The STEADY Diabetic Foot Ulcer Registry: Methods, Insights, and Future Directions
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Diabetic foot ulcers (DFUs) are a major cause of morbidity, amputation, and mortality among individuals with diabetes, with disproportionate impact on underserved populations. Comprehensive real-world data on DFU management and outcomes are lacking. Objective. To describe the design and methodology of the STEADY (Structured Evaluation and Analysis of Diabetic Foot Ulcers in the US) registry, a national prospective cohort study of patients with DFUs in the United States whose objective is to evaluate DFU treatment patterns, outcomes, and health care resource utilization in real-world settings, to assess comparative effectiveness, cost effectiveness, and safety of DFU therapies and therapy combinations, and to advance disease management through risk- and site-stratified treatment optimization models. Methods. STEADY is a 10-year prospective multicenter observational study with an aim of enrolling 5000 adults with active DFUs in the United States. Data sources include electronic case report forms, electronic medical records (EMRs), patient-reported outcomes via mobile app, and optional insurance claims. Primary and secondary end points will include time and incidence of partial and complete wound closure; wound and disease characteristics; rates of recurrence, infection, ischemic events, and amputation; health care utilization, including surgical procedures; health-related quality of life; work productivity; and additional patient reported outcomes. Descriptive, survival, and comparative effectiveness analyses will be performed. Data governance ensures full regulatory compliance and robust data security and integrity, supporting the potential use of the registry dataset as a synthetic control arm in future clinical research. Conclusion. STEADY leverages an artificial intelligence (AI)-enabled platform to integrate multisource data, including wound photography, social determinants of health, patient reported outcomes and caregiver information. The platform uses AI for transcription and interpretation of patient and provider dictation, supports patient-controlled EMR synchronization for comprehensive longitudinal tracking across providers, offers participant incentives to enhance engagement, and ensures rigorous, automated data quality assurance at all stages.
Diabetic foot ulcers (DFUs) are among the most debilitating complications of diabetes, representing a major cause of morbidity, mortality, and health care costs worldwide. Globally, an estimated 589 million adults aged 20 years to 79 years live with diabetes.1 Up to one-third of people with diabetes will experience a DFU in their lifetime.2,3 Studies have found that 35% to 58% of DFUs develop an infection, and that of these, 9% to 17% will result in amputation within 1 year to 2 years.4-6
In the United States, diabetes prevalence in adults is 14.7% to 15.8% when combining diagnosed and undiagnosed cases, more than one-third higher than in rest of the world (11.1%).1,7 DFUs are the leading cause of nontraumatic lower extremity amputations in the United States.2 One study found that less than half of people with diabetes survived 5 years after a lower extremity amputation, and less than one-fourth survived 10 years.8 Not only are these outcomes devastating to patients and caregivers, but they are also associated with significant health care utilization and economic burden.9
The human impact of DFUs extends far beyond clinical complications. Studies have shown that patients with diabetes fear the loss of a limb more than death itself.10 Amputations disproportionately affect underserved populations, including Black, Hispanic, and Native American individuals, who are 2 to 4 times more likely to experience limb loss compared with White individuals.11-13 Socioeconomic status and geography further compound these disparities, with higher amputation rates among patients insured by Medicaid, those living in rural settings without specialist access, and residents of safety net urban neighborhoods.12,14 These inequities highlight the urgent need for real-world data (RWD) and innovative approaches to improve DFU management, prevention, and outcomes.
Challenges in Current DFU Research and Care
Despite decades of clinical research, the overall prognosis for DFUs remains poor. Healing rates are variable, recurrence is common, and mortality following amputation rivals that of many cancers.15,16 The heterogeneity of patients with DFU and their comorbidities complicates both clinical management and research. Randomized controlled trials (RCTs) are often limited by narrow inclusion criteria, modest sample sizes, and a focus on short-term outcomes.17 Consequently, the findings from these trials may not translate effectively to routine practice, especially in underserved and diverse patient populations.
The importance of RWD in health care research is well recognized, but significant challenges persist in accessing comprehensive, high-quality databases (DBs) for analyzing chronic wound care. Primary research on incidence and prevalence rates is often limited by (1) outdated data, with research conducted more than 20 years ago; (2) use of data from a single metro area; (3) use of data collected at a single site of care; and (4) small sample size. The Wound Care Collaborative Community (WCCC), through its Real-World Evidence Work Group, identified a limited number of DBs suitable for its natural history project, highlighting the need for health care systems to develop more accessible and usable DBs that capture the full patient journey and wound care outcomes.18
Existing DFU registries in the United States, such as the U.S. Wound Registry (USWR)19-21 and the National Institutes of Health-sponsored Diabetic Foot Consortium (DFC),22 as well as international efforts such as the Tampere Wound Registry in Finland,23 have advanced the field but exhibit key limitations. Weaknesses include a reliance solely on structured data from electronic health records (EHRs), lack of standardized patient-reported outcomes (PROs), lack of validated wound care classification measures such as the Wagner, University of Texas (UT), or wound, ischemia, and foot infection (WIfI) systems, a failure to capture longitudinal follow-up beyond the episode of care, geographical limitation to a single region, or limitation to a single site of care. Moreover, few registries incorporate real-world imaging, social determinants of health (SDOH), or advanced analytics that support personalized medicine. These gaps create barriers to understanding the full spectrum of DFU care and hinder (1) the development of predictive models to support clinical decision-making, (2) the identification of unmet needs and opportunities for wound product innovation, (3) the creation of robust synthetic control arms to support clinical research, (4) the understanding of effect of therapy on long-term recidivism and the effect of wound closure on mortality, and (5) the ability for health care payers to assess cost-effectiveness and comparative effectiveness of therapeutic treatment options.
The Rationale for STEADY
The protocol for STEADY (Structured Evaluation and Analysis of Diabetic Foot Ulcers in the U.S.) was established to address existing gaps and develop a comprehensive, scalable, and patient-centered data infrastructure. Originating as a key consensus recommendation of the WCCC Driving Innovation Summit held in conjunction with the Symposium on Advanced Wound Care (SAWC) Spring 2024, in Orlando, FL, this initiative brings together experts from the US Food and Drug Administration (FDA), the Centers for Medicare and Medicaid Services (CMS), industry, and academia to overcome barriers and accelerate innovation in wound care.18 The STEADY registry is designed to integrate multisource RWD to advance the understanding, prevention, and management of DFUs as a collaboration between FDA collaborative community WCCC and clinical research organization (CRO) Alira Health.
Key drivers behind the development of STEADY include the need for comprehensive data capture, inclusion of underrepresented groups, advancement of analytics, support for policy and quality improvement, and the potential to develop synthetic control arms.
Need for comprehensive data capture
Current registries often miss critical patient-level details such as percentage area reduction, time to total wound closure, wound classification, imaging, PROs, and longitudinal follow-up. STEADY captures multimodal data from clinical sites, patient-initiated EHR connections, PROs, wound imaging, and claims sources to create a robust, unified dataset. Whereas most registries collect data at only 1 site of care, STEADY will enlist sites across varied locations of DFU care and will collect patient-generated data to fill in any gaps in site-generated data.
Equity and inclusion
Given the disproportionate effect of DFUs on marginalized populations, STEADY incorporates intentional site selection and patient engagement strategies to ensure representation of diverse communities, including rural, safety net, and minority populations. The study’s scientific committee will monitor STEADY’s inclusion of underrepresented groups and adjust study operations as necessary to ensure representativeness.
Advancement of analytics
By leveraging artificial intelligence (AI) and machine learning pipelines, STEADY supports predictive modeling for risk stratification, outcome forecasting, cost comparison summary, and clinical decision support. It also enables the creation of synthetic control arms to enhance the interpretability and efficiency of interventional studies.
Support for policy and quality improvement
Regulatory bodies, including the FDA and CMS, increasingly value real-world evidence (RWE) in regulatory decision-making, leading to greater demand for RWD within wound care.24 STEADY’s data infrastructure is aligned with national standards (Observational Medical Outcomes Partnership [OMOP])25 to facilitate collaboration with regulators, payers, and quality organizations.
Synthetic control arm development
The registry’s comprehensive and standardized dataset allows for the creation of synthetic control arms to support clinical trials. Matching algorithms may use baseline characteristics, ulcer severity, and comorbidities to generate comparable cohorts. This approach reduces the need for placebo arms in interventional studies, accelerates timelines, and lowers costs.26,27
Objectives of the STEADY Registry
The primary objective of the STEADY registry is to conduct comparative effectiveness studies of DFU therapies in real-world settings, including various sites of real-world care. The secondary objectives of the STEADY registry are to assess the real-world and long-term safety and effectiveness of contemporaneous and innovative DFU therapies, especially those approved in the past 5 years that have had less opportunity for the accumulation of evidence; to characterize the DFU patient and treatment journey or journeys, including if there is a standard of care, and where the patients are seen for care; understand the burden of illness of DFU, including on patients’ quality of life; to evaluate the epidemiology of DFU, including short- and long-term outcomes; and to assess the cost-effectiveness of DFU therapies. A comprehensive list of research objectives, including exploratory objectives, is detailed in Appendix 1.
DFUs represent a significant clinical and public health challenge, with persistent gaps in evidence and limited RWD to guide policy and practice. The STEADY registry addresses these challenges by providing a comprehensive, scalable, and patient-centered platform for evidence generation, predictive analytics, and quality improvement. Through its multisource data architecture, equity-focused design, and advanced analytics, STEADY is uniquely positioned to transform DFU research and improve outcomes for patients across the United States. The aim of publishing the STEADY protocol (research question, eligibility, methods, and reporting outcomes) is to minimize the potential for bias, reinforce confidence in data integrity and validity, raise awareness, and facilitate peer feedback.
Methods
Study design
The STEADY registry is a prospective, multicenter, observational cohort study designed to capture comprehensive RWD on patients with DFUs, which represents a novel, innovative project in the field of limb preservation. As noted previously, a comprehensive list of research objectives is detailed in Appendix 1.
Study population
Patients aged 18 years and older with a confirmed diagnosis of a DFU are eligible for inclusion. Both incident and prevalent DFU cases are enrolled across diverse clinical settings, including acute care hospitals, wound care centers, private podiatry offices, primary care settings, and endocrinology practices, as well as through decentralized direct-to-patient recruitment. The inclusion of varied care environments ensures broad representativeness and enhances the generalizability of findings. Exclusion criteria include primary health insurance or DFU care managed in a country other than the United States, inability to provide consent, and malignancy at the wound site.
Recruitment and compliance
Participating sites use a combination of automated EHR screening and manual chart review to identify eligible patients. Patients provide institutional review board (IRB)-approved informed consent either electronically or in person, in compliance with IRB and Health Insurance Portability and Accountability Act (HIPAA) regulations. All participating sites adhere to the principles of the Declaration of Helsinki and Good Clinical Practice guidelines. Research records will be retained for 15 years following study completion. Participant-level research data will be maintained in encrypted, access-restricted systems with full audit logging. AI-enabled data processing will occur within secure, HIPAA-compliant environments; no participant personal health information will be used to train or refine external models. Patients may withdraw consent at any time, which will result in no further contact or data collection. De-identified analytic datasets may be retained for secondary research use as permitted by the informed consent and applicable US regulations.
Data collection framework
Data are collected longitudinally at baseline and at follow-up intervals. The registry leverages structured data capture aligned with the OMOP Common Data Model to ensure interoperability and regulatory readiness. The full list of data variables and schedule of assessments are detailed in Supplement 1 and Supplement 2, respectively.
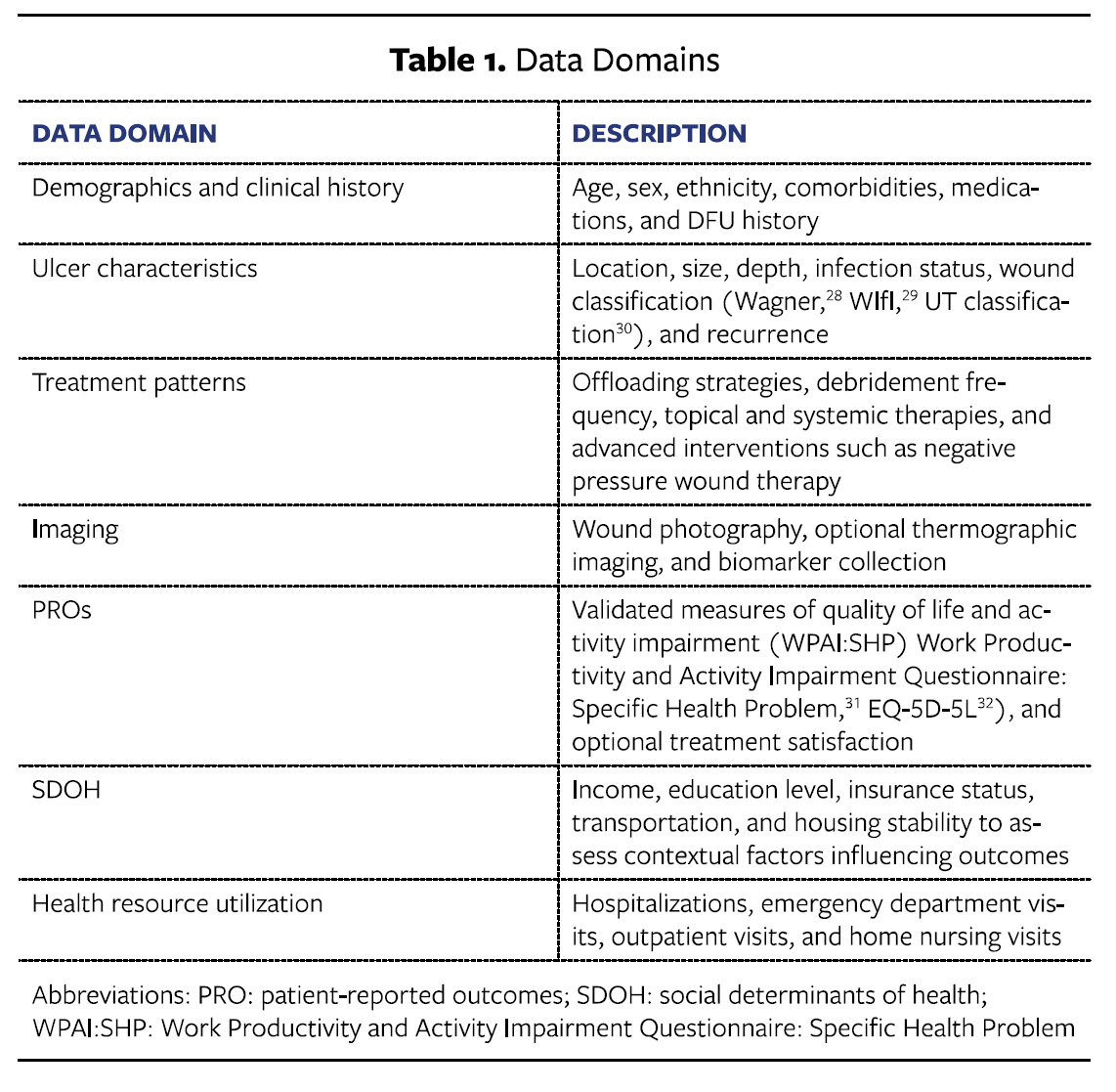
Data domains
Table 1 summarizes the core data domains collected in this study, encompassing patient characteristics, ulcer features, treatment patterns, and outcomes. These domains are designed to provide an assessment of clinical status, care delivery, and factors influencing diabetic foot ulcer outcomes.
Digital health integration
STEADY uses a web portal and patient-facing mobile application that enables participants to synchronize their EHRs via secure application programming interfaces, upload home-monitored data, such as wound photos and step counts, and complete PRO assessments between clinical visits. The system is designed to combine active and passive data collection in a way that minimizes complexity and patient burden.33 The data source integration enhances data granularity and minimizes missing data while promoting patient engagement in self-management.34
Quality assurance and data governance
Data undergo automated and manual validation checks to ensure accuracy and completeness. Discrepancies are flagged for site-level resolution. Governance is overseen by a steering committee composed of clinicians, data scientists, patient representatives, and regulatory advisors to ensure transparency and compliance.35
Statistical analysis plan
The registry aims to enroll 5000 patients over 10 years to enable robust subgroup analyses and predictive modeling. Analyses will be performed approximately on a yearly basis and will include descriptive statistics, Cox regression modeling, and time-to-event analyses for the primary outcome of time to total wound closure, as well as recurrence. Continuous efficacy outcomes will be summarized by weeks as observed values and change from baseline. All analysis will be presented overall and for subgroups of interests. Additional exploratory factorial analysis will be done to determine additional patient clustering. An overview adverse event (AE) table containing at least the number of patients with AEs, serious AEs, and severe AEs will be produced. Each individual sponsor retains the ability to independently perform analyses that may not be specified here.
Propensity score matching and causal inference modeling with sensitivity analyses may be done to adjust for confounders and to estimate treatment effects.36,37 For predictive analytics, machine learning algorithms, including gradient boosting machines and recurrent neural networks, can be trained on de-identified datasets. Performance is assessed using cross-validation and independent test sets, with metrics including area under the curve, calibration plots, and decision curve analysis.38
The precise methods used will depend on the biases detected, but a statistical plan for comparative effectiveness from the registry data is described here.
Default comparative effectiveness analytic strategy for STEADY DFU analyses
Target trial emulation. Analyses will be structured to emulate an RCT using a new-user, active-comparator design to ensure comparability of treatment groups at initiation.27
Propensity score estimation with flexible machine learning. Propensity scores will be estimated using flexible learners, such as gradient boosting algorithms, or ensemble approaches like the super learner.39 These methods improve covariate balance compared with conventional logistic regression.40
Weighting for covariate balance. Overlap weighting or stabilized inverse probability of treatment weighting (IPTW) will be applied to balance baseline covariates.41 Covariate balance will be assessed using standardized mean differences, with poor overlap addressed by trimming noncomparable patients.42
Doubly robust estimation. Treatment effects will be estimated using augmented inverse probability weighting, which combines outcome and treatment models for robustness.43 Targeted maximum likelihood estimation will be conducted as a sensitivity analysis to confirm robustness.44
Time-varying treatments and confounding. For exposures or confounders that change over time (eg, use of different off-loading devices, infection status), marginal structural models with time-varying IPTW will be used.45
Censoring and missing data. Loss to follow-up will be handled with inverse probability of censoring weighting.46 Missing data will be addressed through multiple imputation by chained equations,47 supplemented by sensitivity analyses under different missing data assumptions.
Negative control analyses. Negative control outcome and exposure analyses will be used to detect unmeasured confounding.48 E-values will be calculated to assess the minimum strength of association an unmeasured confounder would need to explain away the observed results.49
Competing risks analysis. In the presence of competing outcomes (eg, amputation vs. death), Fine–Gray subdistribution hazard models50 50 and cause-specific Cox proportional hazards models51 will be applied.
Heterogeneity of treatment effects. Variation in treatment response will be explored using causal forests52 and Bayesian additive regression trees.53 These models allow estimation of individualized treatment effects.
Instrumental variable analyses. Where valid instruments exist (eg, site-level prescribing preferences), instrumental variable (IV) analyses such as 2-stage least squares will be applied. Sensitivity analyses will evaluate potential violations of IV assumptions.
Discussion
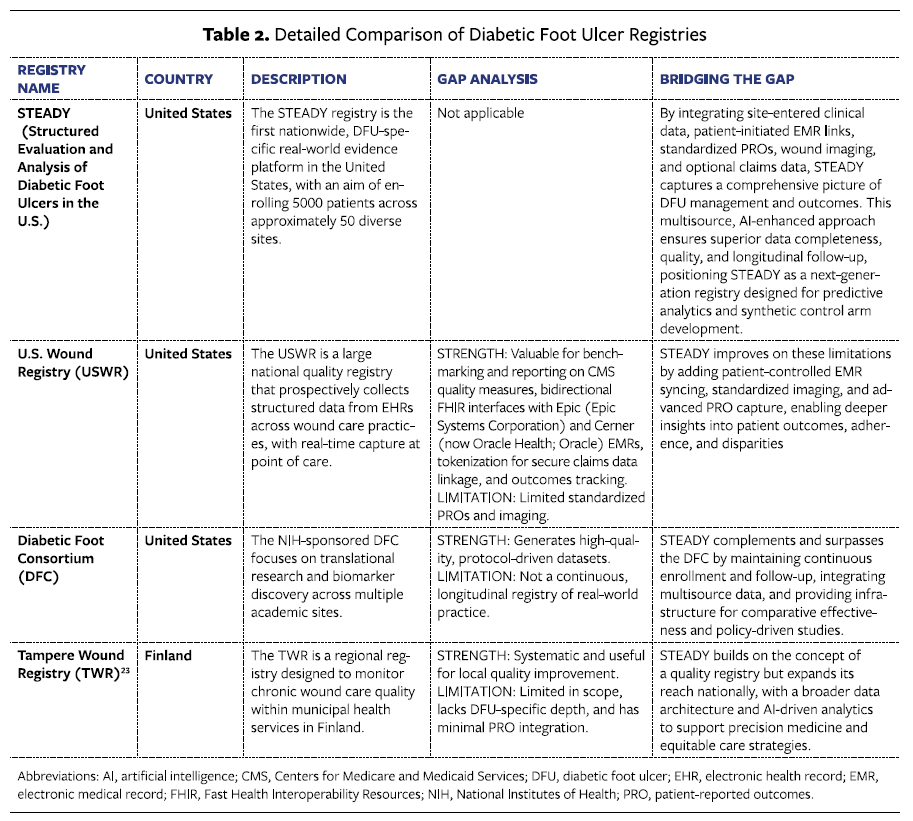
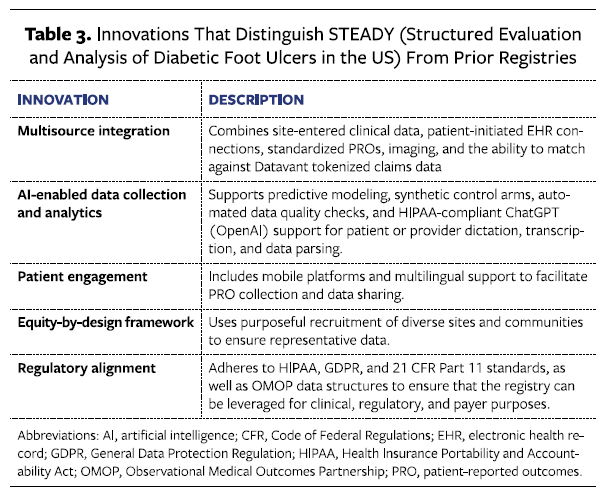
The STEADY registry addresses gaps in existing DFU registries, whose strengths and limitations are summarized in Table 2,19-21,23,54,55 by combining a US-based national scope, multisource data integration, advanced analytics, and unique patient and provider engagement and retention features in the CRO’s technology platform (Table 3). Whereas the USWR primarily captures electronic medical record (EMR) data, STEADY integrates patient-initiated EMR connections, standardized PROs facilitated by active patient engagement teams, AI agents to assist in provider data collection (clinician-reported outcomes [ClinROs], and wound imaging for a more holistic view of DFU care. The DFC, as well as international efforts such as the Tampere Wound Registry, provide valuable research insights but lack the continuous RWE framework and synthetic control arm readiness embedded in STEADY. This positions STEADY as a foundational resource for comparative effectiveness research.
One of STEADY’s defining features is its capacity for advanced predictive modeling using AI and machine learning, supported by the evidence generation and data analytics team at the CRO involved in this project. Predictive algorithms can help clinicians better understand patient journeys and support more complicated treatment plans. The registry’s large, well-characterized cohort facilitates the creation of synthetic control arms for interventional research, a critical innovation in wound care studies, in which recruitment and randomization can be challenging. Synthetic controls have been shown to reduce trial costs, accelerate timelines, and improve generalizability by using real-world cohorts to serve as comparators.36
This approach aligns with recent FDA guidance on RWE, which encourages the use of high-quality, structured data to support regulatory decision-making.56 The STEADY registry has been intentionally designed to be “fit for purpose,” in accordance with FDA draft guidance. Specifically, the registry ensures that data are both relevant—capturing all necessary outcomes, exposures, and covariates in a timely manner that reflects the current standard of care—and reliable. The data collection platform maintains full control over data provenance through a validated source system and robust audit trail, and strategies to address missing data, bias, and confounding are explicitly incorporated into the study design and analysis.
Finally, STEADY will also enable the use of RWE in payer decision-making. Use of RWE is already established for coverage decisions at the national level under CMS coverage with evidence (CED) guidance. Local coverage determinations (LCDs) issued by Medicare Administrative Contractors may also be moving toward increasing use of RWE in decision-making. Hospital purchasers place high value on RWE to assess clinical and economic value, and comparative effectiveness across product brands in routine practice. STEADY will enable generation of the quality, long-term RWE necessary for such purposes.
FDA regulatory considerations for future applications
The FDA has increasingly emphasized the use of RWE to support regulatory decision-making, particularly following the 21st Century Cures Act and subsequent guidance. High-quality observational data can provide important context for regulatory review by characterizing disease natural history, variability in clinical practice, and outcomes. For DFUs specifically, comprehensive natural history data helps FDA reviewers understand expected healing rates, recurrence patterns, and long-term outcomes across diverse patient populations, enabling more informed benefit–risk assessments for new therapies and devices (K. Dev Verma, MD, FDA Center for Drug Evaluation and Research, email, November 13, 2025).
The STEADY registry is designed to generate several forms of evidence aligned with these regulatory interests, including longitudinal effectiveness and safety outcomes, subgroup analyses, and durability of wound healing in real-world settings. The data generated may inform postmarket evaluations or supplement clinical trial evidence where appropriate. Any future regulatory use would require alignment with the FDA regarding end point definitions, data quality, and analytic approaches to ensure fitness for purpose.
CMS and Payer Considerations for Future Applications
In addition to its potential role in future FDA applications, the evidence generated from STEADY may also inform how Medicare and other payers evaluate DFU therapies in real-world practice. Although CMS does not review or comment on manuscripts, the agency has publicly described the types of evidence relevant to coverage, coding, and payment decisions. These considerations are often mirrored in commercial payer medical policies, technology assessments, and hospital value analyses, highlighting the broader utility of robust RWE in wound care.
Across both national coverage determinations and LCDs, CMS evaluates evidence across several domains: clinical meaningfulness of outcomes, including wound closure, infection resolution, amputation, and recurrence57,58; generalizability to Medicare beneficiaries, particularly older adults with multimorbidity, frailty, and social risk factors58,59; comparative effectiveness relative to available therapeutic alternatives and site-of-care variations57,58; durability of benefit and real-world adherence over time57,58; and effect on health care utilization, including hospitalizations, emergency department visits, and postacute care need.57,59
The STEADY registry is intentionally designed to incorporate many of these evidence domains. Its prospective, multisite data collection captures wound severity classifications, longitudinal outcomes, PROs, and SDOH. These features may enhance the relevance and generalizability of registry findings to real-world Medicare populations, addressing evidence gaps frequently cited in DFU LCDs and payer reviews. Moreover, STEADY’s analytic strategy including target trial emulation, propensity score–based weighting, and methods to address confounding and missing data aligns with methodological principles CMS has referenced in prior coverage deliberations and CED precedents.
Although the registry is not designed to fulfill all coverage requirements for any individual product, its national scope and analytic framework may help contextualize clinical and economic value, identify patient subgroups most likely to benefit from specific interventions, and inform future payer-facing or postmarket studies. As with regulatory applications, any payer-directed use would benefit from early alignment with the FDA and relevant stakeholders.
Limitations, Operational Challenges, and Future Directions
While STEADY offers substantial innovations, several challenges must be acknowledged. Data harmonization across diverse sources requires continuous quality assurance, and patient engagement for longitudinal follow-up remains resource intensive. In particular, the underserved patient population disproportionately affected by DFUs is also less likely to engage in research. WCCC’s leadership in selecting representative sites and the CRO’s dedicated patient engagement team and IRB approved patient and site financial remuneration (that are paid out automatically on milestone completion in the platform) will help to ensure this population is represented. Additionally, as predictive analytics become embedded in clinical workflows, ongoing validation and governance are critical to ensure model transparency and continued clinical utility.
As an observational study, STEADY is inherently more susceptible to confounding and selection bias than an RCT and must also account for measurement and time-related sources of bias. To mitigate these risks, the registry incorporates standardized data collection procedures, rigorous quality assurance processes, and proactive patient engagement supported by the CRO’s technology platform. In addition, a layered statistical strategy, detailed in the “Methods” section, has been implemented to explicitly address each of the major categories of bias.
Operational challenges inherent to RWD are anticipated and proactively addressed. Variable missingness is expected, as patients move away from sites, as patients have difficulty synchronizing medical records, or as the appeal of incentives wanes. To mitigate this and enhance longitudinal completeness, STEADY employs redundant data pathways with a data preference hierarchy (site-entered electronic case report forms, EMR abstraction, clinical encounter ambient data capture with AI, and patient-reported data), completeness monitoring, and prespecified analytic strategies, including multiple imputation and inverse probability weighting. EMR integration variability across vendors and institutions may affect depth and timing of capture, and not all participants will have site-entered electronic case report forms; sensitivity analyses will be stratified by data availability. Imaging variability is addressed through standardized user guidance, quality review, and feedback. Financial incentives will be available to encourage engagement, and dedicated support staff are available through integrated chat and teleconferencing features. These measures serve to produce valid outcomes estimates while preserving real-world generalizability.
Future Enhancements
ChatGPT (OpenAI) modules have been developed for the platform on an experimental basis that allow patients and clinicians to dictate responses to open-ended questions and see their answers transcribed and parsed into the registry questionnaire using a custom AI model. Comparing acceptability and validity of this novel method of data collection when compared with traditional PROs and ClinROs is a topic of ongoing research. Possible future enhancements include integration of genomic and microbiome data for precision medicine applications, expansion to international sites for global benchmarking, and development of clinician-facing decision support tools.
Conclusion
Comprehensive RWD on DFU management and outcomes remains a major unmet need in clinical care and research. By combining multisource, longitudinal data with AI-enhanced data collection and analytics, the STEADY registry provides a transformative platform to advance DFU research and wound care product development, inform clinical decision-making, and improve patient outcomes throughout the United States.
Author and Public Information
Authors: Jared R. Adams, MD, PhD1; Jennifer N. Lannon BS, BCPA1; Vickie R. Driver, DPM, MS2,3; David Alper, DPM2,4; Cyaandi Dove, DPM2,5; John C. Lantis II, MD2,6; Joseph Rolley, MSIA2,7; Robert Snyder, DPM, MBA, MSc, CWSP2,8
Affiliations: 1Alira Health, Framingham, MA, USA; 2Wound Care Collaborative Community, Orlando, FL, USA; 3Elson S. Floyd College of Medicine, Washington State University, Spokane, WA, USA; 4Mount Auburn Hospital, Cambridge, MA, USA; 5Harold Schnitzer Diabetes Health Center, Oregon Health & Science University, Portland, OR, USA; 6Icahn School of Medicine at Mount Sinai, New York, NY, USA; 7JTR Business Consulting, LLC, Doylestown, PA, USA; 8Centre for Medical Education, School of Medicine, Cardiff University, Cardiff, Wales, UK
Disclosures: J.N.L. completed this work while a Master of Public Health degree candidate at the Johns Hopkins Bloomberg School of Public Health in Baltimore, MD. J.R.A. and J.N.L. are employees of Alira Health, the developers of the registry technology. The registry is currently supported by the WCCC (Wound Care Collaborative Community) and Alira Health. The AI toolset leverages a Health Insurance Portability and Accountability Act (HIPAA)-compliant ChatGPT instance, but there are no commercial connections to OpenAI. All other authors have consulted for Alira Health but are not employees or shareholders.
Ethical Approval: IRB approval for protocol, recruitment materials, informed consent, study questionnaires, and patient and site renumeration schedule will be acquired before study start.
Correspondence: Jared R. Adams, MD, PHD; 1 Grant St 4th Floor Suite 400, Framingham, MA, 01702; jared.adams@alirahealth.com
Manuscript Accepted: February 25, 2026
Recommended Citation
Adams JR, Lannon JN, Driver VR, et al. The STEADY diabetic foot ulcer registry: methods, insights, and future directions. Wounds. 2026;38(3):97-106. doi:10.25270/wnds/26010
Appendix
|
Appendix 1. Full List of Research Objectives |
|
Primary Objective: Conduct comparative effectiveness studies of DFU therapies in real-world settings, including various sites of real-world care. Secondary Objectives: Assess the real-world and long-term safety and effectiveness of DFU therapies, especially those approved in the last 5 years. Characterize the DFU patient and treatment journey(s), including if there is a standard of care, and where they are seen for care (including academic and non-academic hospitals, Indian Health Service, Veterans Administration, long-term care facilities, mobile wound clinics, podiatry clinics, wound clinics, and diabetes clinics). Understand the burden of illness of DFU, including on patients’ quality of life. Evaluate the epidemiology of DFU, including short- and long-term outcomes. Assess the cost-effectiveness of DFU therapies. Exploratory Objectives: Evaluate the type of offloading devices and their frequency of use and evaluate the ease of access to offloading devices. Draw a comprehensive picture of DFU in the context of diabetes, integrating evidence across networks of care. Assess the level of post-healing care and patient education in general clinical practice. Examine the impact of patient compliance on outcomes and the importance of family and social support. Assess the impact of diabetes medications on outcomes by type in clinical practice. Evaluate the percentage of patients that are offered trial therapy. Evaluate the number and type of caregivers as associated with wound closure and non-recurrence of the wound. Assess the use of advanced imaging modalities and long-term outcomes associated with their use. Assess the prevalence of various types of arterial perfusion tests, including concordance of their use with relevant guidelines. |
References
- International Diabetes Federation. Diabetes facts and figures. Accessed October 20, 2025. https://idf.org/about-diabetes/diabetes-facts-figures/
- Armstrong DG, Boulton AJM, Bus SA. Diabetic foot ulcers and their recurrence. N Engl J Med. 2017;376(24):2367-2375. doi:10.1056/NEJMra1615439
- Zhang P, Lu J, Jing Y, Tang S, Zhu D, Bi Y. Global epidemiology of diabetic foot ulceration: a systematic review and meta-analysis. Ann Med. 2017;49(2):106-116. doi:10.1080/07853890.2016.1231932
- Ndosi M, Wright-Hughes A, Brown S, et al. Prognosis of the infected diabetic foot ulcer: a 12-month prospective observational study. Diabet Med. 2018;35(1):78-88. doi:10.1111/DME.13537
- Prompers L, Huijberts M, Apelqvist J, et al. High prevalence of ischaemia, infection and serious comorbidity in patients with diabetic foot disease in Europe: baseline results from the Eurodiale study. Diabetologia. 2007;50(1):18-25. doi:10.1007/S00125-006-0491-1
- Lavery LA, Armstrong DG, Wunderlich RP, Tredwell J, Boulton AJM. Diabetic foot syndrome: evaluating the prevalence and incidence of foot pathology in Mexican Americans and non-Hispanic whites from a diabetes disease management cohort. Diabetes Care. 2003;26(5):1435-1438. doi:10.2337/diacare.26.5.1435
- Centers for Disease Control and Prevention. National Diabetes Statistics Report. Accessed August 30, 2025. https://www.cdc.gov/diabetes/php/data-research/index.html
- Lavery LA, Hunt NA, Ndip A, Lavery DC, van Houtum W, Boulton AJM. Impact of chronic kidney disease on survival after amputation in individuals with diabetes. Diabetes Care. 2010;33(11):2365-2369. doi:10.2337/dc10-1213
- Driver VR, Fabbi M, Lavery LA, Gibbons G. The costs of diabetic foot: the economic case for the limb salvage team. J Vasc Surg. 2010;52(3 Suppl):17S-22S. doi:10.1016/j.jvs.2010.06.003
- Ragnarson Tennvall G, Apelqvist J. Health-related quality of life in patients with diabetes mellitus and foot ulcers. J Diabetes Complications. 2000;14(5):235-241. doi:10.1016/S1056-8727(00)00133-1
- Skrepnek GH, Mills JL Sr, Lavery LA, Armstrong DG. Health care service and outcomes among an estimated 6.7 million ambulatory care diabetic foot cases in the U.S. Diabetes Care. 2017;40(7):936-942. doi:10.2337/dc16-2189
- Hicks CW, Selvarajah S, Mathioudakis N, et al. Burden of infected diabetic foot ulcers on hospital admissions and costs. Ann Vasc Surg. 2016;33:149-158. doi:10.1016/j.avsg.2015.11.025
- Dickson MC, Skrepnek GH. Hospitalization and health resource utilization in emergency department cases of diabetic foot infections in the U.S. from 2012 to 2021: a nationally representative analysis. J Clin Med. 2024;13(18):5361. doi:10.3390/jcm13185361
- Minc SD, Hendricks B, Misra R, et al. Geographic variation in amputation rates among patients with diabetes and/or peripheral arterial disease in the rural state of West Virginia identifies areas for improved care. J Vasc Surg. 2020;71(5):1708-1717.e5. doi:10.1016/j.jvs.2019.06.215
- Moulik PK, Mtonga R, Gill GV. Amputation and mortality in new-onset diabetic foot ulcers stratified by etiology. Diabetes Care. 2003;26(2):491-494. doi:10.2337/diacare.26.2.491
- Walsh JW, Hoffstad OJ, Sullivan MO, Margolis DJ. Association of diabetic foot ulcer and death in a population-based cohort from the United Kingdom. Diabet Med. 2016;33(11):1493-1498. doi:10.1111/dme.13054
- Kostis JB, Dobrzynski JM. Limitations of randomized clinical trials. Am J Cardiol. 2020;129:109-115. doi:10.1016/j.amjcard.2020.05.011
- Driver VR, Walthall H, Oropallo A, et al. Collaboration encourages innovation: setting new standards in wound care with the Wound Care Collaborative Community expert panel consensus recommendations. Wounds. 2024;36(12):410-418. doi:10.25270/wnds/24186
- US Wound & Podiatry Registries. Home [Internet]. Accessed August 30, 2025. https://uswoundregistry.com/
- Fife CE, Eckert KA, Carter MJ. Publicly reported wound healing rates: the fantasy and the reality. Adv Wound Care (New Rochelle). 2018;7(3):77-94. doi:10.1089/wound.2017.0743
- Fife CE, Walker D, Eckert KA. Qualified Clinical Data Registries: how wound care practitioners can make the most out of the Merit-Based Incentive Payment System. Adv Wound Care (New Rochelle). 2018;7(11):387-395. doi:10.1089/wound.2018.0830
- National Institutes of Health. Diabetic Foot Consortium Clinical Research Unit. Accessed August 30, 2025. https://reporter.nih.gov/project-details/11012856
- Pöyry A, Kimpimäki T, Kaartinen I, Salmi TT. Quality registry improves the data of chronic ulcers: validation of Tampere Wound Registry. Int Wound J. 2023;20(9):3750-3759. doi:10.1111/iwj.14270
- Burns L, Le Roux N, Kalesnik-Orszulak R, et al. Real-world evidence for regulatory decision-making: updated guidance from around the world. Front Med (Lausanne). 2023;10:1236462. doi:10.3389/fmed.2023.1236462
- Hripcsak G, Duke JD, Shah NH, et al. Observational Health Data Sciences and Informatics (OHDSI): opportunities for observational researchers. Stud Health Technol Inform. 2015;216:574-578. doi:10.3233/978-1-61499-564-7-574
- Carrigan G, Whipple S, Capra WB, et al. Using electronic health records to derive control arms for early phase single-arm lung cancer trials: proof-of-concept in randomized controlled trials. Clin Pharmacol Ther. 2020;107(2):369-377. doi:10.1002/cpt.1586
- Hernán MA, Robins JM. Using big data to emulate a target trial when a randomized trial is not available. Am J Epidemiol. 2016;183(8):758-764. doi:10.1093/aje/kwv254
- Wagner FW Jr. The dysvascular foot: a system for diagnosis and treatment. Foot Ankle Int. 1981;2(2):64-122. doi:10.1177/107110078100200202
- Mills JL Sr, Conte MS, Armstrong DG, et al. The Society for Vascular Surgery lower extremity threatened limb classification system: risk stratification based on wound, ischemia, and foot infection (WIfI). J Vasc Surg. 2014;59(1):220-34.e1-2. doi:10.1016/j.jvs.2013.08.003
- Armstrong DG, Lavery LA, Harkless LB. Validation of a diabetic wound classification system: the contribution of depth, infection, and ischemia to risk of amputation. Diabetes Care. 1998;21(5):855-859. doi:10.2337/diacare.21.5.855
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a Work Productivity and Activity Impairment instrument. Pharmacoeconomics. 1993;4(5):353-365. doi:10.2165/00019053-199304050-00006
- Herdman M, Gudex C, Lloyd A, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res. 2011;20(10):1727-1736. doi:10.1007/s11136-011-9903-x
- Greenhalgh T, Wherton J, Papoutsi C, et al. Analysing the role of complexity in explaining the fortunes of technology programmes: empirical application of the NASSS framework. BMC Med. 2018;16(1):66. doi:10.1186/s12916-018-1050-6
- Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44-56. doi:10.1038/s41591-018-0300-7
- Wiens J, Saria S, Sendak M, et al. Do no harm: a roadmap for responsible machine learning for health care. Nat Med. 2019;25(9):1337-1340. doi:10.1038/s41591-019-0548-6
- Franklin JM, Schneeweiss S. When and how can real world data analyses substitute for randomized controlled trials? Clin Pharmacol Ther. 2017;102(6):924-933. doi:10.1002/cpt.857
- Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70(1):41-55. doi:10.1093/biomet/70.1.41
- Luo W, Phung D, Tran T, et al. Guidelines for developing and reporting machine learning predictive models in biomedical research: a multidisciplinary view. J Med Internet Res. 2016;18(12)e23. doi:10.2196/jmir.5870
- Golmakani MK, Polley EC. Super learner for survival data prediction. Int J Biostat. 2020;16(2). doi:10.1515/ijb-2019-0065
- Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424. doi:10.1080/00273171.2011.568786
- Li F, Morgan KL, Zaslavsky AM. Balancing covariates via propensity score weighting. J Am Stat Assoc. 2018;113(521):390-400. doi:10.1080/01621459.2016.1260466
- Stuart EA. Matching methods for causal inference: a review and a look forward. Stat Sci. 2010;25(1):1-21. doi:10.1214/09-sts313
- Bang H, Robins JM. Doubly robust estimation in missing data and causal inference models. Biometrics. 2005;61(4):962-973. doi:10.1111/j.1541-0420.2005.00377.x
- Rosenblum M, van der Laan MJ. Targeted maximum likelihood estimation of the parameter of a marginal structural model. Int J Biostat. 2010;6(2):19. doi:10.2202/1557-4679.1238
- Robins JM, Hernán MA, Brumback B. Marginal structural models and causal inference in epidemiology. Epidemiology. 2000;11(5):550-560. doi:10.1097/00001648-200009000-00011
- Robins JM, Finkelstein DM. Correcting for noncompliance and dependent censoring in an AIDS clinical trial with inverse probability of censoring weighted (IPCW) log-rank tests. Biometrics. 2000;56(3):779-788. doi:10.1111/j.0006-341x.2000.00779.x
- Azur MJ, Stuart EA, Frangakis C, Leaf PJ. Multiple imputation by chained equations: what is it and how does it work? Int J Methods Psychiatr Res. 2011;20(1):40-49. doi:10.1002/mpr.329
- Lipsitch M, Tchetgen Tchetgen E, Cohen T. Negative controls: a tool for detecting confounding and bias in observational studies. Epidemiology. 2010;21(3):383-388. doi:10.1097/ede.0b013e3181d61eeb
- VanderWeele TJ, Ding P. Sensitivity analysis in observational research: introducing the E-value. Ann Intern Med. 2017;167(4):268-274. doi:10.7326/m16-2607
- Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94(446):496-509. doi:10.1080/01621459.1999.10474144
- Andersen PK, Gill RD. Cox’s regression model for counting processes: a large sample study. Ann Stat. 1982;10(4):1100-1120. doi:10.1214/aos/1176345976
- Athey S, Wager S. Estimating treatment effects with causal forests: an application. Obs Stud. 2019;5(2):36-51.
- Chipman HA, George EI, McCulloch RE. BART: Bayesian additive regression trees. Ann Appl Stat. 2010;4(1):266-298. doi:10.1214/09-aoas285
- Kuikko K, Salmi T, Huhtala H, Kimpimäki T. Characteristics of chronic ulcer patients by gender and ulcer aetiology from a multidisciplinary wound centre. Int Wound J. 2024;21(8):e70012. doi:10.1111/iwj.70012
- Jones TLZ, Holmes CM, Katona A, et al. The NIDDK Diabetic Foot Consortium. J Diabetes Sci Technol. 2023;17(1):7-14. doi:10.1177/19322968221121152
- US Food and Drug Administration. Draft: Use of real-world evidence to support regulatory decision-making for medical devices. Accessed September 10, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/draft-use-real-world-evidence-support-regulatory-decision-making-medical-devices
- Centers for Medicare & Medicaid Services. Decision memo for Hyperbaric Oxygen Therapy for Diabetic Foot Ulcers (CAG-00383N). Baltimore, MD; 2002. Also includes: NCA - Hyperbaric Oxygen (HBO) Therapy (Section C, Topical Oxygen) (CAG-00060R) - Decision Memo.
- Centers for Medicare & Medicaid Services Medicare Administrative Contractors. LCD L35041: Application of Skin Substitute Grafts for Treatment of Diabetic Foot Ulcers and Venous Leg Ulcers. Revised 2023. https://www.cms.gov/medicare-coverage-database/view/lcd.aspx?LCDId=35041
- Centers for Medicare & Medicaid Services. Local coverage determination L33831: Surgical Dressings. Revised 2023. https://www.cms.gov/medicare-coverage-database/view/lcd.aspx?LCDId=33831

















