Effect of Modified Transcutaneous Laser Irradiation of Blood on the Healing of Diabetic Foot Ulcers: A Pilot Clinical Trial
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Intravenous laser blood irradiation (ILIB) is a low-intensity systemic laser therapy modality that irradiates blood, promoting biological effects that may enhance tissue repair and healing. Objective. To compare the progression of diabetic foot ulcer healing with modified ILIB combined with conventional treatment vs conventional treatment alone. Methods. This randomized, controlled, single-masked, single-center clinical pilot study included 14 patients allocated to a control group (CG), which received conventional treatment at the study site, or the experimental group, which received modified ILIB combined with conventional treatment (GILIB) (power, 100 mW; wavelength, 660 nm) administered over 10 sessions within 12 days. Sociodemographic and clinical data were collected, ulcer photographs were taken for computerized planimetry analysis, and blood samples were collected for hematological and biochemical analyses. Generalized estimating equation were used to compare groups and time points (days 1, 5, and 12) regarding ulcer areas in centimeters squared, with analyses performed using statistics software at an alpha level of .05. Results. The mean (standard deviation) ulcer duration was 12.0 (8.0) months in the GILIB and 7.7 (7.3) months in the CG, with predominance of superficial, noninfected ulcers (University of Texas grade 1A). Significant differences in mean ulcer area were observed between groups across all time points. Within-group analysis showed a significant reduction in mean ulcer area in the GILIB (P = .02) vs a nonsignificant increase in ulcer area in the CG (P = .23). The GILIB group demonstrated increased leukocyte, band cell, and neutrophil counts, and a reduction in platelets. There were no significant differences in fasting glucose levels. Conclusion. Modified ILIB combined with conventional treatment in individuals with diabetic foot ulcers contributed to a reduction in total ulcer area compared with conventional treatment alone, without clinically significant hematological or biochemical changes.
Conventional management of diabetic foot ulcers (DFUs) is multidisciplinary and involves multifactorial processes related to ulceration, such as glycemic control, pressure reduction, dressing application, debridement of devitalized tissue, wound cleansing, circulation management, application of topical dressings, and infection control.1,2 No ideal approach to managing this complication has been established, however, which necessitates the use of new adjunctive therapies to accelerate healing and prevent amputation,2 including photobiomodulation.
Photobiomodulation, also known as low-level laser therapy, is a therapeutic approach that involves the application of light capable of biostimulating cells and tissues by promoting photochemical and photophysical events.3,4 A wide range of laser application techniques have been used, including local therapy; projection onto internal organs; and intracavitary, transdermal, and intravascular therapy.5 The latter is the method known as intravenous laser irradiation of blood (ILIB), or the intravascular irradiation of blood with a laser. ILIB was first performed in the 1970s by Russian scientists for the treatment of cardiovascular disease.5,6 Originally, this treatment was performed by directly irradiating laser light into the circulating blood using a 2-mm optical fiber inserted through a disposable sterile catheter.
Due to its invasive nature, however, the technique was disadvantageous, leading to the development of new methods, such as modified ILIB.7 This latter method is characterized by its noninvasive nature; it is applied transdermally through a blood vessel.8 Modified ILIB therapy has shown efficacy in systemic, acute, and chronic inflammatory processes,9,10 as well as in promotion of morphological changes in mitochondria,11 leading to increased activity of enzymes against reactive oxygen species and nitrogen species, acting on the redox function of the cell.12,13
Laser light applied to blood vessels can increase the production of arginine and nitric oxide, thus positively influencing several signaling pathways.13 In patients with diabetes, therapies that reduce arginase levels are crucial, because inhibition of this enzyme stimulates nitric oxide synthesis, promoting vasodilation. Additionally, reducing the expression of epidermal growth factor receptor (EGFR) can minimize neuroinflammation, reducing associated secondary damage.14 In summary, arginine stimulates the production of nitric oxide, which improves circulation and promotes wound healing in the feet. In individuals with diabetes, reduced arginase enhances this effect, while EGFR modulation reduces inflammation and damage, promoting tissue repair.
Thus, considering the impaired healing in patients with diabetes due to prolonged inflammation associated with oxidative stress caused by reactive oxygen species, and considering that modified ILIB therapy has no deleterious effects on tissues, it is believed that modified ILIB therapy may act in a complementary fashion in the healing of injuries in these patients.
Despite the beneficial effects of modified ILIB in patients with diabetes already evidenced in the literature,15,16 no studies were found on the use of this technique specifically for the healing of DFUs. Thus, the current study provides innovative findings on the use of modified ILIB for DFU healing, which may contribute to scientific advancement. The aim of the study was to compare the healing progression of DFUs between patients treated with modified ILIB combined with conventional treatment and those who received conventional treatment alone.
Methods
This study was approved by the Ethics Committee for Human Research of the University Hospital at the Federal University of Maranhão (approval number 5,800,722), and it was registered in the Brazilian Clinical Trials Registry of the Ministry of Health (registration number RBR-3v7mrsq). The study was conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent prior to enrollment.
This pilot, randomized, controlled, single-masked clinical trial was conducted from March 1, 2022, through January 31, 2023, at the Diabetic Foot Outpatient Clinic of the Municipal Hospital of Imperatriz (HMI) in the Northeast region of Brazil.
The study population consisted of patients with DFUs followed at the research site. Patients were divided into blocks of 8 participants, with 4 in each group, and were randomly assigned to the experimental group that received modified ILIB therapy in addition to conventional treatment (GILIB) or the control group (CG). Allocation was done through envelope drawing.
The inclusion criteria were as follows: age 18 years or older, clinical diagnosis of type 2 diabetes, follow-up at the study site, ulcer size up to 10 cm, ulcer located on the foot, grade 1 or 2 and stage A or B ulcer according to the University of Texas diabetic wound classification system, and preserved cognitive function as assessed by the Mini-Mental State Examination.
Exclusion criteria were current smoking, alcoholism, neuromuscular or musculoskeletal disorders, peripheral arterial occlusive disease (PAD) as determined by ankle-brachial index (ABI) measurements less than 0.90 or greater than 1.4, and contraindications to ILIB (photosensitivity, pregnancy, complex arrhythmias, heart failure, hematologic malignancies, uncontrolled glaucoma, application over tattoo, pacemaker use, electronic and/or metallic implant at the application site).17
The 2011 American College of Cardiology Foundation/American Heart Association guidelines define as normal ABI values between 1.0 and 1.4. Values less than 0.90 are diagnosed as PAD, and values greater than 1.4 indicate noncompressible arteries.18
Discontinuation criteria for the current study were inability to maintain the standard position for the intervention (supine position, with limbs extended and forearm supinated); topical application to the ulcer site of medication or bandage not used at the study site, as well as oral or injectable medication use (antibiotics and anti-inflammatories); discomfort during the intervention; and nonadherence to intervention and control.
Recruitment took place in the study site waiting room. Sixty-one patients met the inclusion criteria. Of these, 41 were excluded after applying the exclusion criteria, resulting in a total of 20 included patients who were randomized into the 2 groups as shown in the flowchart in Figure 1.
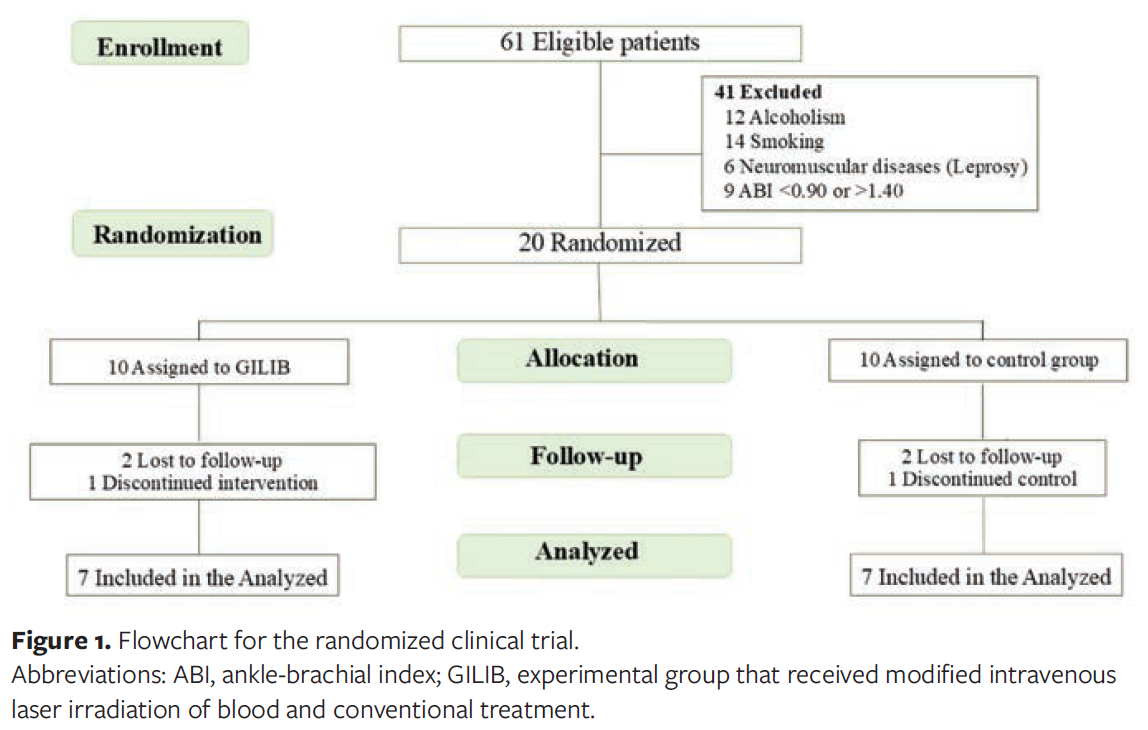
Power was calculated based on the main variables of the groups using G*Power software (version 3.1).19 The chi-square test was used for analysis, with an effect size of 30% and a significance level of P < .05, resulting in a power of 69.35%.
As noted previously, participants were randomized into 2 groups: the GILIB, which underwent modified transcutaneous ILIB on the radial artery in addition to receiving standard treatment at the study site, and the CG, which received only standard treatment at the study site. Both groups received standardized clinical guidance to reduce pressure on the plantar region.
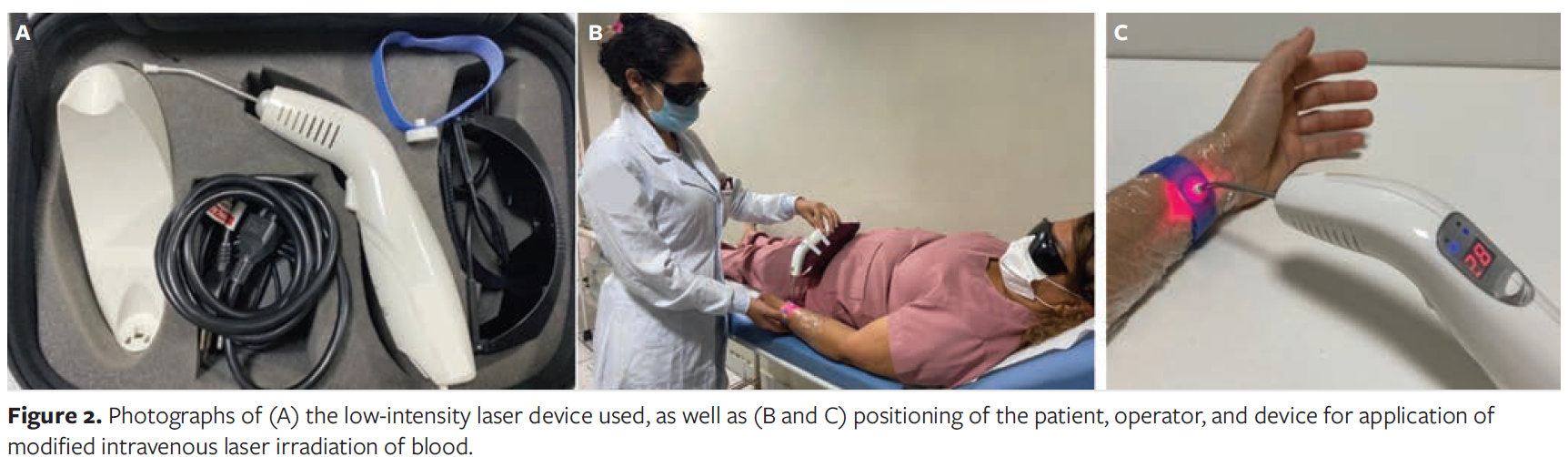
For the intervention, with the participant in the supine position (with limbs extended and the forearm supinated), the laser device was attached on the distal forearm to a wristband designed for this purpose, after first locating and marking the radial artery. The location of the radial artery was determined by palpation (medial to the styloid process of the radius and lateral to the tendon of the flexor carpi radialis) with the aid of a vascular Doppler using a portable ultrasound device. The wrist was then wrapped with polyvinyl chloride film, and the region of the artery was marked with a felt tip pen on paper. The intervention protocol consisted of 10 sessions of 30 minutes each, performed daily over 12 days, with a 2-day interval (weekend) between the fifth and sixth application of the intervention. This protocol was based on studies using this technique.7,11,15,20
The present study used the Therapy EC laser model (DMC Importacao e Exportacao de Equipamentos Ltda), which emits in the red spectrum with a semiconductor diode at a wavelength of 660 nm, a power of 100 mW, a continuous emission mode, a beam cross-sectional area of 0.5026 cm², irradiance of 200 mW/cm², and a dose of 1 J (energy corresponding to 10 seconds of exposure time). For the ILIB application (30 minutes, or 30 × 60 seconds = 1 800 seconds), a total of 180 J was delivered, corresponding to a fluence of 358.14 J/cm².
The device was calibrated by the manufacturer prior to data collection to ensure the power and emission of the required energy for the study. The device used and the method of application are shown in Figure 2.
Data collection was carried out on day 1 (D1) and at follow-up day 5 (D5) and day 12 (D12). Data were obtained using a sociodemographic and clinical questionnaire that included identification data, clinical variables, and DFU characteristics, from which area reduction and healing rate were assessed. Additionally, 10 mL of blood was collected on D1 and D12 for hematological and biochemical analyses, serving as a baseline for evaluating the ulcer healing process.
DFU total area, area reduction, and healing rate were evaluated using images obtained with a EOS Rebel T5i camera (Canon Inc), with a resolution of 18.0 MP. The camera was positioned 30 cm from the ulcer, in ambient light, with the participant’s foot placed on a disposable blue TNT cloth.
Disposable printed rulers, 10 cm in length, were used in all images to calibrate the ImageJ software (version 1.54p), which calculated the total ulcer area from the captured images. This software uses the wound edges for measurement in centimeters squared. Image processing methods or computerized planimetry are considered the standard for assessing wound diameter.21
Wound area reduction corresponds to the progressive decrease in area over time, and it is most accurately assessed in vivo through quantitative measurements of the area taken at different intervals after lesion formation.22
In the current study, area reduction measurements were used to calculate healing indices and percentages (rates) on D5 and D12. The ulcer area reduction index (UARI) is defined as the difference between the initial area (D1) and the final area (D12), divided by the initial area. Thus, the UARI expresses the result of therapeutic follow-up based on the variation in ulcer area, interpreted as follows: UARI equal to 0 indicates unchanged ulcer area; UARI less than 0 indicates an increase in ulcer area; UARI greater than 0 indicates a reduction in ulcer area; and UARI equal to 1 indicates a completely healed ulcer. The ulcer area reduction percentage (UARP) is obtained by multiplying the UARI by 100.23,24
Statistical analyses were performed using SPSS version 24 (IBM Corporation), with alpha set at .05 as the level of statistical significance. Descriptive statistics (absolute and relative frequencies) were used to analyze sociodemographic and clinical data, and the chi-square test was used to verify errors and inconsistencies between groups. After verifying errors and inconsistencies, generalized estimating equation (GEE) models were calculated to compare ulcer area in centimeters squared over time (D1, D5, and D12) in the CG and the GILIB. Comparisons that were significantly different at an alpha level of .05 were subjected to the Bonferroni test. The GEE model is suitable for continuous responses and repeated measures, reflecting the relationship between the independent and variable responses, considering the correlation between measurements at each point in time.25
Results
Of the 20 participants, 4 did not start the intervention due to withdrawal from the study, despite confirming their participation and being randomized. Additionally, 2 participants (1 from each group) did not complete the study protocol. Thus, the final sample consisted of 14 participants who were systematically allocated and randomized between the GILIB (n = 7) and the CG (n = 7).
Regarding sociodemographic data of the sample, 10 of the 14 participants (n = 5 per group) were male (71.4%) and 4 (n = 2 per group) were female (28.6%). The mean (SD) age was 56 (11) years in the GILIB and 60 (10) years in the CG. Illiteracy was prevalent in both the CG (n = 4 [57.1%]) and the GILIB (n = 3 [42.9%]). Four participants in the CG were married (57.1%); in the GILIB, 3 participants were single and 3 were married (42.9% each). In the CG, there was a preponderance of participants earning up to a minimum wage (57.1%). Mixed-race participants predominated in the GILIB (71.4%), while White participants were more prevalent in the CG (42.9%).
All participants presented with 1 ulcer each, all of which were first episodes in that anatomic region. However, 1 participant in the GILIB and 4 participants in the CG reported previous ulcers in another anatomic region (14.3% and 57.1%, respectively).
The mean (SD) ulcer duration prior to inclusion in the study was longer in the GILIB (12 [8.0] months) compared with the CG (7.7 [7.3] months). This difference was not statistically significant. The predominant location of ulcers was in the plantar region, occurring in 6 participants in the GILIB and in 4 in the CG (85.7% and 57.1%, respectively). In both groups, all ulcers were classified according to the University of Texas system. In both groups, 4 participants (57.1%) had grade 1 ulcers and 3 (42.9%) had grade 2 ulcers, while 5 participants (71.4%) presented with stage A and 2 (28.6%) with stage B ulcers. These data indicate that, prior to the intervention, the groups were comparable in terms of ulcer severity and stage.
Regarding the type of product used at the wound site during the study period, essential fatty acids (EFAs) were the most common in both groups, used by all participants in the CG (100%) and 6 (85.7%) in the GILIB. The EFA used was Dersani (Laboratório Daudt Oliveira Ltda). There were no significant differences in sociodemographic characteristics between the groups (P > .05).
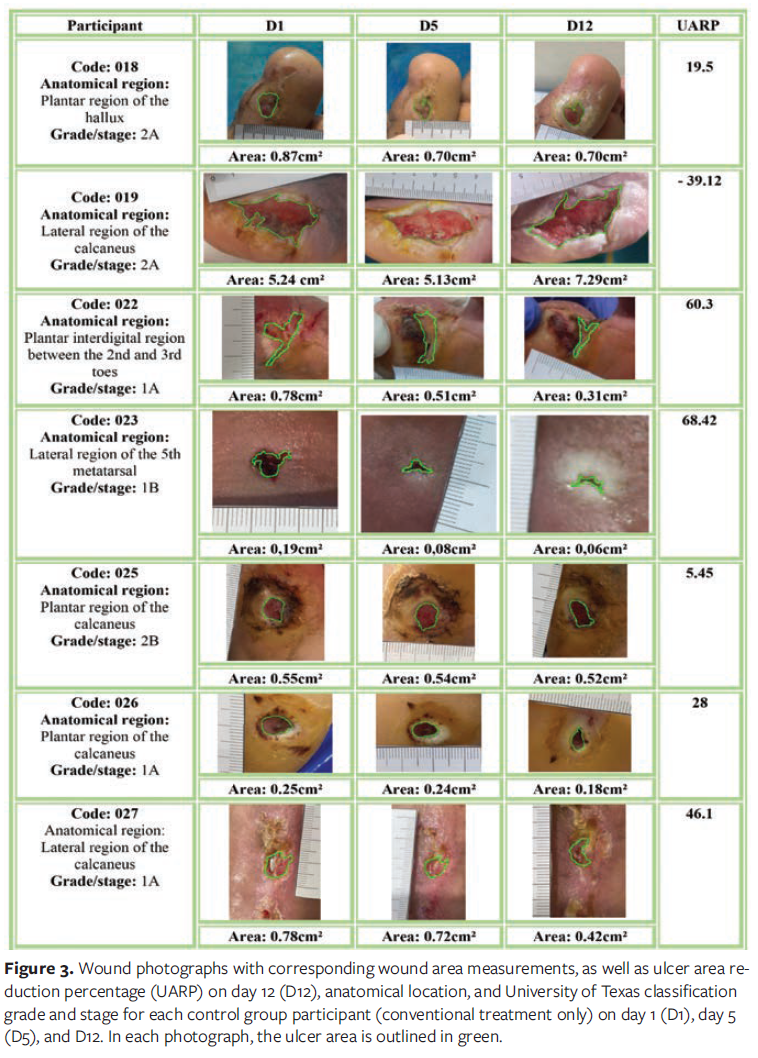
Figure 3 shows the CG participant codes, ulcer location, grade and stage of each wound, the wound area measurements in centimeters squared at each evaluation time point (D1, D5, and D12), and the UARP at D12. Ulcer evolution analysis showed that in the CG, subject 023 had a healing rate of 68.4%—the highest observed in this group. Meanwhile, participant 019 had a negative rate (−39.12%), indicating an increase in ulcer area over the study period. Overall, ulcer reduction was observed in 6 participants (018, 022, 023, 025, 026, and 027) in this group, with varying UARPs.
Figure 4 shows the participant codes, ulcer location, grade and stage of each wound, wound area measurements in centimeters squared at each evaluation time point (D1, D5, and D12), and UARP at D12 for the GILIB participants. In this group, the best result was observed in participant 013, who achieved complete healing. Participants 017 and 021 had the lowest area reduction rates in the group (5.1% and 5.5%, respectively). All participants experienced a reduction in total wound area, with variable UARPs.
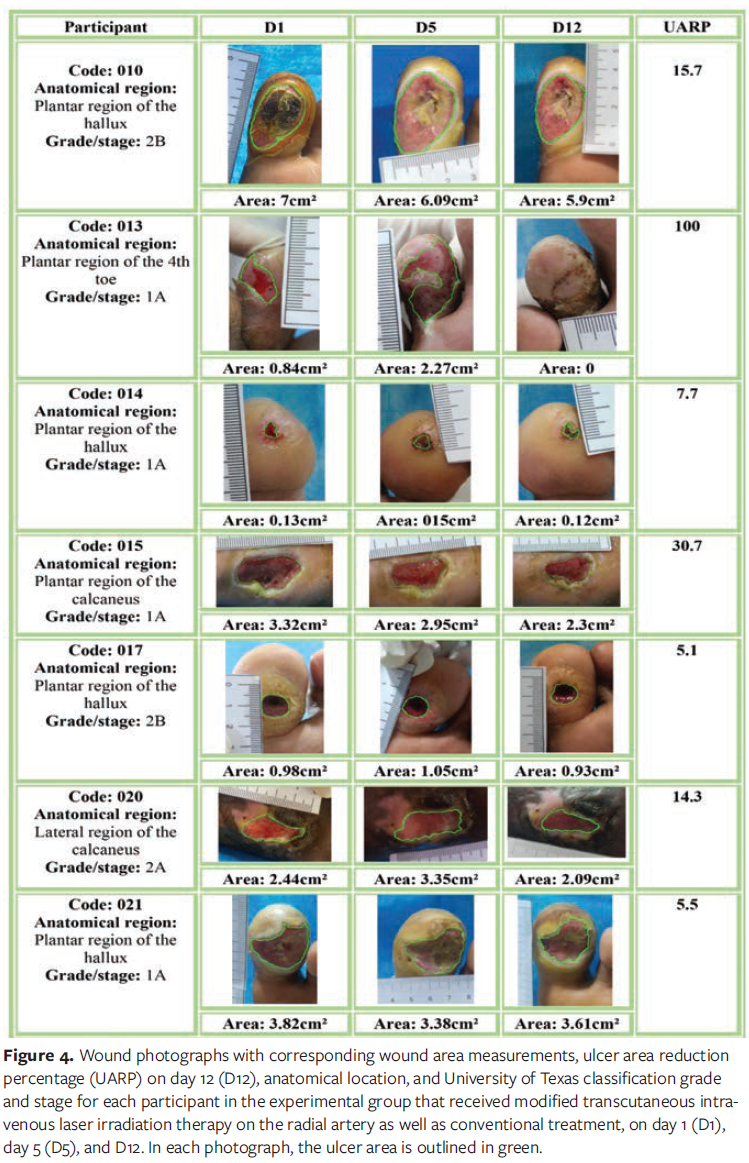
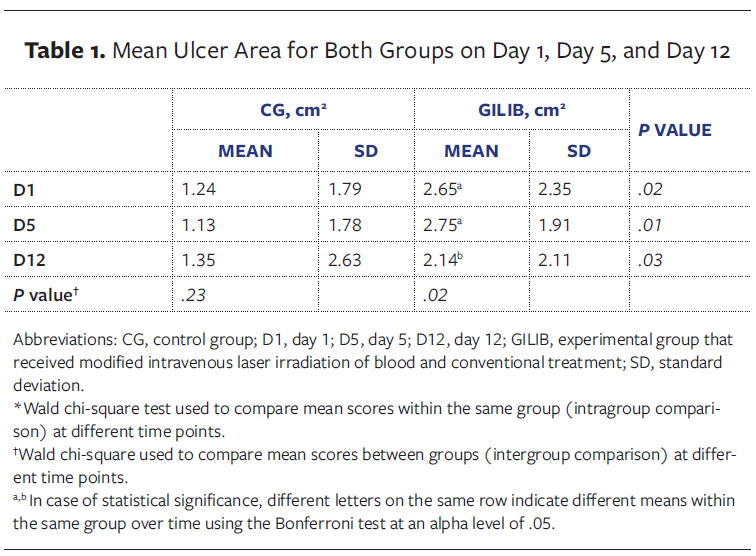
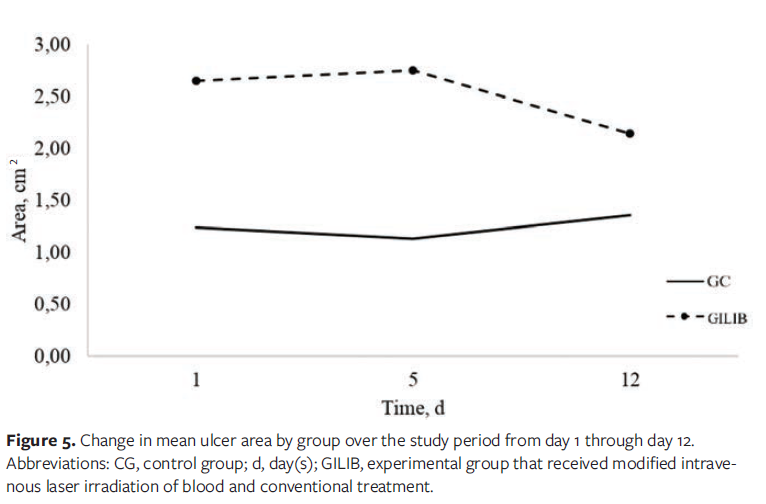
Table 1 shows the mean ulcer area for participants in both groups on D1, D5, and D12. A statistically significant difference was observed between the groups at different time points throughout the study (intergroup comparison). In intragroup comparison, the GILIB showed a statistically significant decrease in mean ulcer area over the study period (P = .02), while the CG group showed a statistically nonsignificant increase in ulcer area (P = .23). Figure 5 shows the change in mean ulcer area by group over the study period, illustrating a clear reduction in area for the GILIB and an increase in area for the CG.
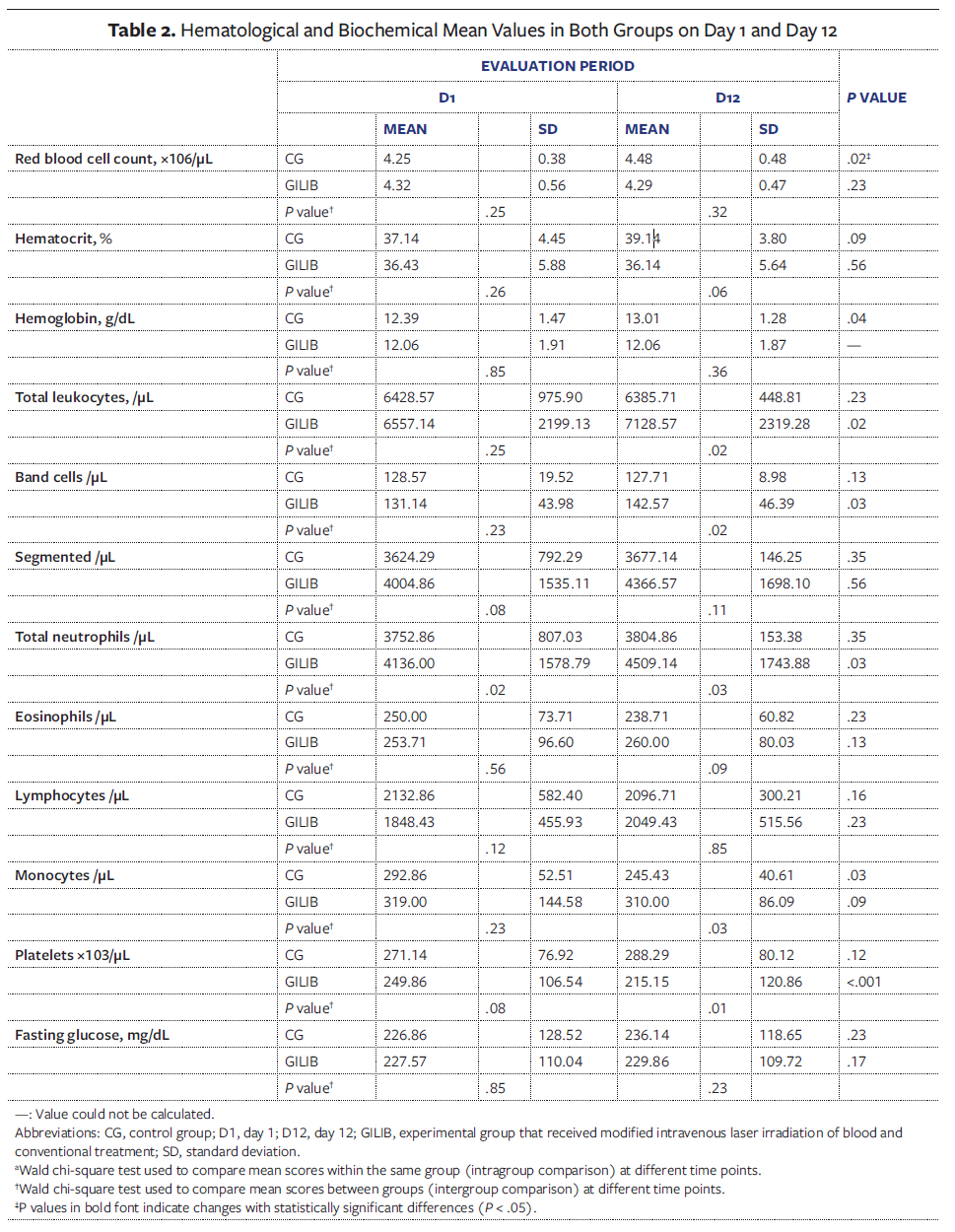
Blood samples were collected on D1 and D12 for hematological and biochemical analyses, including complete blood count and fasting blood glucose tests. The mean hematological and biochemical results of participants in the CG and GILIB at both time points of the study protocol are presented in Table 2.
In the intragroup analysis, the CG showed a significant change between D1 and D12 in red blood cell count, hemoglobin level, and monocyte level, with increased mean values of red blood cells, hemoglobin, and a reduction in monocytes. In the GILIB, significant differences were observed throughout the study period in total leukocyte, band cell, neutrophil, and platelet counts, characterized by increases in leukocytes, band cells, and neutrophils, and a decrease in platelets. However, all observed variations remained within normal reference ranges.
At the beginning of the study (D1), a significant difference between the groups was observed only in the total neutrophil count (P = .02). The remaining hematological parameters did not show relevant differences. In the between-group comparison at D12, significant differences were observed in total leukocyte count (P = .02), band cells (P = .02), total neutrophils (P = .03), monocytes (P = .03), and platelets (P = .01).
Discussion
DFUs represent a significant global health problem; Brazil, in particular, has high amputation rates. Thus, effective therapies for the treatment of these lesions are essential. This study is pioneering in investigating the use of modified ILIB in diabetic patients with foot ulcers. The findings suggest a favorable effect of ILIB on ulcer healing in these patients.
In the current study, participants in the GILIB demonstrated a statistically significant reduction in ulcer area over the 12-day follow-up period (P = .02), after receiving 10 irradiation sessions. In contrast, the CG showed a nonsignificant increase in ulcer area (P = .23).
Transcutaneous intravascular laser irradiation of blood is believed to have significant effects on the healing of diabetic ulcers due to the effects triggered by the photomodification of small amounts of blood irradiated transcutaneously, which subsequently act throughout the entire blood volume.26 Studies indicate that this therapy promotes a decrease in pro-inflammatory cytokines such as transforming necrosis factor-a and interleukin 1b,8,27 a reduction in endothelin-1, an increase in nitric oxide release and arginine production,28,29 and a reduction in arginase levels and EGFR expression.12,13 These changes stimulate gene expression related to tissue regeneration and repair,30,31 thus contributing to the improvement of inflammation, vasodilation, and cellular regeneration.
High arginase activity in patients with type 2 diabetes and hypertension is associated with endothelial dysfunction and reduced nitric oxide availability.32 Thus, the reduction of arginase and EGFR promoted by ILIB may restore vascular function, decrease neuroinflammation, and reduce tissue damage. Studies indicate that arginine influences the release of hormones such as glucagon and insulin, in addition to reducing tissue hypoxia, improving oxygenation, and contributing to the normalization of cellular metabolism.7,33,34 These effects, combined with inflammatory modulation, are among the benefits of ILIB in the healing of diabetic ulcers.
Concerning tissue healing with the use of modified ILIB, a study suggested a beneficial effect of ILIB on the inflammatory and healing processes of neurotendinous injury in a diabetic and renal model.33 The authors of that study evaluated 64 mice with induced renal disease and diabetes that were subjected to injury to the tibial nerve and common calcaneal tendon. The mice were randomized to 1 of 3 treatments: immersion cryotherapy, irradiation with low-intensity laser therapy at the injury site, and modified ILIB. The therapeutic protocol lasted 5 consecutive days.
Diseased animals in both the cryotherapy group and the modified ILIB group exhibited a reduction in inflammatory exudate and better organization of collagen fibers, as well as absence of signs of tissue necrosis. Moreover, a statistically significant reduction in inflammatory parameters was observed following ILIB therapy compared with other treatments. Histopathological analysis revealed that the modified ILIB group demonstrated superior tissue healing outcomes, reinforcing the benefits of this light-based therapy and supporting the positive results observed in this research.
ILIB therapy promotes increased arginine and nitric oxide production, as well as a significant reduction in arginase and EGFR levels, thus contributing to the reduction of neuroinflammation and secondary tissue damage.12,13 Arginine also participates in the release of hormones such as glucagon and insulin, which may reduce tissue hypoxia, improve oxygenation, and normalize tissue metabolism.31,34 Thus, individuals with DFUs may benefit from ILIB therapy, particularly due to their low serum levels of insulin and glucagon and high levels of fatty acids.33 In patients with chronic conditions, such as diabetes and kidney disease, ILIB exerts beneficial effects by increasing oxygen availability, reducing carbon dioxide pressure, normalizing tissue metabolism, promoting cytokine release, modulating the production of growth factors, and stimulating angiogenesis.35
In the current study, the results of hematological and biochemical analyses showed statistically significant changes in total leukocytes, band cells, total neutrophils, and platelets in the GILIB (P < .05). However, it is important to note that all values remained within the established reference ranges for normal parameters, indicating that despite these variations, there was no significant hematological dysregulation or clinical risk associated with ILIB therapy.
These findings reinforce evidence suggesting that ILIB interventions promote adaptive blood alterations, including increased red blood cell counts, a reduced sedimentation rate, and enhanced functions of lymphatic cells and macrophages.9,36,37 Experimental studies have pointed out structural changes in peripheral blood cells following ILIB, such as reticulocytosis and significant increases in neutrophils, eosinophils, basophils, and lymphocytes.36,16 These alterations are attributed to the stimulation of leukopoiesis and the migration of mature cells from the bone marrow, spleen, and pulmonary vessels, reflecting a systemic modulatory effect of the laser on the vascular and immune systems.9,16
The scientific literature recognizes the fundamental role of neutrophils in the innate immune response and tissue repair. Studies have shown that neutrophils are recruited to sites of injury, where they perform essential functions in pathogen defense and the promotion of tissue healing.38,39 Furthermore, activation of the innate immune system, including neutrophil elevation, is associated with resolution of inflammation and initiation of the tissue repair process.39,40
It has been reported that complete repair of injured tissues results from successive alternations of anabolic and catabolic reactions, with leukocytes being among the most important protagonists.40 Two leukocyte populations sequentially dominate the inflammatory events of wound healing: neutrophils and macrophages. Both play a critical role in wound debridement, while macrophages also promote the recruitment and activation of other cells necessary for subsequent stages of wound healing.41
Modified ILIB applied for 30 minutes proved to be more effective in increasing platelet and neutrophil counts compared with 60 minutes of modified ILIB.42 However, in the current study, an increase was observed only in neutrophils. A study involving oncology patients confirmed the efficacy of modified ILIB, with a wavelength of 660 nm applied for 30 minutes and 60 minutes, as an alternative treatment to maintain or increase minimal levels of blood components in patients undergoing chemotherapy.42 The reported efficacy ranged from 85% to 100%, depending on the blood component evaluated: hemoglobin (85% for 30 minutes; 86% for 60 minutes), platelets (100% for both durations), and neutrophils (95% for 30 minutes; 92% for 60 minutes).
Tissue healing is affected by hyperglycemic status. Hyperglycemia is involved in the development of nearly all diabetic complications, including those related to wound healing.43 In the current study, no improvement in glycemic control was observed in participants who received modified ILIB. This finding was also reported in a randomized clinical trial involving 21 participants with type 2 diabetes and periodontitis who were divided into 2 groups (control and intervention/ILIB).44 Mean patient age was 61 years. Participants in the ILIB group underwent 10 modified ILIB laser sessions with a wavelength of 660 nm, combined with conventional periodontal treatment. Regarding fasting glucose and glycated hemoglobin (hemoglobin A1C) levels, the authors of that study did not observe statistically significant improvements in either intergroup or intragroup analyses (P > .05).
In contrast, a study that investigated the use of nonmodified ILIB for glycemic control in patients with type 2 diabetes reported a reduction in mean (SD) plasma glucose level from 190 (17) mg/dL at baseline to 165 (20) mg/dL (measured 30 minutes after a single application) (P = .0018).45 In that study, the assessment was conducted immediately after a single ILIB session, without evaluating the longevity of the treatment. It is believed that this factor, combined with differences in the technique used, may explain why a reduction in glycemia was not observed in the present investigation.
It should be noted that the speculative effects and the lack of robust evidence in the literature have limited the clinical use and establishment of appropriate ILIB parameters.17 Finally, it is important to emphasize that no adverse effects were observed during the study period in the current study; thus, treatment with modified ILIB can be considered a safe adjuvant therapy.
Limitations
This study has several limitations. Individuals with wounds, particularly in the lower extremities, often face difficulties accessing health care services. Additionally, low levels of education and low socioeconomic status may have hindered the ability to recruit a larger sample, resulting in limited statistical power. Another limitation is the lack of patients with larger ulcers or those with ischemic etiology. The limited number of cases in this study may have influenced the results, and this should be considered when interpreting the conclusions and generalizing the findings. The relatively short follow-up period is another limitation, given that typically there are delays in the phases of healing with DFUs. Despite these limitations, the findings are significant and contribute to a better understanding of the effects of modified ILIB in patients with DFUs. This research provides valuable insights into the therapeutic potential of this approach.
The study did not include specific inflammatory markers, which may have provided a deeper understanding of the effects of ILIB. Although blood work was performed for clinical monitoring of the participants, these tests are not sufficient to specifically highlight the anti-inflammatory and tissue microenvironment-modulating mechanisms potentially associated with the therapy. Finally, although all patients received routine local care, debridement was not standardized. This variation in local care may have influenced the results and, thus, represents a limitation of the study.
Conclusions
The current study demonstrated that modified ILIB therapy combined with conventional treatment promoted a significant reduction in total ulcer area in patients with DFUs compared with conventional treatment alone. The findings highlight the potential of modified ILIB as a safe and effective complementary approach in wound care. No hematological or biochemical alterations outside normal ranges were observed in the present study. Furthermore, the increase in total leukocytes, band cells, and neutrophils in the GILIB suggests immunomodulatory effects of the therapy.
Despite these promising results, future studies are needed to evaluate the effects of ILIB therapy on additional biological markers in patients with DFU, such as oxidative stress parameters and growth factors. Future studies also should include longer follow-up periods and larger sample sizes in order to better understand the mechanisms of action and long-term benefits of modified ILIB in the healing of DFUs.
Author and Public Information
Authors: Hellyangela Bertalha Blascovich, MSc1; Lívia Maia Pascoal, PhD1; Ana Cristina Pereira de Jesus Costa, PhD1; Francisca Aline Arrais Sampaio Santos, PhD1; Thiago Moura de Araújo, PhD2; Leonardo Hunaldo dos Santos, PhD1; Marcelino Santos Neto, PhD1; Floriacy Stabnow Santos, PhD1; and Lilia Jannet Saldarriaga Sandoval, PhD3
Affiliations: ¹Federal University of Maranhão, Imperatriz, Brazil; 2University for International Integration of the Afro-Brazilian Lusophony, Redenção, Brazil; 3National University of Tumbes, Tumbes, Peru
Disclosure: The authors disclose no financial or other conflicts of interest.
Ethical Approval: This study was approved by the Ethics Committee for Human Research of the University Hospital at the Federal University of Maranhão (approval number 5,800,722), and it was registered in the Brazilian Clinical Trials Registry of the Ministry of Health (registration number RBR-3v7mrsq).Written informed consent was obtained from the patients for publication of this study and accompanying images. This study was conducted in accordance with the Declaration of Helsinki.
Correspondence: Hellyangela Bertalha Blascovich, MSc; Postgraduate Program in Health and Technology, Federal University of Maranhão, Avenida da Universidade, S/N, Dom Afonso Felipe Gregory, Imperatriz, MA, 65915-240, Brazil; hellyangela.bb@unitins.br
Manuscript Accepted: February 20, 2026.
References
- Andrade LL, Carvalho GCP, Valentim FAAA, Siqueira WA, Melo FMAB, Costa MML. Characteristics and treatment of diabetic foot ulcers in an ambulatory care. Rev Pesqui Cuid Fundam Online. 2019;11(1):124-128. doi:10.9789/2175-5361.2019.v11i1.124-128
- Ananian CE, Davis RD, Johnson EL, et al. Wound closure outcomes suggest clinical equivalency between lyopreserved and cryopreserved placental membranes containing viable cells. Adv Wound Care (New Rochelle). 2019;8(11):546-554. doi:10.1089/wound.2019.1028
- Hanna R, Dalvi S, Benedicenti S, et al. Photobiomodulation therapy in oral mucositis and potentially malignant oral lesions: a therapy towards the future. Cancers (Basel). 2020;12(7):1949. doi:10.3390/cancers12071949
- Tsai SR, Hamblin MR. Biological effects and medical applications of infrared radiation. J Photochem Photobiol B. 2017;170:197-207. doi:10.1016/j.jphotobiol.2017.04.014
- Moskvin SV. Low-level laser therapy in Russia: history, science and practice. J Lasers Med Sci. 2017;8(2):56-65. doi:10.15171/jlms.2017.11
- Chiran DA, Litscher G, Weber M, Ailioaie LM, Ailioaie C, Litscher D. Intravenous laser blood irradiation increases efficacy of etanercept in selected subtypes of juvenile idiopathic arthritis: an innovative clinical research approach. Evid Based Complement Alternat Med. 2013;2013:168134. doi:10.1155/2013/168134
- Kazemikhoo N, Sarafnejad AF, Ansari F, Mehdipour P. Modifying effect of intravenous laser therapy on the protein expression of arginase and epidermal growth factor receptor in type 2 diabetic patients. Lasers Med Sci. 2016;31(8):1537-1545. doi:10.1007/s10103-016-2012-x
- Meneguzzo DT, Ferreira LS, De Carvalho EM, Nakashima CF, Hamblin MR. Intravascular laser irradiation of blood. In: Hamblin MR, de Sousa MVP, Agrawal T, eds. Handbook of Low-Level Laser Therapy. Jenny Stanford Publishing; 2017:943-945.
- Mikhaylov VA. The use of intravenous laser blood irradiation (ILBI) at 630-640 nm to prevent vascular diseases and to increase life expectancy. Laser Ther. 2015;24(1):15-26. doi:10.5978/islsm.15-OR-02
- Silva HCDA, Acioli S, Fuly PSC, Nóbrega MML, Lins SMSB, Menezes HF. Construction and validation of nursing diagnoses for people with diabetic foot ulcers. Rev Esc Enferm USP. 2022;56:e20220022. doi:10.1590/1980-220X-REEUSP-2022-0022en
- Karu TI. Mitochondrial signaling in mammalian cells activated by red and near-IR radiation. Photochem Photobiol. 2008;84(5):1091-1099. doi:10.1111/j.1751-1097.2008.00394.x
- Kazemi Khoo N, Iravani A, Arjmand M, et al. A metabolomic study on the effect of intravascular laser blood irradiation on type 2 diabetic patients. Lasers Med Sci. 2013;28(6):1527-1532. doi:10.1007/s10103-012-1247-4
- Huang SF, Tsai YA, Wu SB, Wei YH, Tsai PY, Chuang TY. Effects of intravascular laser irradiation of blood in mitochondria dysfunction and oxidative stress in adults with chronic spinal cord injury. Photomed Laser Surg. 2012;30(10):579-586. doi:10.1089/pho.2012.3228
- Xu Y, Lin Y, Gao S. Study on the selection of laser wavelengths in the intravascular low-level laser irradiation therapy. Lasers Med Sci. 2015;30(4):1373-1376. doi:10.1007/s10103-015-1732-7
- da Silva Leal MV, Lima MO, Nicolau RA, et al. Effect of modified laser transcutaneous irradiation on pain and quality of life in patients with diabetic neuropathy. Photobiomodul Photomed Laser Surg. 2020;38(3):138-144. doi:10.1089/photob.2019.4714
- Tomé RFF, Silva DFB, Santos CAO, Neves GV, Rolim AKA, Gomes DQC. ILIB (intravascular laser irradiation of blood) as an adjuvant therapy in the treatment of patients with chronic systemic diseases—an integrative literature review. Lasers Med Sci. 2020;35(9):1899-1907. doi:10.1007/s10103-020-03100-4
- Rezende L, Lenzi J. Eletrotermofototerapia em Oncologia: da Evidência à Prática Clínica. 1st ed. Rio de Janeiro: Thieme Revinter; 2019.
- Rooke TW, Hirsch AT, Misra S, et al. 2011 ACCF/AHA focused update of the guideline for the management of patients with peripheral artery disease (updating the 2005 guideline): A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2011;124(18):2020-2045. doi:10.1161/CIR.0b013e31822e80c3
- Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. 2007;39(2):175-191. doi:10.3758/bf03193146
- Houreld NN, Abrahamse H. Laser light influences cellular viability and proliferation in diabetic-wounded fibroblast cells in a dose- and wavelength-dependent manner. Lasers Med Sci. 2008;23(1):11-18. doi:10.1007/s10103-007-0445-y
- Foltynski P, Ladyzynski P, Ciechanowska A, Migalska-Musial K, Judzewicz G, Sabalinska S. Wound area measurement with digital planimetry: improved accuracy and precision with calibration based on 2 rulers. PLoS One. 2015; 7;10(8):e0134622. doi: 10.1371/journal.pone.0134622
- Lammers G, Verhaegen PD, Ulrich MMW, et al: An overview of methods for the in vivo evaluation of tissue-engineered skin constructs. Tissue Eng Part B Rev. 2011;17(1):33-55. doi:10.1089/ten.TEB.2010.0473
- Taradaj J, Shay B, Dymarek R, et al. Effect of laser therapy on expression of angio- and fibrogenic factors, and cytokine concentrations during the healing process of human pressure ulcers. Int J Med Sci. 2018;15(11):1105-1112. doi:10.7150/ijms.25651
- Mehl AA, Schneider B Jr, Schneider FK, Carvalho BHK. Measurement of wound area for early analysis of the scar predictive factor. Rev Lat Am Enfermagem. 2020;28:e3299. doi:10.1590/1518-8345.3708.3299
- Costa PRF, Assis AMO, Cunha CM, et al. Hypertriglyceridemic waist phenotype and changes in the fasting glycemia and blood pressure in children and adolescents over one-year follow-up period. Arq Bras Cardiol. 2017;109(1):47-53. doi:10.5935/abc.20170067
- Malavazzi TCS, Andreo L, Martinelli A, et al. Preventive and therapeutic vascular photobiomodulation decreases the inflammatory markers and enhances the muscle repair process in an animal model. J Photochem Photobiol B. 2024;256:112921. doi:10.1016/j.jphotobiol.2024.112921
- Zhevago NA, Samoilova KA. Pro- and anti-inflammatory cytokine content in human peripheral blood after its transcutaneous (in vivo) and direct (in vitro) irradiation with polychromatic visible and infrared light. Photomed Laser Surg. 2006;24(2):129-139. doi:10.1089/pho.2006.24.129
- Derkacz A, Szymczyszyn A, Szahidewicz-Krupska E, Protasiewicz M, Poręba R, Doroszko A. Effect of endovascular coronary low-level laser therapy during angioplasty on the release of endothelin-1 and nitric oxide. Adv Clin Exp Med. 2017;26(4):595-599. doi:10.17219/acem/62535
- Kazemikhoo N, Ansari F, Nilforoushzadeh. The hypoglycemic effect of intravenous laser therapy in diabetic mellitus type 2 patients: a systematic review and meta-analyses. Med Clin Rev. 2016;1(1):7. doi:10.21767/2471-299X.1000007
- Karu T. Photobiology of low-power laser effects. Health Physics. 1989;56(5):691-704.
- Makela AM. Role of L-arginine in the biological effects of blue light. In: Proceedings SPIE 5968: Laser Florence 2004. A Window on the Laser Medicine World. 2005;596805:32-44. doi:10.1117/12.660038
- Shosha E, Xu Z, Narayanan SP, Lemtalsi T, Fouda AY, Rojas M, Xing J, Fulton D, Caldwell RW, Caldwell RB. Mechanisms of Diabetes-Induced Endothelial Cell Senescence: Role of Arginase 1. Int J Mol Sci. 2018; 19(4):1215. Doi:10.3390/ijms19041215
- Silva PH, Silva PH, Facco GG, Corazza AV, da Silva JG, Silva IS. Effect of electrophysical resources on healing of neurotendinous injury in an experimental model of type I diabetes and kidney disease. Acta Cir Bras. 2022;37(4):e370402. doi:10.1590/acb370402
- Dunnill C, Patton T, Brennan J, et al. Reactive oxygen species (ROS) and wound healing: the functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int Wound J. 2017;14(1):89-96. doi:10.1111/iwj.12557
- Razzaghi MR, Ghanei E, Malekian S, Mazloomfard MM. Intravenous laser therapy in patients with acute kidney injury: a randomized clinical trial. J Lasers Med Sci. 2021;12:e49. doi:10.34172/jlms.2021.49
- Moskvin S, Askhadulin E, Kochetkov A. Low-level laser therapy in prevention of the development of endothelial dysfunction and clinical experience of treatment and rehabilitation of COVID-19 patients. Rehabil Res Pract. 2021; 6626932. Published 2021 Jan 26. doi:10.1155/2021/6626932
- Sarycheva, T.G.; Tsybzhitova, E.B.; Popova, O.V.; Aleksandrov, O.V. Morphometry and electrophoretic mobility of red blood cells from patients with asthma in the intravenous blood laser irradiation. Klin. Lab. Diagn. 2009, 3, 13–14. https://pubmed.ncbi.nlm.nih.gov/19391239/
- Yang L, Shi F, Cao F, et al. Neutrophils in tissue injury and repair: molecular mechanisms and therapeutic targets. MedComm (2020). 2025;6(5):e70184. doi:10.1002/mco2.70184
- Peiseler M, Kubes P. More friend than foe: the emerging role of neutrophils in tissue repair. J Clin Invest. 2019;129(7):2629-2639. doi:10.1172/JCI124616
- Anitua E, Troya M, Alkhraisat MH. Beyond killing: the overlooked contribution of neutrophils to tissue repair. Int J Mol Sci. 2025;26(17):8669. doi:10.3390/ijms26178669
- Balbino CA, Pereira LM, Curi R. Mecanismos envolvidos na cicatrização: uma revisão. Rev Bras Cienc Farm. 2005;41(1):27-51. doi:10.1590/S1516-93322005000100004
- Oliveira De Lima, T, Spin, M, Lizarelli, RFZ, Minicucci, EM, Freitas, KABS, Bocchi, SCM. Transcutaneous laser therapy for hematopoietic adverse effects of antineoplastic chemotherapy: A randomized clinical trial. Nursing Edição Brasileira. 2022; 25(288), 7826–7840. doi:10.36489/nursing.2022v25i288p7826-7840
- Saeedi P, Petersohn I, Salpea P, et al; IDF Diabetes Atlas Committee. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. 2019;157:107843. doi:10.1016/j.diabres.2019.107843
- da Silva Júnior FL, Silva DNA, Azevedo MLS et al. Efficacy of ILIB on periodontal clinical parameters and glycemic control in patients with periodontitis and type II diabetes—randomized clinical trial. Lasers Med Sci. 2022; 37: 1945–1952. doi: 10.1007/s10103-021-03455-2
- Kazemi Khoo N, Iravani A, Arjmand M. et al. A metabolomic study on the effect of intravascular laser blood irradiation on type 2 diabetic patients. Lasers Med Sci. 2013: 28;1527–1532 (2013). doi: 10.1007/s10103-012-1247-4


















