound therapies (ie, honey, collagen dressing, NPWT, skin graft).
The current series provides novel evidence describing cutaneous safety and tolerability of pHA use in fragile neonatal populations for whom alternative cleansing options are limited. These findings, along with supportive adult and pediatric literature, highlight the need for future prospective studies to more clearly define cutaneous safety and to evaluate efficacy outcomes in neonatal wound care.
Limitations
Several limitations should be considered when interpreting these findings. The retrospective, single-center design may limit generalizability, and the absence of a comparator group precludes assessment of relative efficacy. The study was not designed to evaluate wound healing efficacy, bioburden reduction, or antimicrobial effects. Wound healing outcomes were not formally quantified beyond descriptive follow-up, reflecting the primary focus on cutaneous safety and tolerability. Nevertheless, the consecutive sampling approach, consistency of provider assessments, and inclusion of a broad range of wound types strengthen the internal validity of the results.
Conclusion
In this series of 100 preterm infants and neonates, encompassing 766 cumulative applications of pHA, no cutaneous adverse effects or wound-related complications were observed, including in an infant born at 21 weeks’ gestation. These findings provide the strongest evidence to date that pHA is a safe and well-tolerated wound cleanser in this highly vulnerable population. Future prospective, multicenter studies are warranted to further characterize cutaneous safety and to evaluate efficacy outcomes in neonatal wound care.
Author and Public Information
Authors: Rene Amaya, MD; and Emily Heisler, MSN, RN, FNP-C
Affiliations: Pediatrix Neonatology of Houston, Houston, TX, USA; Department of Pediatrics, The Woman’s Hospital of Texas, Houston, TX
Disclaimer: The views expressed in this article, based on our analysis and manuscript preparation, are solely those of the authors and do not represent the official position of the institution.
Disclosure: The authors declare no conflicts of interest and received no external funding for this study.
Ethical Approval: Approval was obtained from the University of Houston Institutional Review Board (IRB number STUDY00004982). The requirement for informed consent was waived, because only previously documented clinical data were analyzed and no interventions outside standard care were introduced.
Acknowledgments: Both authors contributed to the conception and design of the study, participated in data acquisition and management, performed the data analysis and interpretation, and were responsible for drafting the manuscript.
Correspondence: Rene Amaya, MD; Pediatrix Neonatology of Houston, 7400 Fannin St, Suite 750 Houston, TX 77054; rene.amaya@pediatrix.com
Manuscript Accepted: February 9, 2026
References
- Visscher MO, Carr AN, Narendran V. Epidermal immunity and function: origin in neonatal skin. Front Mol Biosci. 2022;9:894496. doi:10.3389/fmolb.2022.894496
- Visscher MO, Hu P, Carr AN, et al. Newborn infant skin gene expression: remarkable differences versus adults. PLoS One. 2021;16(10):e0258554. doi:10.1371/journal.pone.0258554
- Choi EH. Skin barrier function in neonates and infants. Allergy Asthma Immunol Res. 2025;17(1):32-46. doi:10.4168/aair.2025.17.1.32
- Ji F, Li D, Lyu T, Yang T, Yuan H, Huang X, Hu X. Iatrogenic skin injuries in infants admitted to neonatal intensive care units: An investigation in 22 Chinese units. J Tissue Viability. 2024;33(2):197-201.
- Mulinda C, Manthalu P, Nkhoma D, et al. Pre-procedural topical antisepsis in the neonate: A systematic review evaluating risk factors for skin injury. Int J Nurs Stud. 2025;149:104623.
- Fassino B, Ferrario S, Sorrentino G, et al. Hospital-acquired skin lesions in the neonatal intensive care unit: a retrospective analysis of temporal trends and quality improvement strategies. J Pediatr Nurs. 2023;70:40-46. doi:10.1016/j.pedn.2023.01.009
- Marty D, Sorum K, Smith K, Nicoski P, Al Sayyed B, Amin S. Nosocomial infections in the neonatal intensive care unit. Neoreviews. 2024;25(5):e254-e264. doi:10.1542/neo.25-5-e254
- Boyar V, Visscher M, Lund C, Narendran V; Tiny Baby Collaborative Steering Committee. Seeing beyond the obvious: pragmatic skin care guidance for infants 22-24 weeks gestational age. J Perinatol. 2025. doi:10.1038/s41372-025-02375-1
- Mulinda C, Suhail S, Sutherland B, Lauren CT, Hunt RD. Pre-procedural topical antisepsis in the neonate: a systematic review evaluating risk factors for skin injury. Pediatr Dermatol. 2025;42(1):31-40. doi:10.1111/pde.15773
- Sathiyamurthy S, Banerjee J, Godambe SV. Antiseptic use in the neonatal intensive care unit—a dilemma in clinical practice: an evidence based review. World J Clin Pediatr. 2016;5(2):159-171. doi:10.5409/wjcp.v5.i2.159
- McCord H, Fieldhouse E, El-Naggar W. Current practices of antiseptic use in Canadian neonatal intensive care units. Am J Perinatol. 2019;36(2):141-147. doi:10.1055/s-0038-1661406
- Oropallo A, Rao AS, Del Pin C, Ranire-Maguire M, Mathew A. An objective comparative study of non-surgical cleansing techniques and cleanser types in bacterial burden management. Int Wound J. 2024;21(2):e14730. doi:10.1111/iwj.14730
- Fligor CW, Duplechain AB, Chaffin AE. A 12-month retrospective review of pressure injury plastic surgical flap techniques utilizing pure hypochlorous acid (pHA) preserved wound solution for wound bed preparation. Wound Manag Prev. 2024;70(4). doi:10.25270/wmp.24001
- Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78(9):1461-1466. doi:10.1016/j.joms.2020.06.029
- Gold MH, Andriessen A, Bhatia AC. Topical stabilized hypochlorous acid: the future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020;19(2):270-277. doi:10.1111/jocd.13280
- Mayer DO, Tettelbach WH, Ciprandi G, Downie F, Hampton J, Hodgson H, et al. Best practice for wound debridement. J Wound Care. 2024;33(6 Suppl C):S1-S32.
- Boecker D, Zhang Z, Breves R, Herth F, Kramer A, Bulitta C. Antimicrobial efficacy, mode of action and in vivo use of hypochlorous acid (HOCl) for prevention or therapeutic support of infections. GMS Hyg Infect Control. 2023;18:Doc07. doi:10.3205/dgkh000433
- Elsass FT. Adjunctive debridement with hypochlorous acid leads to a healing trajectory of complex wounds in children. Ostomy Wound Manage. 2016;62(4):8-10.
- Faust E. Wound cleansing with a hypochlorous acid–preserved wound cleanser in pediatric patients with burns. Wound Manag Prev. 2022;68(2):8-10.
- Boyar V. Hypochlorous acid in pediatric wound management: from cleansing to healing wounds. Wound Manag Prev. October 2020. Accessed March 3, 2026. https://www.hmpgloballearningnetwork.com/site/wmp/article/hypochlorous-acid-pediatric-wound-management-cleansing-healing-wounds.
- Marshall-Hanson C. Healing complex wounds and skin conditions in pediatric patients using a pH-controlled cleanser containing hypochlorous acid. Wound Manag Prev. 2020. Accessed March 3, 2026. https://www.hmpgloballearningnetwork.com/site/wmp/article/healing-complex-wounds-and-skin-conditions-pediatric-patients-using-ph-controlled-cleanser.
- Fernandez R, Griffiths R, Ussia C. Water for wound cleansing. Cochrane Database Syst Rev. 2022;(4):CD003861. doi:10.1002/14651858.CD003861
- Ringblom A, Ivory J, Adlerberth I, Wold AE, McIntosh C, Wolf A: Wound cleansing solutions versus normal saline in the treatment of diabetic foot ulcers - a systematic review. J Tissue Viability. 2024;33(4):591-597. doi:10.1016/j.jtv.2024.08.010
- Ortega-Llamas L, Quiñones-Vico MI, García-Valdivia M, et al. Cytotoxicity and wound closure evaluation in skin cell lines after treatment with common antiseptics for clinical use. Cells. 2022;11(9):1395. doi:10.3390/cells11091395
- Schultz GS, Sibbald RG, Falanga V, et al. Wound bed preparation: a systematic approach to wound management. Wound Repair Regen. 2003;11(Suppl 1):S1-S28. doi:10.1046/j.1524-475x.11.s2.1.x
- Tsai CF, Chung JJ, Ding SJ, Chen CC. In vitro cytotoxicity and antibacterial activity of hypochlorous acid antimicrobial agent. J Dent Sci. 2024;19(1):345-356. doi:10.1016/j.jds.2023.07.007

















