Diagnostic Accuracy of the Mangled Extremity Severity Score for Predicting Amputation in Adult Lower Limb Trauma: A Systematic Review and Meta-Analysis
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. The Mangled Extremity Severity Score (MESS) was developed to guide surgeons in deciding between limb salvage and amputation in severe lower limb trauma. Its accuracy has been questioned, with inconsistent results reported across different health care systems. Objective. To assess the diagnostic accuracy of the MESS for predicting amputation in adults with severe lower limb trauma and to explore differences between high-income countries (HICs) and low- and middle-income countries (LMICs). Methods. The authors conducted a systematic review and meta-analysis following Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines. Observational studies reporting MESS diagnostic accuracy were identified. Data were extracted in duplicate and appraised using the Joanna Briggs Institute checklist for systematic reviews. Pooled diagnostic odds ratios (DORs) were calculated with a random-effects model, and subgroup analyses explored differences by income setting and injury mechanism. Results. Thirteen studies with a combined total of 793 participants were identified, of which 7 studies comprising 385 patients from 7 countries were included in the meta-analysis. Overall, the MESS demonstrated strong predictive value, with a pooled DOR of 29.37 (95% CI, 7.79-109.55; P < .001). Patients scoring above the cutoff were approximately 29 times more likely to undergo amputation than those with a score below the cutoff. Heterogeneity was moderate-to-substantial (I² = 62.92%, tau² = 1.99, Q = 16.51; P = .011). Publication bias assessment showed minimal concerns (fail-safe N = 165, Kendall tau = 0.429; P = .239) (Egger regression = 1.955; P = .051). The MESS had the highest performance in lower-middle-income countries, with an amputation rate of 32.1%. In upper-middle-income countries the mean DOR was 58.8, with a lower amputation rate of 10.0%. The poorest performance was demonstrated in the HIC of Austria, with a sensitivity of 50.0%, specificity of 76.2%, and a DOR of 3.20. Conclusion. This study provides the first global synthesis of MESS diagnostic accuracy in severe lower limb trauma. The findings highlight that the MESS remains a valuable decision-making tool, particularly in LMICs and high-energy trauma; however, its predictive power is weaker in advanced care settings where salvage options have expanded.
Severe lower limb trauma and the management of mangled extremities continue to pose difficult challenges in orthopedic practice. A mangled extremity is a limb with an injury to at least 3 of 4 systems: soft tissue, bone, nerves, and vessels.1 For surgeons, the decision to salvage or amputate a mangled limb is rarely straightforward, and definitive guidelines have been lacking.2 Advances in microsurgery, vascular repair, and intensive care have expanded the boundaries of limb salvage, but success often comes at a cost. Patients who undergo repeated reconstruction may face functional impairment, secondary amputations, prolonged hospital stays, and long-term psychosocial and financial strain.3–5
The global burden of mangled extremities is considerable. Road traffic accidents (RTAs), particularly high-energy motorcycle crashes, account for most cases, disproportionately affecting young men in their most productive years.6–8 In India, RTAs have been reported in greater than 70% of severe limb trauma cases,3 while Egyptian series place the figure above 80%.7 Faced with such injuries, clinicians must make decisions in what has been described as a “grey zone.” Some cases demand immediate amputation, such as when ischemia has exceeded 6 hours or when circulation cannot be restored.5,9 Popliteal artery trauma, for example, may result in amputation in up to one-third of cases, with survivors often experiencing phantom limb pain, musculoskeletal strain, and depression.10 The management decision often requires a multidisciplinary team, considering patient age, comorbidities, and preferences alongside the extent of tissue damage, vascular involvement, ischemia time, and available resources.6,9 Even taking these considerations into account, predicting which limbs will ultimately require amputation and which will be functional remains uncertain.
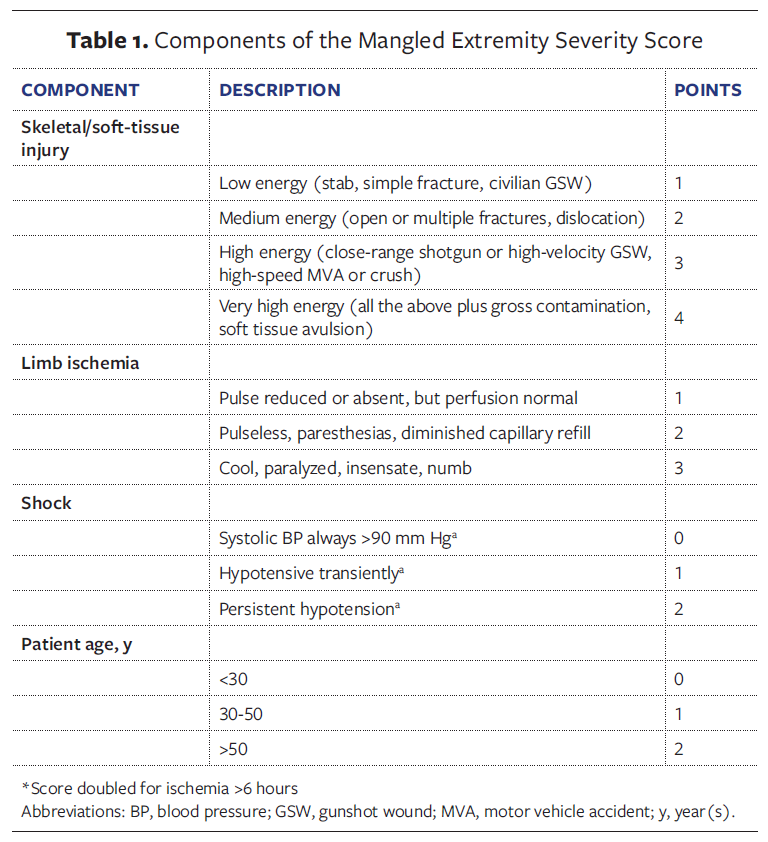
To bring structure to this complex decision-making process, several scoring systems have been introduced. The Mangled Extremity Severity Score (MESS) is the most widely used, in part because of its simplicity. It incorporates skeletal and soft-tissue injury, ischemia, shock, and patient age,3,11 with early studies suggesting that a score of 7 or higher predicted amputation with near-perfect accuracy2,12,13 (Table 1). However, research from different health settings has shown that the score does not perform consistently.
In high-income countries (HICs), where advanced trauma care is widely available, the MESS has been criticized for its poor predictive value. An Austrian study reported that its performance was little better than chance,14 while research in the United States found that more than half of patients with a MESS greater than or equal to 7 underwent successful salvage.15 Japanese investigators have even suggested raising the threshold to 8 to improve accuracy.16 In contrast, studies from low- and middle-income countries (LMICs) often report higher sensitivity and specificity, with some Indian and Egyptian series citing a sensitivity greater than 90%.3,7 These findings are far from uniform, however, with studies in other LMICs finding the scoring tool to be less reliable, particularly in centers with dedicated trauma services and improved resources.9,17
Other scoring systems, including the Ganga Hospital Open Injury Severity Score (GHOISS), the Limb Salvage Index (LSI), and the Hannover Fracture Scale, have been proposed to overcome limitations of the MESS. Some appear more accurate in certain contexts, for example, the GHOISS for Gustilo IIIB injuries,17 but large multicenter studies such as the Lower Extremity Assessment Project (LEAP) trial have concluded that no single score reliably predicts long-term function.15
Despite decades of use of the MESS, no comprehensive systematic review and meta-analysis has synthesized its diagnostic accuracy across global settings. The absence of such evidence leaves clinicians to rely on fragmented and sometimes contradictory findings. With differences emerging between high-income and low-resource contexts, a pooled evaluation is urgently needed. The objective of this study was to assess the diagnostic accuracy of the MESS for predicting amputation in adults with severe lower extremity trauma. The authors of the present study also sought to determine any differences between HICs and LMICs. Clarifying the true predictive value of the MESS, as well as identifying where it remains useful or falls short, has the potential to guide more evidence-based decisions, reduce unnecessary amputations, and improve outcomes for patients with these life-changing injuries.
Methods
Protocol and registration
This systematic review followed Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines and was registered in the international systematic review registry PROSPERO (CRD420251140417). Ethical approval was not required, because only published data were analyzed.
Database and search strategy
The PICO framework was used to develop the study question: population: adult lower limb trauma patients; intervention: not applicable; comparison: amputation vs salvage; and outcome: MESS diagnostic accuracy. The authors searched PubMed, Scopus, Web of Science, and Lens.org for studies published between January 1, 2010, and August 31, 2025. Search terms included Mangled Extremity Severity Score, amputation, limb salvage, and lower limb trauma (Supplementary File). Medical Subject Headings (MeSH) and free-text terms were used, along with Boolean operators (AND, OR) to refine searches. No language restrictions were applied, but only English-language studies or translations were included.
Study selection
Eligible studies were observational and peer-reviewed studies that reported MESS diagnostic accuracy in adult lower limb trauma and included global or LMIC settings. Case reports, reviews, non-adult or upper limb studies, and non-English-
language texts without translations were excluded. Two reviewers (E.M., M.A.) screened titles and abstracts using Rayyan, with full-text disputes resolved by a third reviewer (J.K.).
Data extraction and quality assessment
Data were extracted in duplicate using a standardized Excel (Microsoft) form, capturing study characteristics and MESS cutoffs. Diagnostic measures including true positives (correctly predicted amputations) and true negatives (correctly predicted salvages) were also extracted. Quality was assessed using the Joanna Briggs Institute (JBI) checklist for systematic reviews,18 with 6 or more of 11 criteria met indicating high-quality studies.
Income classification of study countries was based on the World Bank country income groups.19 Countries were categorized as low-income, lower-middle-income, upper-middle-income, or high-income according to their gross national income per capita at the time of publication. For analysis, the authors of the present study grouped LMICs together and contrasted them with HICs.
Statistical analysis
Diagnostic odds ratios (DORs) were logit-transformed and pooled using a
random-effects model (restricted maximum likelihood) in Jamovi (version 2.6.44; the jamovi project). Heterogeneity was assessed with I², tau², and Cochran Q. Publication bias was evaluated using fail-safe N, Kendall tau, Egger regression, and funnel plots. Subgroup analyses examined income level and trauma mechanism. Equivalence testing used two one-sided tests (TOSTs) with bounds of plus or minus 0.5 logit units. A P value less than .05 was considered statistically significant.
Results
Study selection
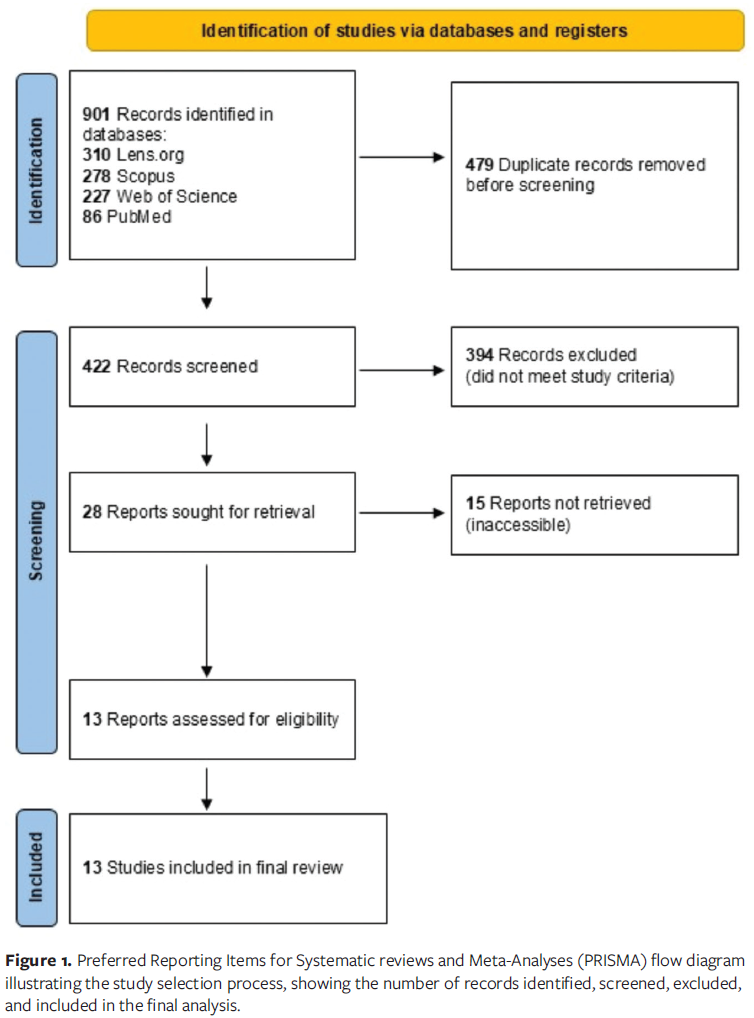
From 901 records (PubMed, 86; Scopus, 278; Web of Science, 227; Lens.org, 310), 479 duplicates were removed. Of 422 screened records, 28 were sought for retrieval, and 13 studies (n = 793 patients) from the United States, India, Nepal, Egypt, Brazil, Nigeria, Austria, and China were included in the final analysis (Figure 1). Seven of these 13 studies provided complete diagnostic accuracy data (true positives, false positives, true negatives, false negatives) necessary for calculating DORs and were included in the quantitative meta-analysis. The remaining 6 studies were retained for qualitative synthesis but were excluded from meta-analysis due to incomplete reporting of diagnostic measures required for pooled analysis.
Study characteristics
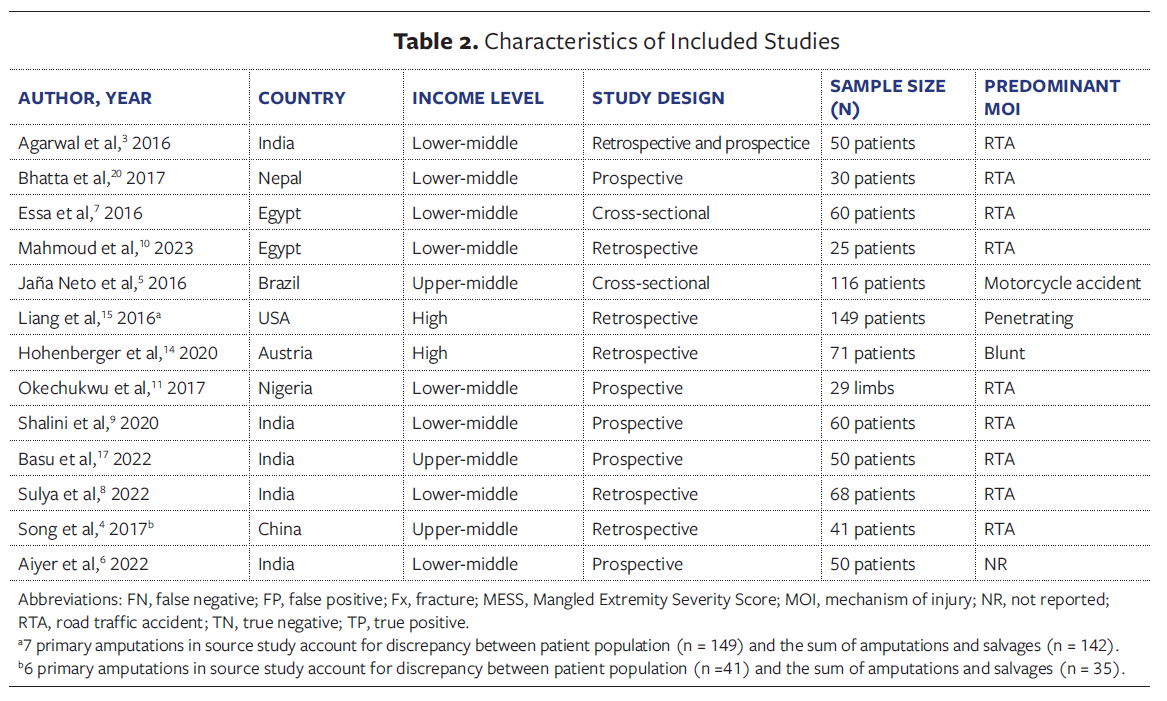
Table 2 summarizes the 13 included studies, detailing country, World Bank-
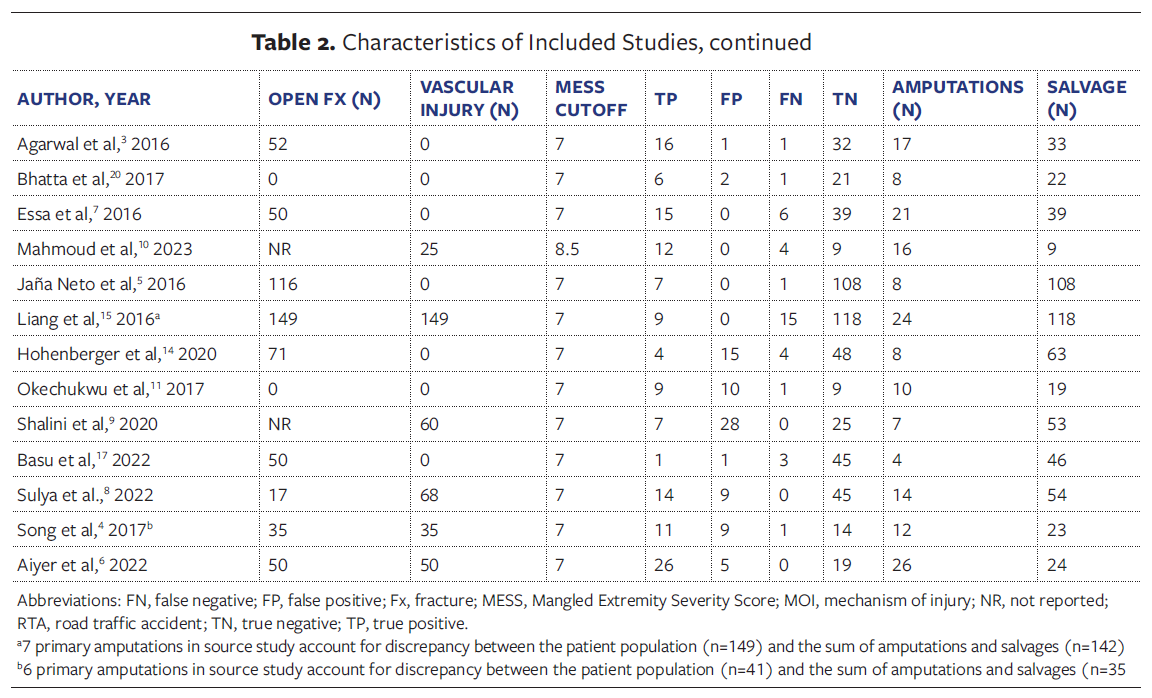
defined income level, study design, sample size, predominant mechanism of injury, and MESS cutoffs (7 to 8.5). The majority of the studies were conducted in LMICs (10 studies [77%]),3,4,6-9,10,11,17,20 with 2 in HICs (Austria, United States)14,15 and 1 in an upper-middle-income country.5 Most studies (12 of 13 [92.3%])3-7,9,11,14,15,17,20 used a MESS cutoff value of 7 to determine need for amputation. RTAs were the predominant mechanism of injury across settings.
Meta-analysis results
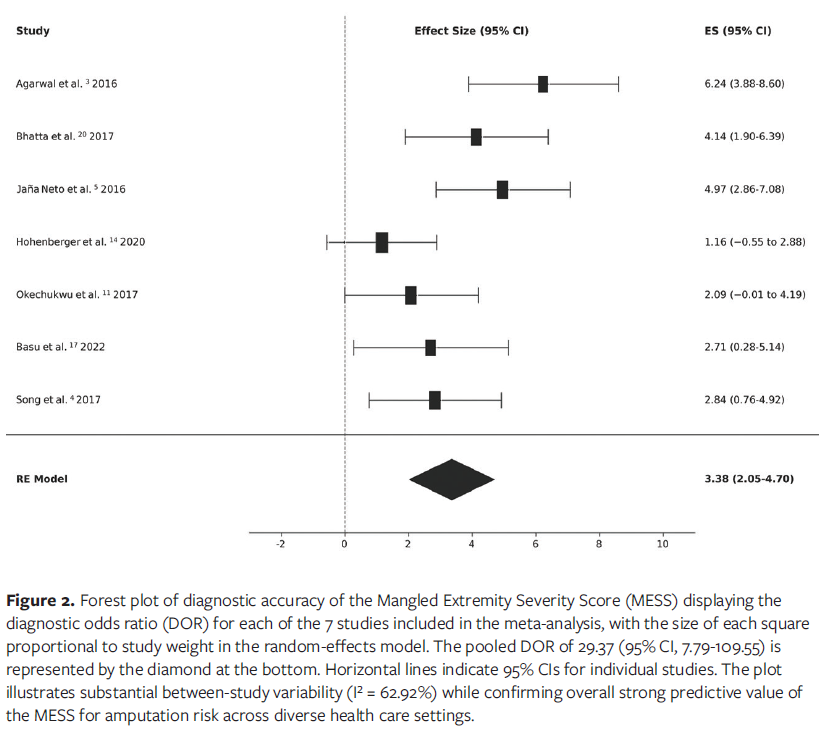
Given the variability amongst studies, it was not possible to perform a formal meta-analysis. Seven studies were included in a random-effects meta-analysis.3-5,11,14,17,20 The random-effects meta-analysis of 7 studies showed a pooled DOR of 29.37 (95% CI, 7.79-109.55; P < .001), indicating that patients with MESS scores above the cutoff are approximately 29 times more likely than those with MESS scores below the cutoff to require amputation. The results were statistically significant, and the CI shows the range of likely values for the DOR (Table 3).

The wide CI reflects the heterogeneity across study settings while confirming substantial overall predictive strength. A forest plot illustrating the DOR for each study and the pooled estimate is provided in Figure 2.
Heterogeneity and publication bias assessment
Heterogeneity analysis revealed an I² value of 62.92% and tau² = 1.99, indicating moderate-to-substantial variation across studies. This suggests that differences in results are attributable to true variation in study characteristics, such as health care setting, injury severity, available resources, and patient populations, rather than sampling error alone. The Q statistic (Q = 16.509; P = .011) confirms that this variation is significant (Table 4).
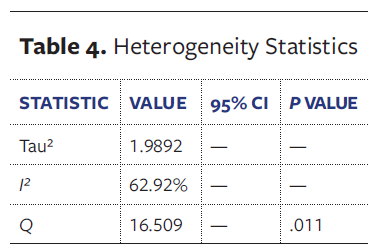
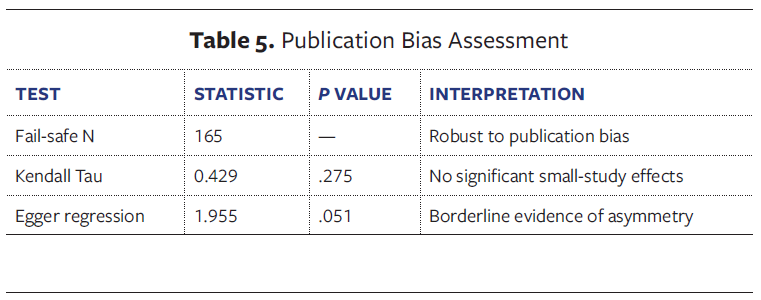
Minimal publication bias was detected (fail-safe N = 165, Kendall tau = 0.429; Egger regression 1.955 (Table 5). The fail-safe N of 165 suggests that it would take 165 null studies to change the results, indicating robustness. However, the Egger regression test approached statistical significance, suggesting borderline evidence of potential publication bias. A funnel plot supported these findings (Figure 3).
Equivalence testing
The TOST equivalence test was nonsignificant, indicating that the DOR is not equivalent to zero, with a significant null hypothesis test (P < .001) (Table 6). The nonsignificant equivalence test and significant null hypothesis test confirm that the MESS has a real, nonzero effect in predicting amputation.

Subgroup analysis
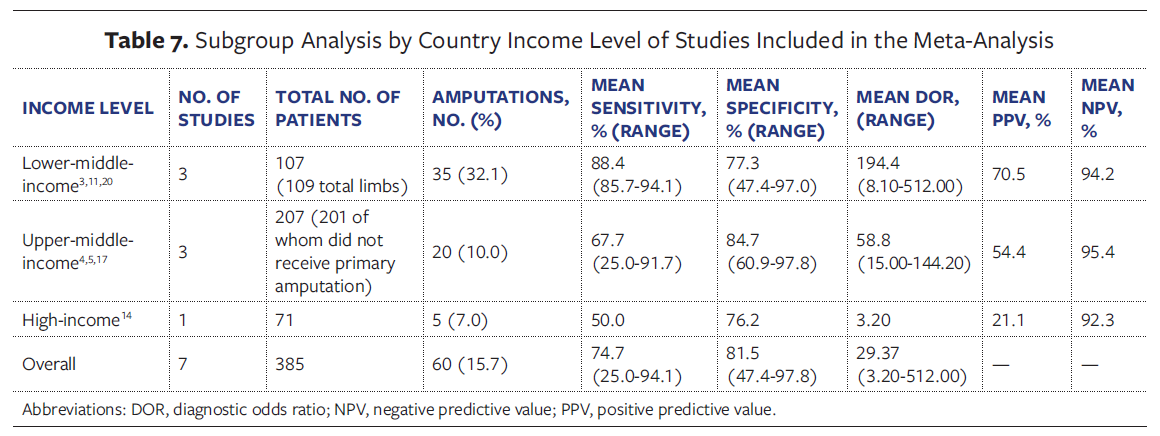
By income level, the MESS showed its strongest performance in lower-middle-income countries (3 studies, 107 patients, 109 total limbs),3,11,20 with high sensitivity (mean, 88.4% [range, 85.7%-94.1%]), consistently strong negative predictive values (mean, 94.2% [range, 90.0%-97.0%]), and the highest mean DOR of 194.4 (range, 8.10-512.00). The amputation rate was 32.1% (35 of 109 limbs).
Upper-middle-income countries (3 studies, 207 patients, 201 of whom did not receive primary amputation)4,5,17 demonstrated lower and more variable sensitivity (mean, 67.7% [range, 25.0%-91.7%]) but maintained good specificity (mean, 84.7%). The mean DOR was 58.8, with a lower amputation rate of 10.0% (20 of 201). In contrast, the single HIC study in the random-effects meta-analysis (Austria, 71 patients) showed the poorest performance, with a sensitivity of 50.0%, specificity of 76.2%, and DOR of 3.20, representing an approximately 60-fold reduction in discriminatory ability compared with lower-middle-income settings. Although the overall amputation rate was only 7.0% (5 of 71), a MESS greater than or equal to 7 substantially overpredicted amputation necessity, with 79% of patients scoring above the threshold ultimately undergoing successful limb salvage (15 of 19). Overall, the MESS appeared to be most accurate in resource-limited settings, whereas in high-resource centers it tended to overestimate the need for amputation, likely reflecting advances in microsurgical and critical care (Table 7).
Study quality assessment
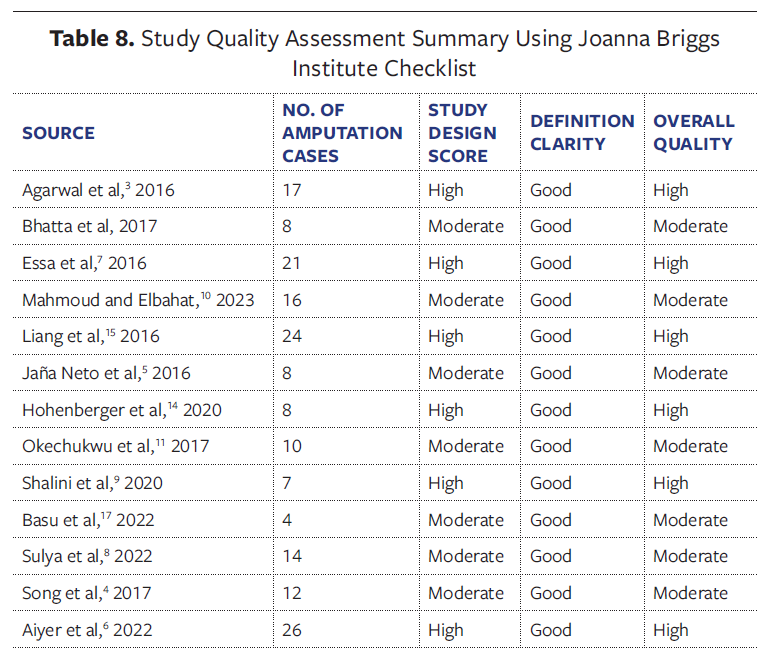
The quality of the 13 studies was assessed using the aforementioned JBI checklist.18 High-quality studies (≥6/11 criteria met) had strong methods and clear definitions, while moderate-quality studies had limitations such as smaller sample sizes or less robust designs. Most studies were reliable, but some had minor methodological issues (Table 8).
Discussion
To the authors’ knowledge, this systematic review and meta-analysis is the first global evaluation of the MESS in adult lower limb trauma. Thirteen studies with a combined total of 793 participants were identified, of which 7 studies comprising 381 patients from 7 countries were included in the meta-analysis. Overall, the MESS retains significant predictive value for amputation, with a pooled DOR of 29.37. In practical terms, patients with a score above the cutoff were approximately 29 times more likely to undergo amputation than those with a score below the cutoff. This confirms that the MESS remains clinically useful, particularly when used to support decision-making in the early hours after injury. The substantial heterogeneity in the present analysis (I² = 62.92%) is not surprising given the diversity of health care systems, injury mechanisms, and patient populations represented. Even so, equivalence testing confirmed that the DOR was significantly greater than zero, reinforcing the clinical relevance of the findings.
Subgroup analysis suggests that the MESS performs better in LMICs than in HICs. In LMICs, the pooled DOR reached 194.4, with a mean sensitivity of 88.4%. This contrasts sharply with the single HIC study from Austria, which showed a DOR of only 3.20 and sensitivity of 50.0%, representing an approximately 60-fold difference in discriminatory ability.14 These findings echo reports from India and Egypt, both LMICs. Agarwal et al3 reported a specificity of 98.5%, and Essa et al7 demonstrated a specificity of 100% of the MESS using the traditional cutoff of 7. However, Hohenberger et al14 concluded in their Austrian study that the MESS was not a good predictor for amputation, with an area under the receiver operating characteristic curve of 0.57. The positive predictive value of only 21.1% in that study indicates that 4 out of 5 patients with a MESS greater than or equal to 7 ultimately underwent successful limb salvage, highlighting significant overprediction of amputation necessity in high-resource settings.14
The stronger performance of the MESS in LMICs may reflect the injury patterns seen in these regions. High-energy RTAs dominate, producing more predictable injury profiles that fit the MESS framework. Additionally, decision-making environments in LMICs are often closer to the conditions under which the MESS was first validated, with limited access to microsurgery, prolonged intensive care, and long-term rehabilitation needs.6,21
Nevertheless, results from LMICs are not uniform. Shalini et al9 described the MESS as a poor predictor in their Indian series, while Basu et al17 reported lower sensitivity and specificity than earlier studies. Sharma et al,22 in another Indian series, noted that whereas the MESS was excellent for predicting primary amputation, it lacked usefulness in predicting limb salvage. These differences likely reflect variability in trauma system maturity, surgical expertise, and resource availability within LMICs.
In contrast, studies from HICs consistently highlight limitations of the MESS. Hohenberger et al,14 in an Austrian study, suggested the score performed little better than chance. Similarly, Liang et al15 showed that more than half of patients with a score greater than 7 went on to successful salvage, undermining the reliability of the traditional threshold. This finding is in line with studies in other HICs. An evaluation of the American Association for the Surgery of Trauma PROspective Vascular Injury Treatment registry demonstrated a reduced diagnostic accuracy of the MESS in predicting amputation, emphasizing the need for further research evaluating additional predictors of amputation.23
This reduced accuracy is likely due to the evolution of trauma care. Modern techniques such as early revascularization, free tissue transfer, negative pressure wound therapy, and advanced rehabilitation have expanded the boundaries of salvageability; limbs once deemed unsalvageable are now viable.14,16 Studies in Japan and the United States proposed raising the MESS cutoff to 8 in order to improve specificity while maintaining acceptable sensitivity.16,23 Other studies suggest decreasing the value for primary amputation by 0.5 point or 1 point to avoid futile efforts of limb salvage.22 This illustrates how local adaptation of scoring thresholds may improve clinical utility.
The MESS also performed differently depending on the mechanism and type of injury. The score showed higher diagnostic accuracy for RTAs and other high-energy mechanisms than for mixed or blunt trauma. This is consistent with the pathophysiology of crush injuries with tibia and fibula fractures that produce extensive tissue and vascular damage, aligning with MESS parameters.3 In contrast, lower-energy or isolated vascular injuries are less predictable within the scoring framework.10,23
Certain injury subtypes merit special mention. Mahmoud and Elbahat10 evaluated the validity of the MESS in patients with popliteal artery injuries. In the case of traumatic popliteal artery injuries, they found that a MESS lower than 8 was a positive predictor of limb salvage in patients with popliteal artery trauma.10 Their research is in agreement with Van Nut et al,24 who also demonstrated that a higher MESS was associated with amputation in popliteal artery injuries. This suggests that injury-specific thresholds may enhance clinical relevance.
Although the MESS remains the most widely used tool for predicting amputation risk, other systems show promise. The GHOISS has repeatedly outperformed the MESS in predicting salvageability, particularly in Gustilo IIIB fractures.6,17 Similarly, the LSI has been validated in several series, with thresholds around 6 predicting amputation with good accuracy.3,8 The Predictive Salvage Index had high sensitivity but low specificity in predicting limb salvage. Overall, it has been advised to use any lower extremity severity score above the amputation threshold with caution.25,26 Limb salvage is not always synonymous with success. Many salvaged limbs remain painful or poorly functional, requiring secondary procedures or eventual amputation. These outcomes carry significant physical, emotional, and financial burdens for patients and families.3,4 However, amputation itself is far from a simple solution, with phantom limb pain, musculoskeletal strain, and psychosocial effects well documented.10 Large multicenter studies concluded that none of these systems reliably predict long-term function or quality of life, limiting their standalone value.27,28,29
The findings of the present meta-analysis support the MESS as a useful adjunct, not a replacement for clinical judgment, particularly in resource-limited settings and cases involving high-energy mechanisms. The best outcomes are achieved through multidisciplinary collaboration among orthopedic, vascular, plastic, and trauma surgeons. Patient-specific factors, including age, comorbidities, cultural background, and personal preference, remain central to decisions. Ischemia time, quality of vascular repair, and availability of advanced trauma resources all influence outcomes, while economic considerations can shape treatment choices, particularly in LMICs.4,9
Limitations
Limitations of this study include moderate heterogeneity, variations in MESS thresholds (7 to 8.5), and the predominance of LMIC studies, which may limit generalizability to HIC contexts. Most studies were hospital-based, potentially overrepresenting severe cases. Retrospective designs in some series also raise the risk of bias. Importantly, most studies reported binary outcomes (salvage vs amputation) without systematically reporting postoperative wound complications, dehiscence rates, or comparative effectiveness of wound management protocols (eg, negative pressure wound therapy vs primary closure) stratified by MESS score data that would be valuable for wound care providers in predicting healing trajectories and guiding treatment decisions. Finally, most studies lacked determination of long-term functional outcomes, quality-of-life assessments, and cost-effectiveness analyses, limiting the authors’ ability to determine whether anatomical salvage translates to meaningful patient-centered outcomes. Future studies must go beyond anatomical survival to evaluate quality of life, functional recovery, and cost-effectiveness. These factors ultimately matter most to patients.
Conclusion
This review confirms that the MESS remains a valuable tool for predicting amputation risk, particularly in LMICs and in high-energy trauma. However, its accuracy is limited in advanced care settings, where modern reconstructive options have shifted the boundaries of salvage. Clinicians should use the MESS as part of a broader, context-specific decision-making framework, alongside multidisciplinary input and patient-centered considerations. Future research should refine MESS thresholds, validate its use in specific injury subtypes, and, most importantly, incorporate measures of long-term function and quality of life. The ultimate goal is not simply to save a limb but to restore meaningful life and function for patients facing these devastating injuries.
Author and Public Information
Authors: Enock Mukiibi, MBBS1; Musilim Abdulai, MBBS1; Jackson Kakooza, MBSS1; Catherine R. Lewis, MD, PhD2; Prosper Akankwasa, MBBS3; Ahmed Kiswezi Kazigo, MBBS, MSurg1; Evelyn Njawuzi Balondemu, MS4; Bienfait Mumbere, MBBS, MSurg1; Sedrick Bukyana, MBBS1; Abass Alao Safiriyu, PhD5; John Turyagumanawe, MBBS1; Marcel Idi Ehanga, MBBS1; Eunice Inomu, MBBS, MSurg1; Francis Mamadi Owera, MBBS, MSurg1; Michael Mugenyi, MBBS, MSurg1
Affiliations: 1Department of Surgery, Kampala International University, Western Campus, Ishaka, Uganda; 2Department of Surgery, St. Joseph’s Hospital Kitovu, Masaka, Uganda; 3Department of Obstetrics and Gynaecology, Kampala International University, Western Campus, Ishaka, Uganda; 4Department of Clinical Psychology, Makerere University, Kampala, Uganda; 5Department of Physiology, Kampala International University, Western Campus, Ishaka, Uganda
Acknowledgments: The authors thank their colleagues for assistance with literature searches and data extraction.
Disclosure: The authors declare no financial or other conflicts of interest.
Correspondence: Michael Mugenyi, MBBS, MSurg; Department of Surgery, Kampala International University, Western Campus, Ishaka, Uganda; email: mugenyimichael4@gmail.com
Manuscript Accepted: January 6, 2026
References
-
- Prasarn ML, Helfet DL, Kloen P. Management of the mangled extremity. Strategies Trauma Limb Reconstr. 2012;7(2):57-66. doi:10.1007/s11751-012-0137-4
- Lange RH. Limb reconstruction versus amputation decision making in massive lower extremity trauma. Clin Orthop Relat Res. 1989;(243):92-99.
- Agarwal V, Agarwal S, Singh A, et al. An evaluation of the clinical utility of mangled extremity severity score in severely injured lower limbs. Int J Res Med Sci. Published online 2016:1661-1665. doi:10.18203/2320-6012.ijrms20161245
- Song W, Zhou D, Dong J. Predictors of secondary amputation in patients with grade IIIC lower limb injuries. Medicine. 2017;96(22):e7068. doi:10.1097/md.0000000000007068
- Jaña Neto FC, Canal M de P, Alves BAF, Ferreira PM, Ayres JC, Alves R. Análise das características dos pacientes com fratura exposta de tíbia grau III de Gustilo e Anderson. Rev Bras Ortop (Sao Paulo). 2016;51(2):143-149. doi:10.1016/j.rbo.2015.06.002
- Aiyer S, Wankhade A, Bava S, Vaghasia D, Nahatkar T, Salve A. The Corelation Between GHOIS and MESS Score to Assess the Salvageability Outcomes of Patients with Vascular Injury. Int J Sci Res. 2022;11(9):1-3. doi:10.36106/ijsr/8400117
- Essa AA, El-Shaboury IM, El-Beltagy YE. Evaluation of prognostic predictors of mangled extremity severity scoring system on the outcome of traumatic extremities injuries at emergency department in Suez Canal University Hospitals. International Surgery Journal. 2016;4(1):75. doi:10.18203/2349-2902.isj20164446
- Sulya D, Vairagar SR, Saichandran B V, et al. A Descriptive Study of Concomitant Vascular and Bone Injuries of the Limbs in a Tertiary Care Hospital in South India. Indian Journal of Vascular and Endovascular Surgery. 2022;9(5):364-369. doi:10.4103/ijves.ijves_71_22
- Shalini A, Singh D, Dachipalli S, Maddu S, Ram L. The validity of the mangled extremity severity score scoring system for lower limb vascular trauma in a tertiary care center. Indian Journal of Vascular and Endovascular Surgery. 2020;7(2):141. doi:10.4103/ijves.ijves_63_19
- Mahmoud AAH, Elbahat MAA. Validity of Mangled Extremity Score System to Predict Limb Salvage on Cases of Traumatic Popliteal Artery Injuries: A Case Series and Review of the Literature. J Endovasc Resusc Trauma Manag. 2023;7(2). doi:10.26676/jevtm.283
- Okechukwu BA, Akinmokun OI, Enweluzo GO, Giwa SO. Validation of mangled extremity severity score in assessing the need for extremity amputation. Nigerian Journal of Medicine. 2017;26(2):156. doi:10.4103/1115-2613.278288
- Johansen K, Daines M, Howey T, Helfet D, Hansen ST. Objective Criteria Accurately Predict Amputation following Lower Extremity Trauma. The Journal of Trauma: Injury, Infection, and Critical Care. 1990;30(5):568-573. doi:10.1097/00005373-199005000-00007
- Helfet DL, Howey T, Sanders R, Johansen K. Limb salvage versus amputation: Preliminary results of the Mangled Extremity Severity Score. Clin Orthop Relat Res. 1990;(256):80-86.
- Hohenberger GM, Konstantiniuk P, Cambiaso-Daniel J, et al. The Mangled Extremity Severity Score Fails to be a Good Predictor for Secondary Limb Amputation After Trauma with Vascular Injury in Central Europe. World J Surg. 2019;44(3):773-779. doi:10.1007/s00268-019-05263-w
- Liang NL, Alarcon LH, Jeyabalan G, Avgerinos ED, Makaroun MS, Chaer RA. Contemporary outcomes of civilian lower extremity arterial trauma. J Vasc Surg. 2016;64(3):731-736. doi:10.1016/j.jvs.2016.04.052
- Omoto R, Umemura Y, Hanaoka K, et al. Re-evaluation of cutting criteria for limb amputation using the Mangled Extremity Severity Score. Trauma Surg Acute Care Open. 2025;10(1):e001694. doi:10.1136/tsaco-2024-001694
- Basu A, Nandy SN, Sarkar PS. Evaluation of Management of Gustilo Type IIIb Open Wound According to Ganga Hospital Open Injury Severity Score (GHOISS). Int J Sci Res. 2022;11(3):29-33. doi:10.36106/ijsr/0509016
- Aromataris E, Fernandez R, Godfrey CM, Holly C, Khalil H, Tungpunkom P. Summarizing systematic reviews: methodological development, conduct and reporting of an umbrella review approach. Int J Evid Based Healthc. 2015;13(3):132-140. doi:10.1097/XEB.0000000000000055
- The World Bank. The World Bank Atlas method - detailed methodology. Accessed November 7, 2025. https://datahelpdesk.worldbank.org/knowledgebase/articles/378832-the-world-bank-atlas-method-detailed-methodology
- Bhatta TR, Gyawali B, Tamrakar R, et al. Utility of mangled extremity severity score in severely injured lower limbs. Journal of Society of Surgeons of Nepal. 2017;18(1):23-25. doi:10.3126/jssn.v18i1.17210
- Hoogervorst LA, Hart MJ, Simpson PM, et al. Outcomes of severe lower limb injury with Mangled Extremity Severity Score ≥ 7. Bone Joint J. 2021;103-B(4):769-774. doi:10.1302/0301-620X.103B4.BJJ-2020-1647.R1
- Sharma S, Devgan A, Marya KM, Rathee N. Critical evaluation of mangled extremity severity scoring system in Indian patients. Injury. 2003;34(7):493-496. doi:10.1016/S0020-1383(02)00214-0
- Loja MN, Sammann A, DuBose J, et al. The mangled extremity score and amputation. Journal of Trauma and Acute Care Surgery. 2017;82(3):518-523. doi:10.1097/TA.0000000000001339
- Van Nut L, Thanh Son H, Lam Vuong N. Revisiting the Mangled Extremity Severity Score (MESS) in Popliteal Artery Injury: A Single-Centre Experience in Vietnam. Cureus. Published online May 10, 2023. doi:10.7759/cureus.38813
- Bosse MJ, MacKenzie EJ, Kellam JF, et al. A prospective evaluation of the clinical utility of the lower-extremity injury-severity scores. J Bone Joint Surg Am. 2001;83(1):3-14. doi:10.2106/00004623-200101000-00002
- Schirò GR, Sessa S, Piccioli A, Maccauro G. Primary amputation vs limb salvage in mangled extremity: a systematic review of the current scoring system. BMC Musculoskelet Disord. 2015;16(1):372. doi:10.1186/s12891-015-0832-7
- Higgins TF, Klatt JB, Beals TC. Lower Extremity Assessment Project (LEAP)--the best available evidence on limb-threatening lower extremity trauma. Orthop Clin North Am. 2010;41(2):233-239. doi:10.1016/j.ocl.2009.12.006
- Swiontkowski MF, Mackenzie EJ, Bosse MJ, Jones AL, Travison T. Factors Influencing the Decision to Amputate or Reconstruct after High-Energy Lower Extremity Trauma. Journal of Trauma and Acute Care Surgery. 2002;52(4):641-649.
- Patterson BM, Agel J, Swiontkowski MF, Mackenzie EJ, Bosse MJ. Knee dislocations with vascular injury: outcomes in the Lower Extremity Assessment Project (LEAP) Study. J Trauma. 2007;63:855-8.