Regulatory Classification vs Real-World Outcomes in CAMPs: Poster Insights from SAWC Spring
A large real-world evidence analysis presented at SAWC Spring challenges a foundational assumption in Medicare reimbursement policy: that FDA regulatory classification (361 HCT/P, 510(k), or PMA) predicts the clinical performance of cellular, acellular, and matrix-like products (CAMPs) used in chronic wound care.
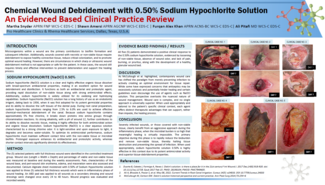
Using a CMS limited dataset spanning 2016–2024, investigators evaluated more than 3.3 million wound episodes, including 2.65 million lower extremity diabetic ulcers (LEDUs) and 745,411 venous leg ulcers (VLUs). CAMP-treated cases were rigorously matched to standard of care (SOC) cohorts using four-way matching (age, sex, comorbidities, treatment timing, debridement depth, and episode year), with additional stratification by regulatory pathway.
The findings were striking: FDA regulatory classification did not meaningfully predict clinical outcomes. Across matched cohorts, differences between CAMPs categorized as PMA, 510(k), or 361 products were largely non-significant. In contrast, all CAMP-treated groups demonstrated superior outcomes compared with SOC alone, including lower mortality (LEDUs: 9.5–11.1% vs 12.7%; VLUs: 10.0–12.2% vs 13.2%), fewer amputations, and reduced emergency department, ICU, and critical care utilization. These benefits were consistent regardless of wound size or product classification.
While SOC episodes showed slightly shorter length of treatment, they were associated with substantially higher complication rates—highlighting a tradeoff between treatment duration and clinical outcomes.
From a reimbursement and policy perspective, these results directly challenge current Medicare frameworks that tier coverage or payment based on FDA regulatory pathways. If outcomes are statistically comparable across CAMP categories, regulatory classification may not be a valid proxy for clinical value.
For clinicians and stakeholders, this study reinforces the importance of real-world effectiveness data in guiding both treatment decisions and policy reform. It suggests that future reimbursement models may need to shift away from regulatory-based tiering toward outcomes-driven frameworks.
Reference
Tettelbach W. Rethinking Regulatory Tiers: Medicare Real-World Evidence Shows Cellular, Acellular, and Matrix-Like Product (CAMP) Outcomes Are Independent of FDA Regulatory Classification. SAWC Spring Poster, 2026.
© 2026 HMP Global. All Rights Reserved.
All information regarding reimbursement, legislation, regulations, policy, and legal proceedings, is provided as a service to our audience. Commercially reasonable efforts have been made to ensure the accuracy of the information within this resource but HMP Global, their employees, their affiliates, contributors, commenters, and reviewers do not represent, guarantee, or warranty that any information provided within this resource is error-free. HMP Global, their employees, their affiliates, contributors, and reviewers disclaim all liability attributable to the use of any information, guidance, or advice contained in this resource. The responsibility for verifying information accuracy for individual use and in individual circumstances lies solely with the audience member. The information in this resource is also not a substitute for legal, medical, or business advice, and is for educational purposes only. Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.