Emergency Cardiac Surgery After Percutaneous Coronary Intervention: Insights From the PROGRESS-Complications Registry
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/25.00383. Epub April 16, 2026.
Abstract
Objectives. Emergency surgery is an infrequent but severe complication of percutaneous coronary intervention (
Methods. The authors analyzed the clinical characteristics and outcomes of 15 patients who underwent emergency surgery after PCI from the PROGRESS-COMPLICATIONS registry.
Results. Of 18 691 patients who underwent PCI at 2 tertiary care centers between 2016 and 2023, 15 (0.08%) required emergency surgery: 14 underwent coronary artery bypass graft surgery (CABG) and 1 underwent aortic valve replacement and CABG. Patients had high prevalence of comorbidities such as hypertension (93.3%), diabetes mellitus (73.3%), dyslipidemia (93.3%), and prior heart failure (53.3%). The most common presentations included non-ST segment elevation acute myocardial infarction (40.0%) and stable angina (33.3%). Target lesions were complex and often had moderate/severe calcification (60.0%) or involved a bifurcation (40.0%). Mechanical circulatory support was used in 53.3% (intra-aortic balloon pump 12.5%; Impella 2.5 [Abiomed] 12.5%; Impella 5.0 37.5%; Impella CP 12.5%; venoarterial extracorporeal membrane oxygenation 25.0%). In-hospital mortality was 33.3%. The most common indications for emergent CABG were coronary dissection (46.7%), aortocoronary dissection (26.7%), and coronary perforation (26.7%). Post-CABG complications included death (33.3%), arrhythmia (33.3%), hemodynamic instability (26.7%), and cardiogenic shock (13.3%). The median hospital stay was 10.0 days (6.0-18.5). During a median follow-up of 24 months, 53.3% of patients experienced major adverse cardiovascular events (MACE), 46.7% died, and 26.7% required target vessel revascularization.
Conclusions. Patients who underwent emergency surgery after PCI had multiple comorbidities, complex coronary anatomy, and high incidence of MACE.
Introduction
Emergency surgery after percutaneous coronary intervention (PCI) is infrequent (reported incidence 0.07-0.12%)1,2
There is limited data on the characteristics and outcomes of patients who require emergency surgery after PCI. We examined the incidence, clinical features, and outcomes of patients who underwent emergency cardiac surgery after PCI at 2 high-volume tertiary care centers.
Methods
Of 18 691 PCI cases performed at 2 tertiary care PCI centers (Abbott Northwestern Hospital and the University of Washington) between 2016 and 2023, 15 patients (0.08%) required emergency cardiac surgery during the index hospitalization due to PCI-related complications and were included in the PROGRESS-COMPLICATIONS Registry (Prospective Global Registry for the Study of Complications, NCT05100940). We examined the clinical characteristics, angiographic and procedural features, and in-hospital and long-term outcomes of patients who underwent emergency surgery as a complication of PCI. The Minneapolis Heart Institute Foundation hosted REDCap (Research Electronic Data Capture) electronic data capture tools, which were used to collect and manage study data.3 The institutional review board at Minneapolis Heart Institute Foundation (MHIF) approved the
Emergency surgery was defined as a non-elective cardiac surgical intervention (eg, coronary artery bypass grafting [CABG] or other cardiac surgery) performed because of PCI-related complications such as dissection, perforation, or hemodynamic instability.4 Confirmation of emergency surgery was based on operative reports and clinical documentation, with the date, time, and type of surgery abstracted for each case.
Technical success of PCI was defined as revascularization with less than 30% residual diameter stenosis and restoration of Thrombolysis in Myocardial Infarction (TIMI) grade 3 antegrade flow in the treated segment.5 Procedural success was defined as achievement of technical success without the occurrence of any major adverse cardiovascular events (MACE). In-hospital MACE was defined as a composite of all-cause mortality, myocardial infarction (MI), emergency surgery, stroke, urgent repeat revascularization, or major bleeding.6 MI was adjudicated according to the Fourth Universal Definition of MI, specifically as type 4a MI related to PCI. Major bleeding events were classified according to the Bleeding Academic Research Consortium (BARC) criteria as type 3 or higher.7
Hypotension was defined as a sustained systolic blood pressure of less than 90 mm Hg during the PCI procedure requiring pharmacologic or mechanical hemodynamic support.8 Equipment loss was defined as the unintended retention or loss of interventional equipment, such as a guidewire or device component, within the coronary vasculature during PCI. Dissection refers to dissection of the target coronary vessel identified during the perioperative period.9
Postoperative complications were defined as events occurring during the index hospitalization following emergency cardiac surgery. Arrhythmia was defined as a clinically significant rhythm disturbance requiring medical or electrical intervention.10 Renal failure was defined as a new acute kidney injury requiring medical management or dialysis.11
Thrombus was identified based on angiographic findings as determined by the treating operators. Intracoronary imaging was not systematically used for thrombus assessment. Thrombus was defined as persistent angiographic thrombus observed during the PCI procedure, including both thrombus present prior to PCI and thrombus identified during the intervention.9
Follow-up MACE was defined as a composite of death, MI, stroke, or target vessel revascularization by either PCI or CABG.5
Continuous variables were reported as mean ± standard deviation or median with interquartile range (IQR). Categorical variables were expressed as frequencies and percentages. Time-to-event outcomes were assessed using the Kaplan-Meier method, and results were presented descriptively given the small sample size. All statistical analyses were conducted using R software, version 4.4.1 (R Foundation for Statistical Computing).
Results
Between 2016 and 2023, the incidence of emergency cardiac surgery after PCI was 0.08% (15 of 18 691 PCIs): 14 underwent emergency CABG and 1 underwent aortic valve replacement plus CABG. Baseline characteristics are summarized in Table 1. The mean age was 68.4 ± 8.5 years, and 66.7% were male. Comorbidities included hypertension in 93.3%, diabetes mellitus in 73.3%, dyslipidemia in 93.3%, and prior heart failure in 53.3%. Other clinical histories included prior MI (46.7%), prior PCI (60.0%), and prior CABG (6.7%). The mean body mass index was 30.58 ± 6.75 kg/m2, and the mean left ventricular ejection fraction was 49.4 ± 13.3%. Most patients presented with non-ST segment elevation acute MI (40.0%), followed by stable angina in 33.3%, unstable angina in 13.3%, ST-segment elevation MI in 6.7%, and asymptomatic ischemia in 6.7%.
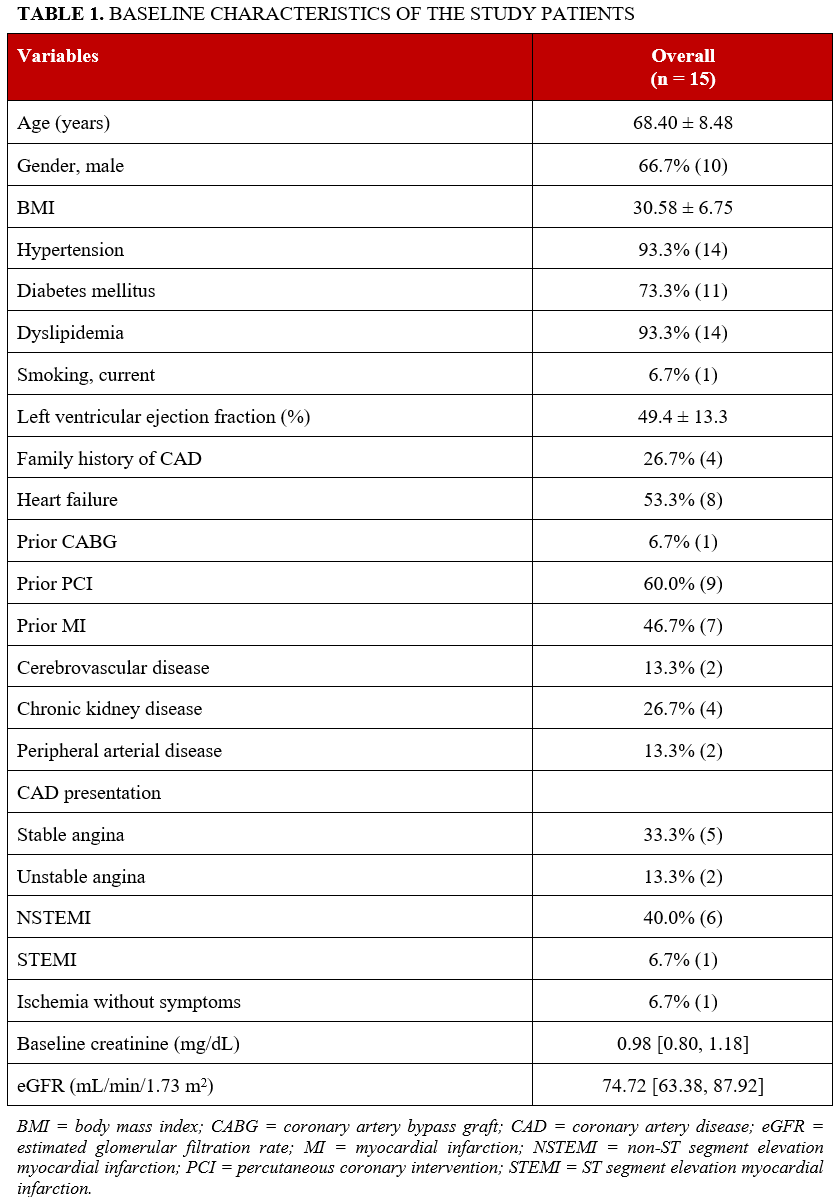
Angiographic characteristics are summarized in Table 2. All patients underwent PCI via femoral access. The treated vessels were the left main coronary artery (33.3%), left anterior descending artery (33.3%), and right coronary artery (26.7%), with the left circumflex artery treated in 6.7% of cases. Bifurcation lesions were present in 40.0%, moderate to severe calcification in 60.0%, moderate to severe tortuosity in 20.0%, in-stent restenosis (ISR) in 13.3%, and thrombus-containing lesions in 13.3% (there were no cases of stent thrombosis). The median lesion length was 20.0 (IQR: 10.0-57.5) mm, and the median target vessel diameter was 3.25 (2.81-3.88) mm. The Medina classification among bifurcation lesions was 1,1,1 in 50.0%; 0,1,1 in 16.7%; and 1,0,0 in 33.3%.
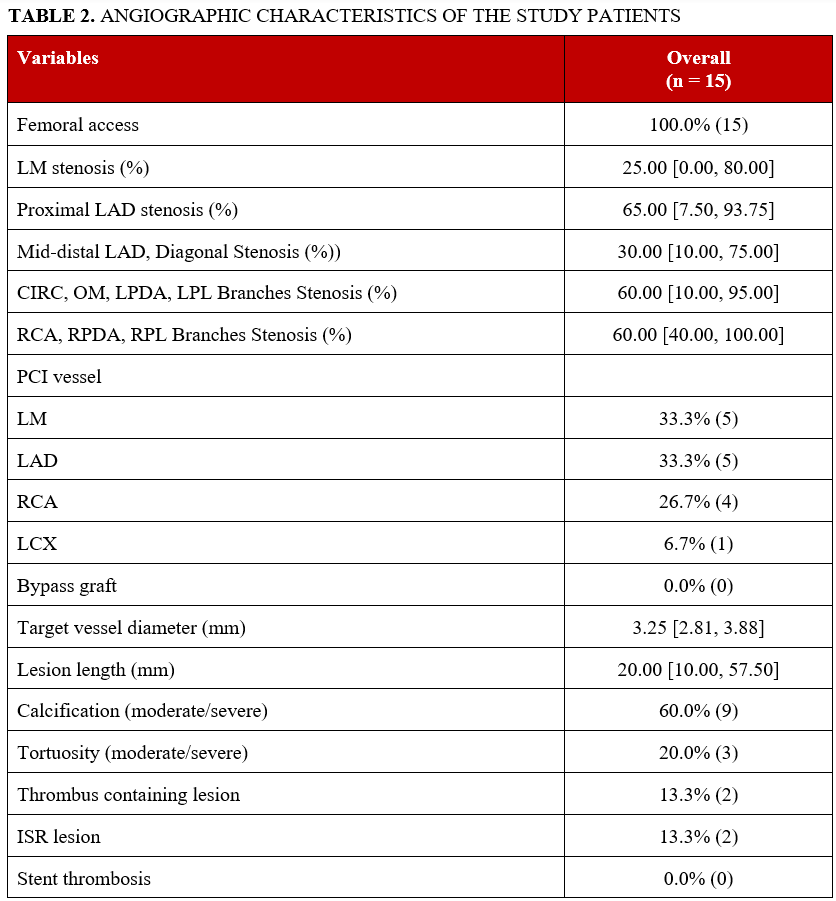
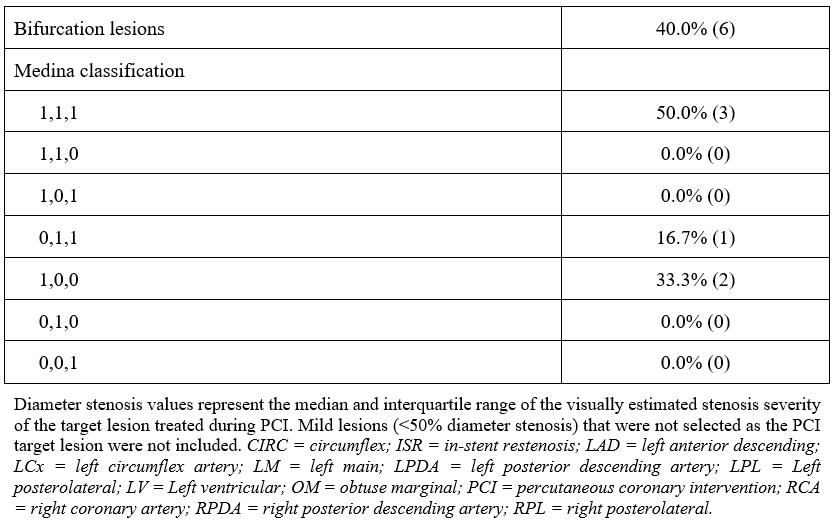
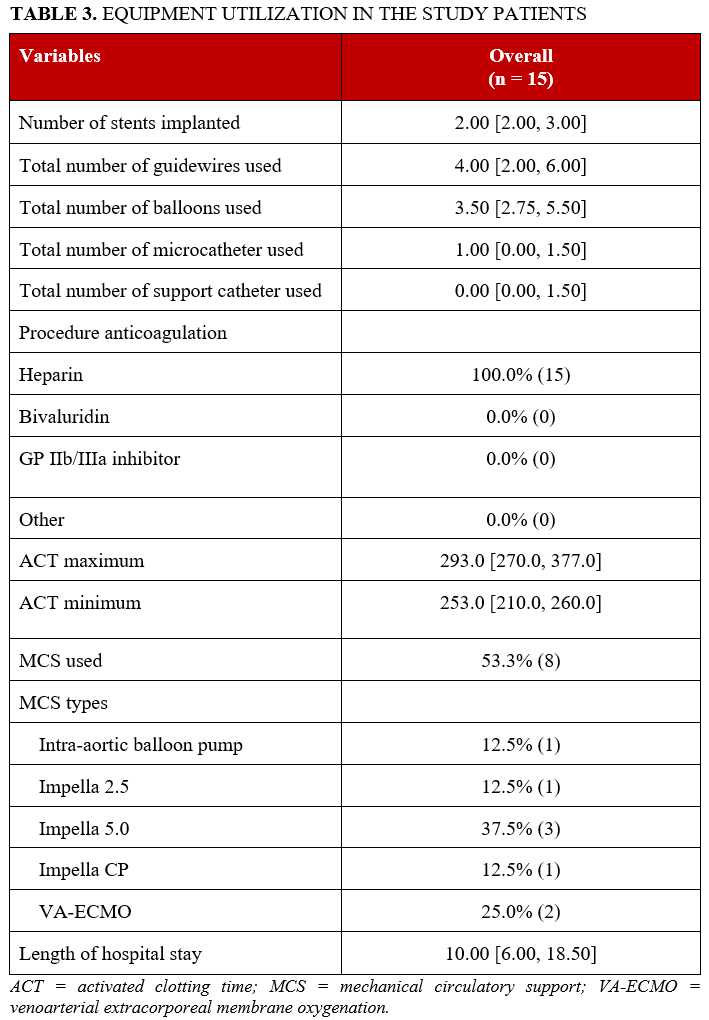
Procedural equipment use is summarized in Table 3. The median number of stents implanted was 2.0 (2.0-3.0), the median number of guidewires was 4.0 (2.0-6.0), the median number of balloons was 3.5 (2.75-5.5), and the median number of microcatheters was 1.0 (0.0-1.5). All patients received heparin for anticoagulation. The median minimum activated clotting time (ACT) was 253 (210-260) seconds, and the median maximum ACT was 293 (270-377) seconds. Mechanical circulatory support (MCS) was used in 53.3% of patients. Devices included intra-aortic balloon pump (12.5%), Impella 2.5 (Abiomed) (12.5%), Impella 5.0 (37.5%), Impella CP (12.5%), and venoarterial extracorporeal membrane oxygenation (25.0%). The median hospital stay was 10.0 (6.0-18.5) days.
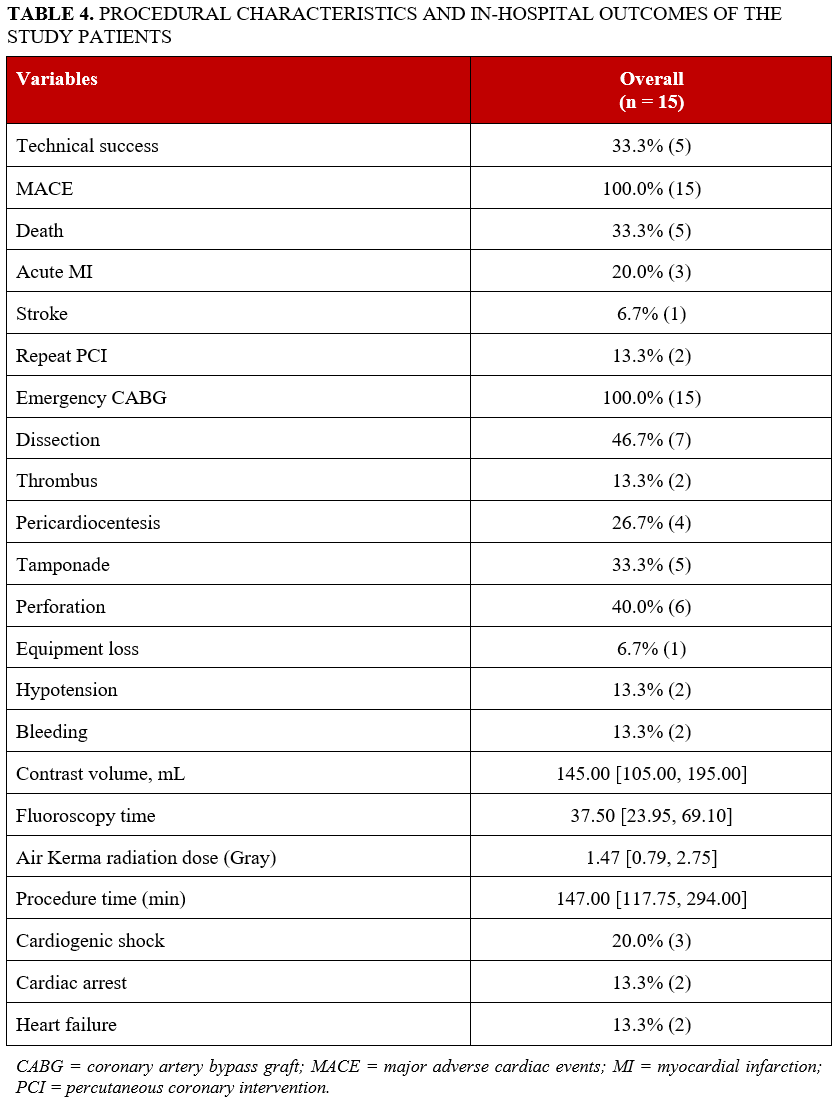
Procedural characteristics and in-hospital outcomes are detailed in Table 4. Technical success was 33.3%, with an in-hospital mortality rate of 33.3%. Indications for CABG included coronary dissection (46.7%), aortocoronary dissection (26.7%), and perforation (26.7%). Preoperative complications included tamponade (33.3%), pericardiocentesis (26.7%), dissection (46.7%), thrombus (13.3%), bleeding (13.3%), hypotension (13.3%), cardiac arrest (13.3%), and cardiogenic shock (20.0%) (Table 5). The median procedure time was 147 (118-294) minutes, median fluoroscopy time was 37.5 (23.95-69.10) minutes, median contrast volume was 145 (105-195) mL, and median air kerma radiation dose was 1.47 (0.79-2.75) Gray.

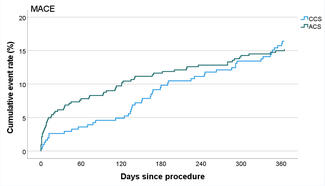
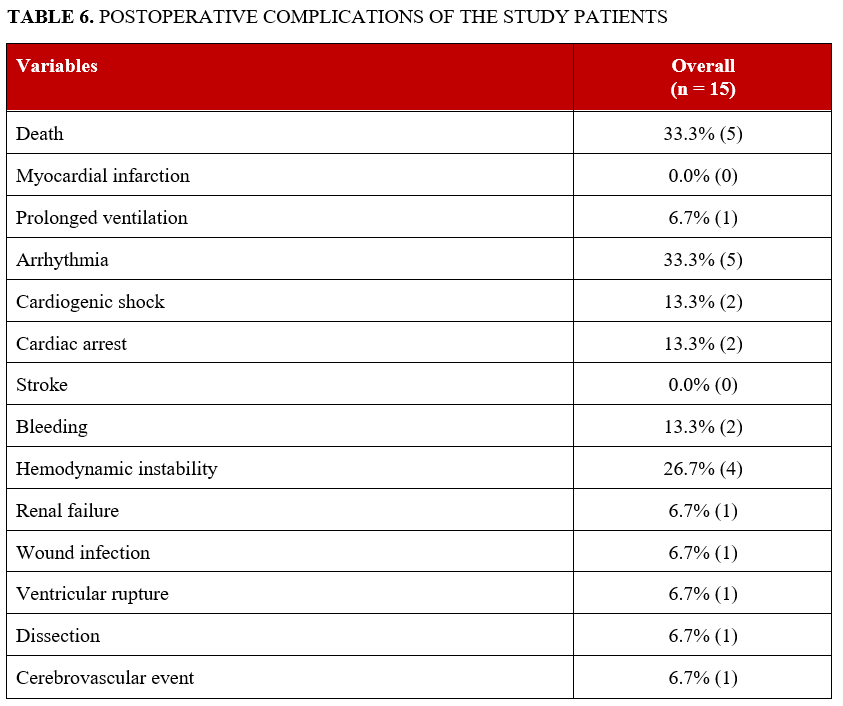
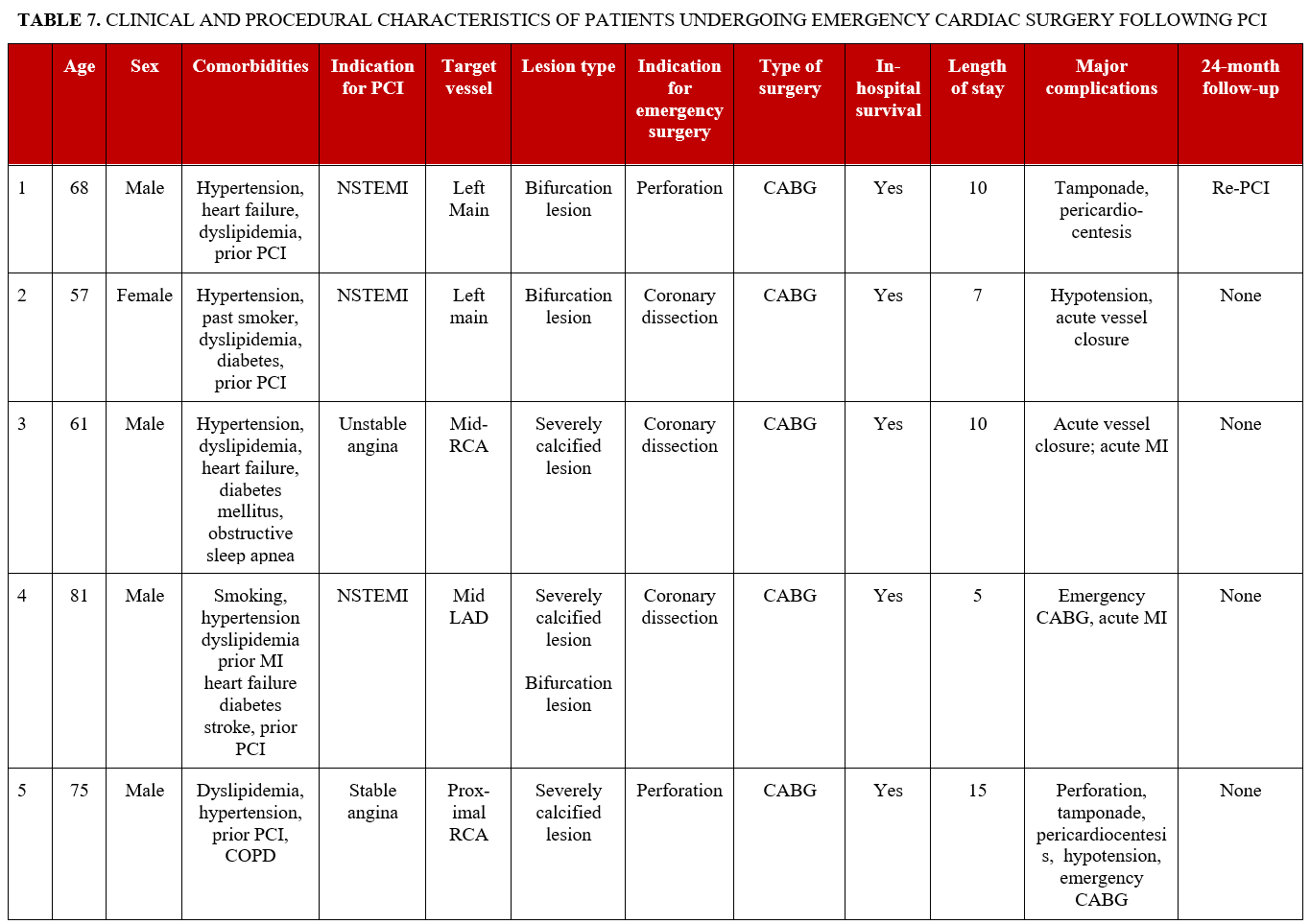
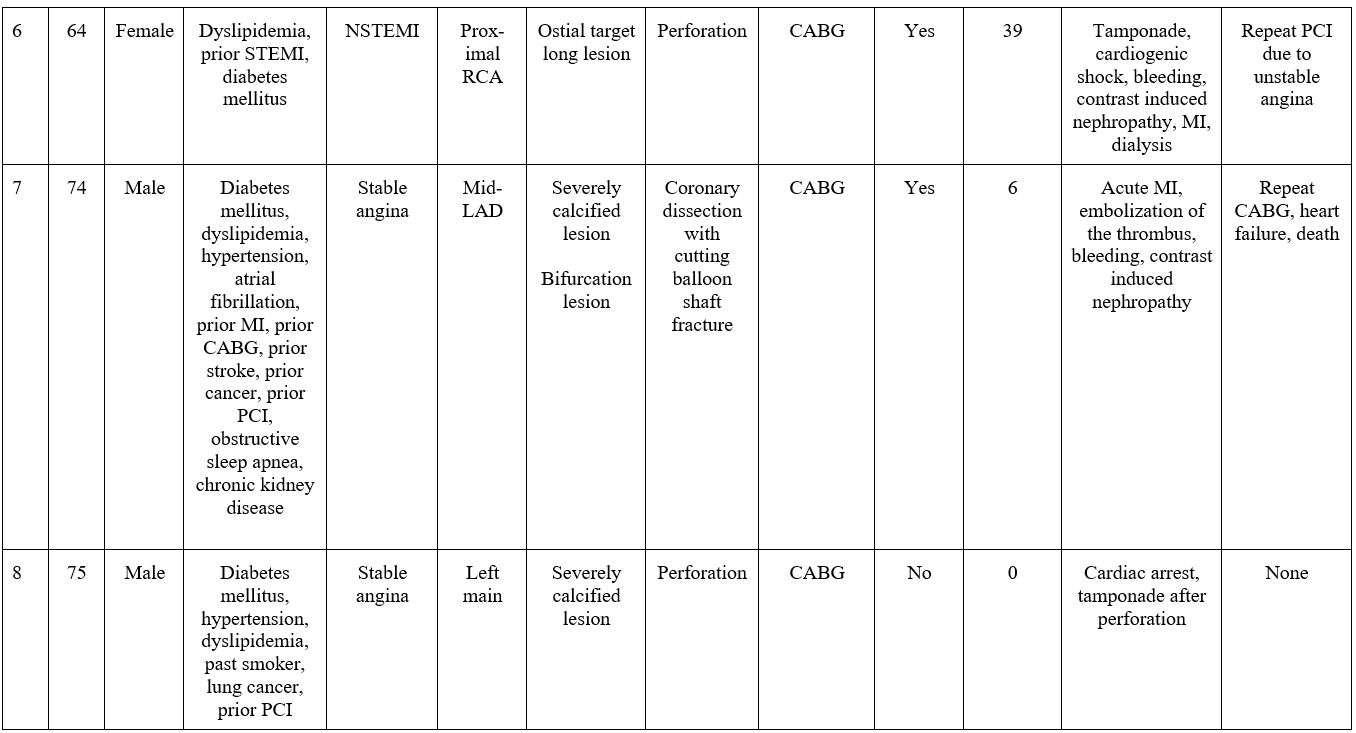
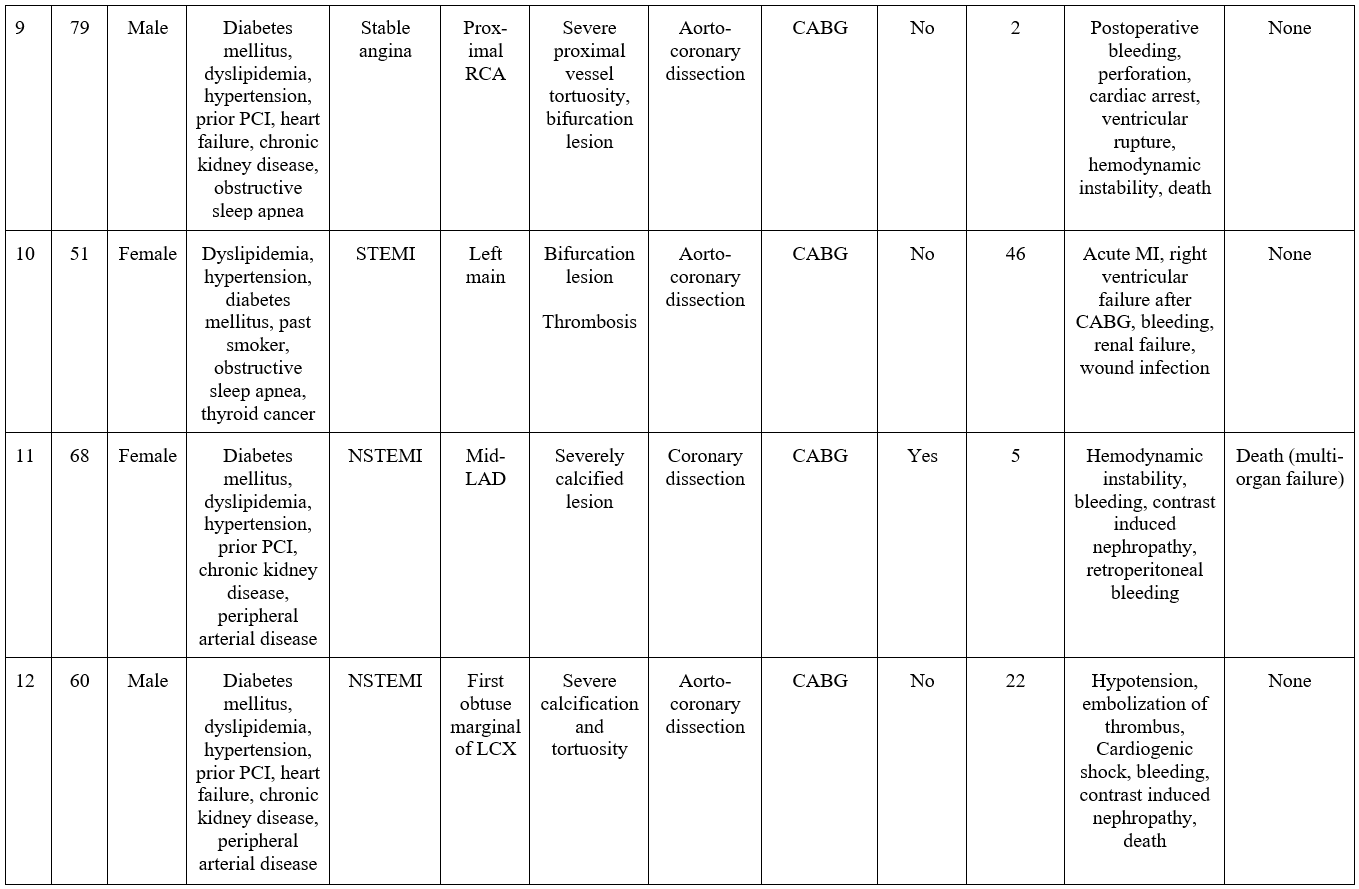
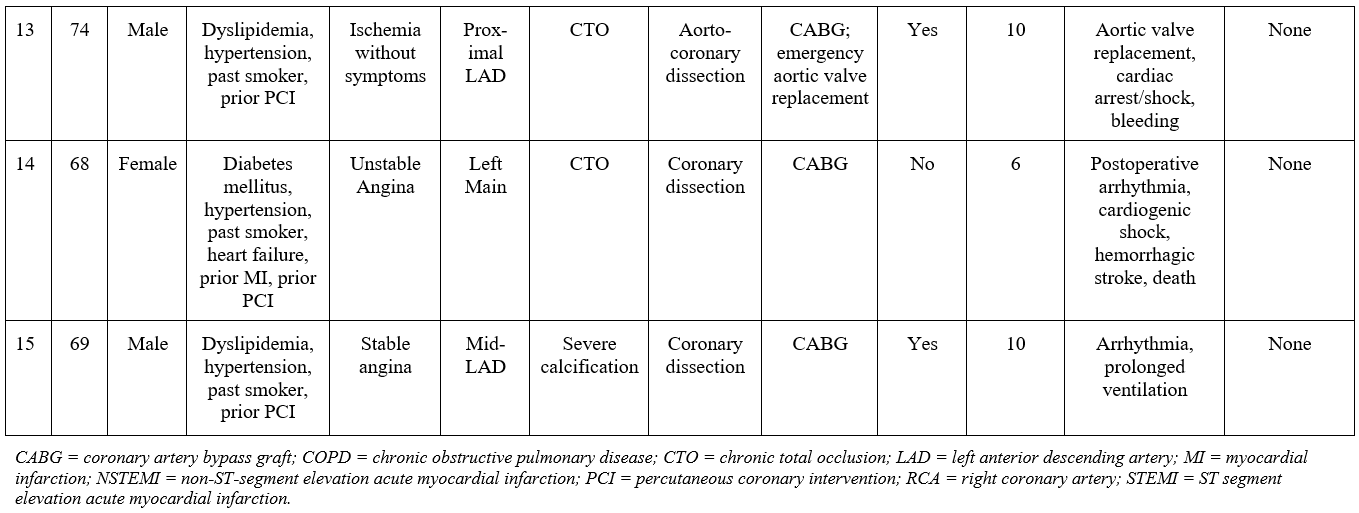
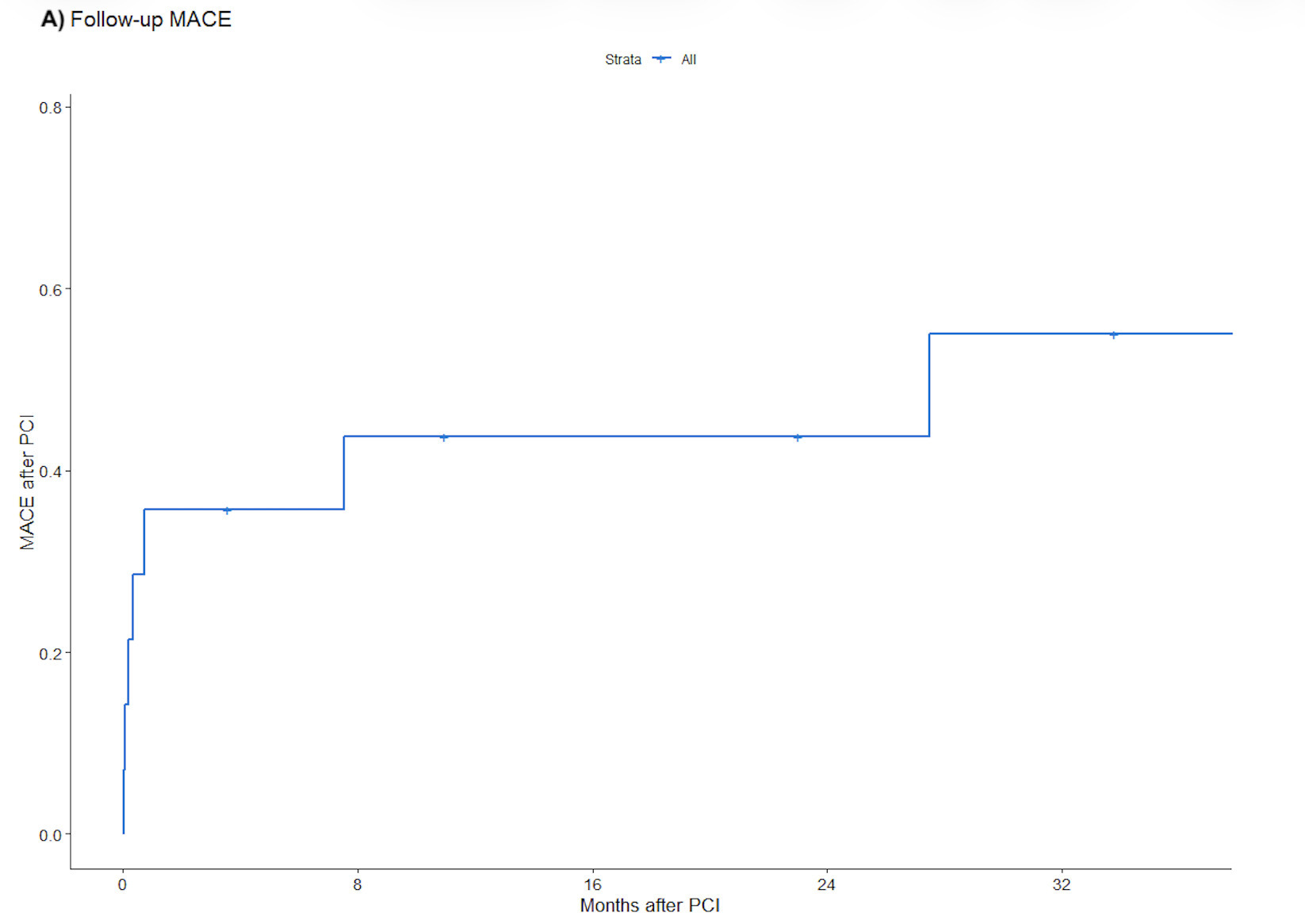
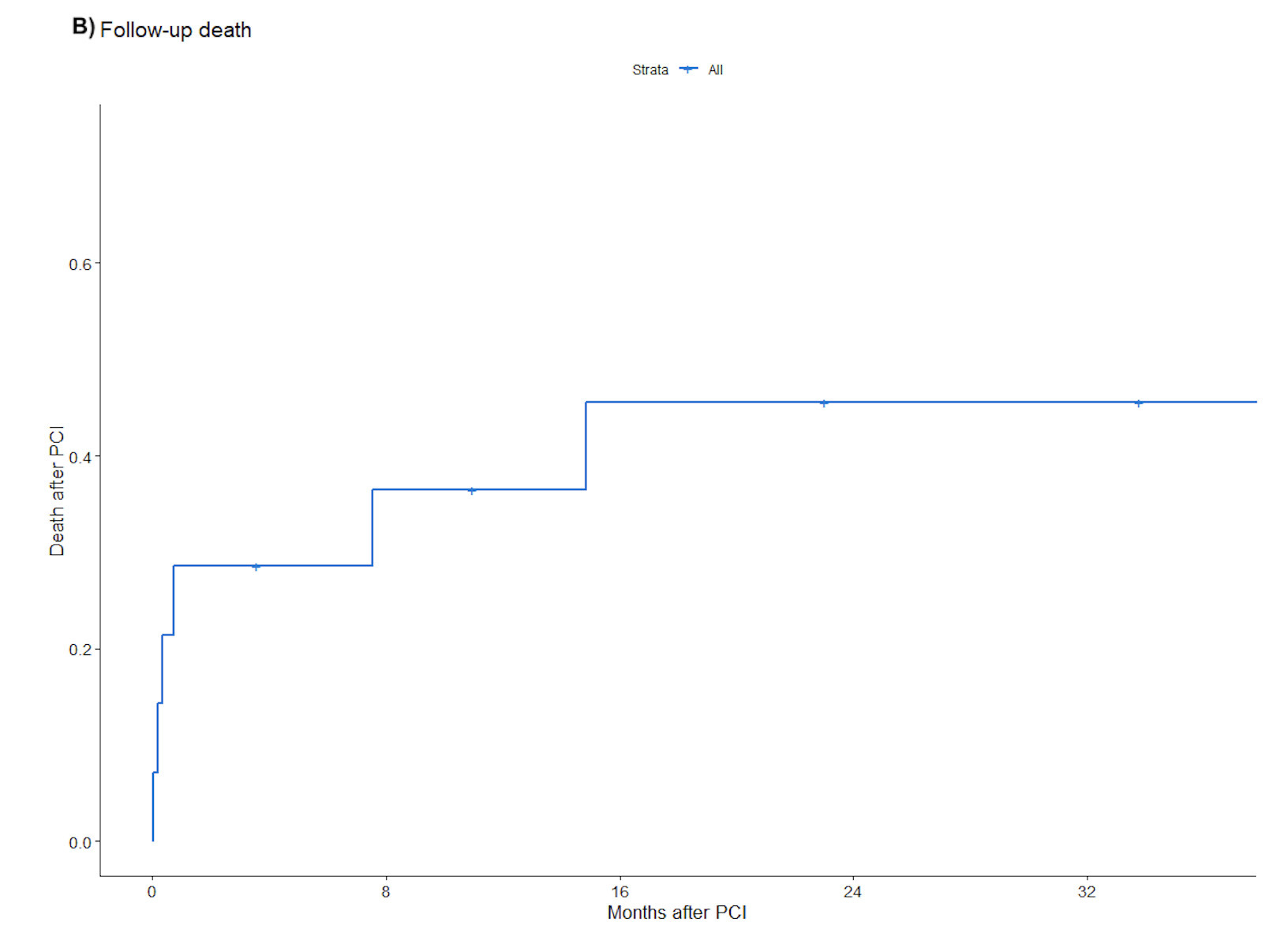
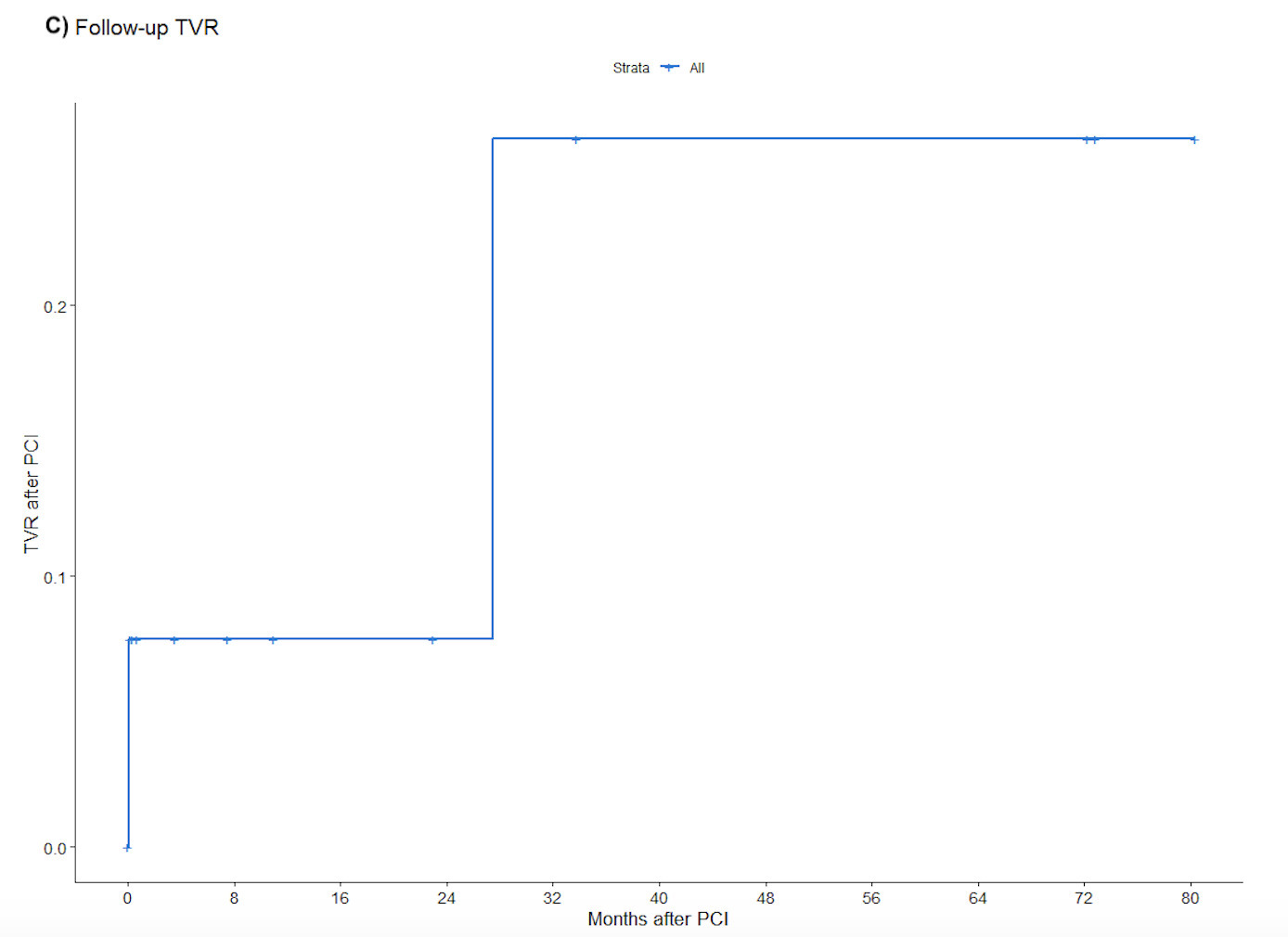
Postoperative complications are summarized in Table 6; these included death (33.3%), arrhythmia (33.3%), hemodynamic instability (26.7%), cardiogenic shock (13.3%), cardiac arrest (13.3%), bleeding (13.3%), acute renal failure (6.7%), prolonged ventilation (6.7%), wound infection (6.7%), ventricular rupture (6.7%), coronary dissection (6.7%), and cerebrovascular event (6.7%). Stroke, MI, tamponade, perforation, and hematoma were not observed during follow-up. During a median follow-up of 24 months, 53.3% of patients experienced MACE (Figure 1A), 46.7% died (Figure 1B), and 26.7% required target vessel revascularization (Figure 1C). Individual clinical and procedural characteristics of the patients in the study are listed in Table 7.
Discussion
The major findings of our study are that PCI-related emergency cardiac surgery (a) occurred in 0.08% of cases, with consistently low incidence throughout the study period; (b) was most commonly due to coronary dissection, aortocoronary dissection, or coronary perforation; (c) occurred in patients with complex angiographic features, including calcification, bifurcation lesions, and long lesion length; and (d) was associated with high rates of in-hospital and long-term MACE and mortality, including a 33.3% in-hospital mortality and a 46.7% 2-year mortality. Although the absolute incidence of these events is extremely low in contemporary practice, the clinical consequences remain severe, emphasizing the importance of rapid recognition and management of catastrophic PCI complications.
In our study, the incidence of emergency cardiac surgery post-PCI was 0.08%, which is similar to prior studies (0.07%-0.12%).1,12 The incidence has been decreasing in recent years, likely because of stricter safety protocols, including improved operator training and use of intravascular imaging for lesion assessment and strategy selection.13,14 Large contemporary registry analyses, including data from the National Cardiovascular Data Registry (NCDR) Cath-PCI Registry, have similarly demonstrated that the need for emergent or salvage CABG after PCI now occurs in approximately 0.1% of procedures, highlighting the rarity of this complication in modern interventional practice.15
Identifying factors associated with PCI-related emergency surgery is essential for developing effective prevention strategies. Patients in our cohort were typically older with a high burden of comorbidities, including diabetes mellitus, prior PCI, and MI. Patients frequently presented with acute coronary syndromes or cardiogenic shock. Similar risk profiles have been associated with poor outcomes in both PCI and cardiac surgery populations from other studies. Diabetes has repeatedly been linked to higher long-term mortality and major adverse events after PCI and CABG.16 In emergency CABG cohorts, particularly those operated after failed or complicated PCI, critical preoperative state, recent MI, low ejection fraction, and cardiogenic shock are major predictors of in-hospital and long-term mortality.17 Although direct data combining diabetes, prior MI, and emergency cardiac surgery after PCI are less abundantly reported, the overall evidence supports the notion that the high-risk profile we observed (older age, diabetes, prior PCI/MI, cardiogenic shock) is consistent with a group at increased risk of adverse post-PCI events. In addition, complex lesion morphology, such as heavy calcification, bifurcation anatomy, and long lesion length, is a recognized contributor to procedural difficulty and may increase the risk of complications during PCI.18
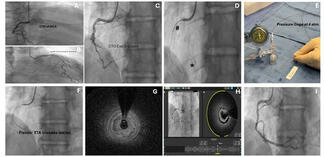
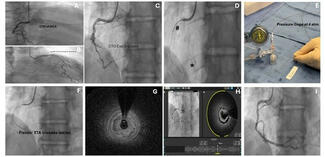
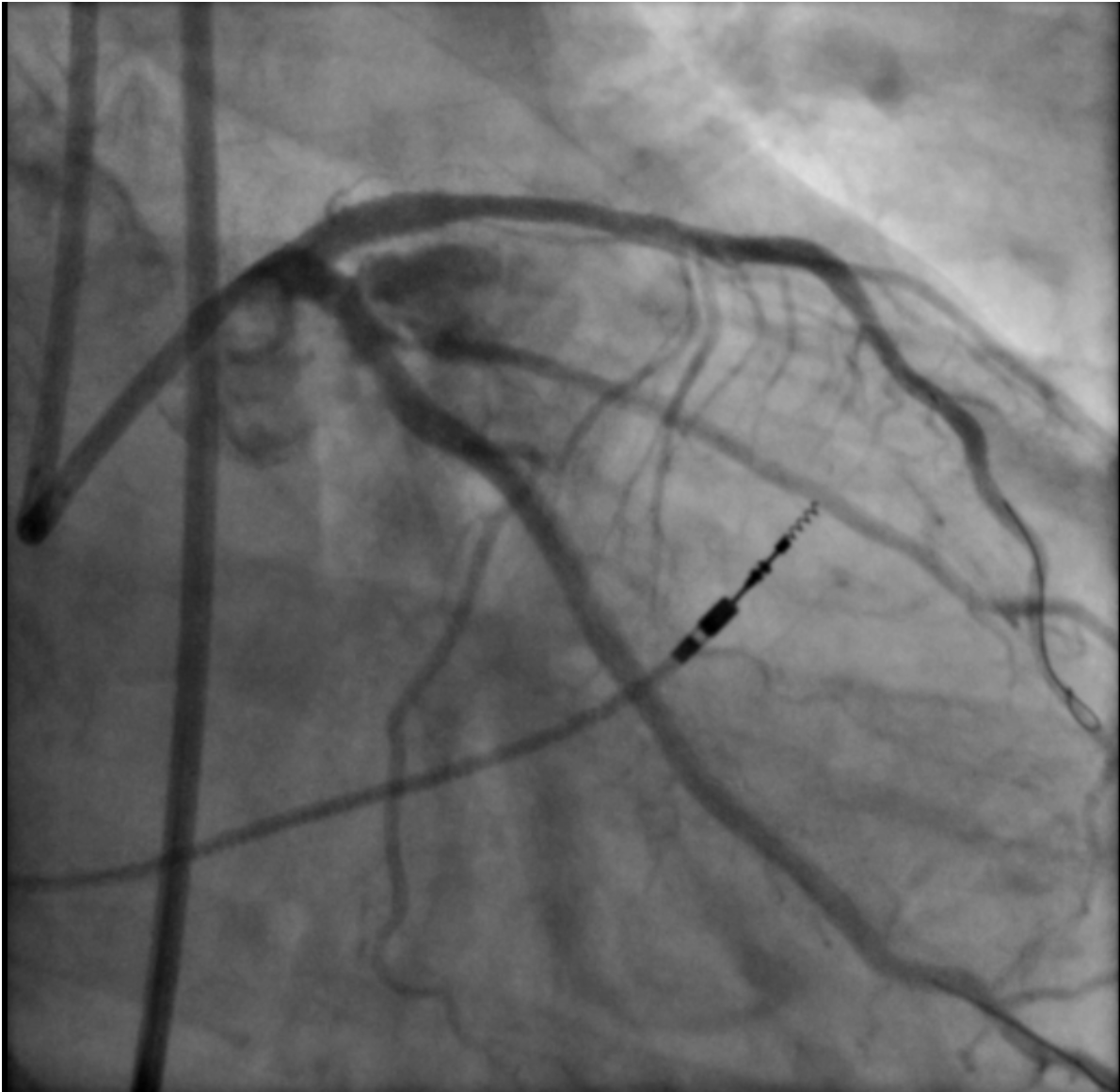
Coronary dissection (46.7%), aortocoronary dissection (26.7%), and coronary perforation (26.7%), including Ellis grade 3 perforations (26.7%), were the predominant indications for emergency cardiac surgery in our cohort. These severe complications can arise suddenly, leading to rapid hemodynamic deterioration through tamponade, myocardial ischemia, or cardiac arrest.19 Our observations parallel those of Darwazah et al, who identified failed angioplasty, dissection, and perforation as the principal drivers of surgical conversion.18 Importantly, while most coronary perforations can be stabilized with covered stents, prolonged balloon tamponade, or pericardiocentesis, a subset remains unresponsive to these strategies and requires immediate surgical intervention.20 An illustrative case from our cohort is shown in Figure 2. This highlights the need for interventional teams to maintain readiness for prompt surgical collaboration when faced with refractory dissection or perforation, as timely escalation can be lifesaving.
In-hospital mortality was 33.3% in our series, vs 21.0% reported in Thielman et al.21 Despite advances in PCI technology, recent studies have not shown substantial improvements in either the short- or long-term prognosis in this high-risk population.22 Our study adds to this field by presenting contemporary, multicenter data on the incidence and outcomes of PCI-related surgical rescue, demonstrating that although the absolute incidence is now very low, the associated mortality remains strikingly high. Furthermore, our findings highlight the ongoing importance of rapid recognition and escalation when refractory dissection or perforation occurs, reinforcing the relevance of surgical backup even in high-volume PCI centers. These results also have potential implications for PCI performed at centers without on-site cardiac surgery or in ambulatory surgical centers, as rare catastrophic complications may still require immediate surgical management.23
Limitations
The present study was observational and retrospective with all inherent limitations. Additionally, the procedures were conducted at a center with experienced PCI operators, which may restrict the generalizability of the findings to less experienced centers. Additional limitations should also be acknowledged. Angiographic lesion characteristics were determined by visual estimation by the operators and were not adjudicated by a central angiographic core laboratory. Detailed procedural variables such as the systematic use of intravascular imaging were not consistently captured within the registry dataset. Finally, the registry included only patients who ultimately underwent emergency cardiac surgery and therefore could not capture patients who experienced catastrophic PCI complications but did not undergo surgical intervention.
Affiliations and Disclosures
Harmanpreet Kaur, BS1,2 ; Michaella Alexandrou, MD1; Ozgur Selim Ser, MD1; Deniz Mutlu, MD1; Dimitrios Strepkos, MD1;
From the 1Minneapolis Heart Institute and Minneapolis Heart Institute Foundation, Abbott Northwestern Hospital, Minneapolis, Minnesota; 2Johns Hopkins University, Baltimore, Maryland; 3University of Washington, Seattle, Washington.
Acknowledgments: The authors are grateful for the philanthropic support of generous anonymous donors (2) and the philanthropic support of Dr. Mary Ann and Donald A Sens; Mrs Diane and Dr Cline Hickok; Mrs Wilma and Mr Dale Johnson; the Mrs Charlotte and Mr Jerry Golinvaux Family Fund; the Roehl Family Foundation; the Joseph Durda Foundation; Ms Marilyn and Mr William Ryerse; and Mr Greg and Mrs Rhoda Olsen. The generous gifts of these donors to the Minneapolis Heart Institute Foundation’s Science Center for Coronary Artery Disease (CCAD) helped support this research project.
Disclosures: Dr Sandoval is a consultant for and serves on the advisory board of Abbott; is a consultant for, serves on the advisory board of, and is a speaker for Roche Diagnostics; is a consultant for, serves on the advisory board of, and is a speaker for Philips; serves on the advisory board of Zoll; is a consultant for and serves on the advisory board of GE Healthcare; is a consultant for CathWorks; is a speaker for HeartFlow, Inc.; is a speaker for and has received a research grant from Cleerly Health; is an associate editor for JACC Advances; and he and others hold patent 20210401347. Dr Brilakis receives consulting/speaker honoraria from Abbott Vascular, the American Heart Association (associate editor, Circulation), Boston Scientific, Cardiovascular Innovations Foundation (Board of Directors), Cordis, Elsevier, GE Healthcare, IMDS, Medtronic, SIS Medical, Teleflex, and Terumo; research support from Boston Scientific, GE Healthcare; is the owner of Hippocrates LLC; and is a shareholder in Cleerly Health, LifeLens Technologies, Inc., MHI Ventures, Stallion Medical, and TrueVue, Inc. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Address for correspondence: Emmanouil S. Brilakis, MD, PhD, Minneapolis Heart Institute, 920 E 28th Street #300, Minneapolis, MN 55407, USA. Email: esbrilakis@gmail.
References
1. Kwok CS, Sirker A, Nolan J, Zaman A, et al. A national evaluation of emergency cardiac surgery after percutaneous coronary intervention and postsurgical patient outcomes. Am J Cardiol. 2020;130:24-29. doi:10.1016/j.amjcard.2020.05.041
2. Li K, Kalwani NM, Heidenreich PA, Fearon WF. Elective Percutaneous coronary intervention in ambulatory surgery centers. JACC Cardiovasc Interv. 2021;14(3):292-300. doi:10.1016/j.jcin.2020.10.015
3. Harris PA, Taylor R, Minor BL, et al; REDCap Consortium. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/j.jbi.2019.103208
4. Yang JH, Lee JM, Park TK, et al. The proximal optimization technique improves clinical outcomes when treated without kissing ballooning in patients with a bifurcation lesion. Korean Circ J. 2019;49(6):485-494. doi:10.4070/kcj.2018.0352
5. Lunardi M, Louvard Y, Lefèvre T, et al. Definitions and standardized endpoints for treatment of coronary bifurcations. EuroIntervention. 2023;19(10):e807-e831. doi:10.4244/EIJ-E-22-00018
6. Thygesen K, Alpert JS, Jaffe AS, et al; Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth universal definition of myocardial infarction (2018). Circulation. 2018;138(20):e618-e651. doi:10.1161/CIR.0000000000000617
7. Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123(23):2736-2747. doi:10.1161/CIRCULATIONAHA.110.009449
8. Jentzer JC, Burstein B, Van Diepen S, et al. Defining shock and preshock for mortality risk stratification in cardiac intensive care unit patients. Circ Heart Fail. 2021;14(1):e007678. doi:10.1161/CIRCHEARTFAILURE.120.007678
9. Brilakis ES. Manual of Coronary Chronic Total Occlusion Interventions: A Step-by-Step Approach. Elsevier/Academic Press; 2014.
10. Moscucci M. Grossman & Baim’s Cardiac Catheterization, Angiography, and Intervention. 9th ed. Wolters Kluwer; 2020.
11. Bindroo S, Quintanilla Rodriguez BS, Challa HJ. Renal Failure (Archived). In: StatPearls. StatPearls Publishing; 2026. Accessed April 9, 2026. http://www.ncbi.nlm.nih.gov/books/NBK519012/
12. Mutlu D, Rempakos A, Alexandrou M, et al. Emergency coronary artery bypass surgery after chronic total occlusion percutaneous coronary intervention: insights from the PROGRESS-CTO registry. Int J Cardiol. 2024;405:131931. doi:10.1016/j.ijcard.2024.131931
13. Doll JA, Kataruka A, Manandhar P, et al. Failure to rescue after percutaneous coronary intervention: insights from the National Cardiovascular Data registry. Circ Cardiovasc Interv. 2024;17(8):e013670. doi:10.1161/CIRCINTERVENTIONS.123.013670
14. Pancholy SB, Patel GA, Patel NR, et al. Trends, outcomes, and predictive score for emergency coronary artery bypass graft surgery after elective percutaneous coronary intervention (from a nationwide dataset). Am J Cardiol. 2021;144:46-51. doi:10.1016/j.amjcard.2020.12.060
15. Peterson ED, Dai D, DeLong ER, et al; NCDR Registry Participants. Contemporary mortality risk prediction for percutaneous coronary intervention: results from 588,398 procedures in the National Cardiovascular Data Registry. J Am Coll Cardiol. 2010;55(18):1923-1932. doi:10.1016/j.jacc.2010.02.005
16. Verevkin A, von Aspern K, Leontyev S, Lehmann S, Borger MA, Davierwala PM. Early and long-term outcomes in patients undergoing cardiac surgery following iatrogenic injuries during percutaneous coronary intervention. J Am Heart Assoc. 2019;8(1):e010940. doi:10.1161/JAHA.118.010940
17. Wilson SR, Vakili BA, Sherman W, Sanborn TA, Brown DL. Effect of diabetes on long-term mortality following contemporary percutaneous coronary intervention: analysis of 4,284 cases. Diabetes Care. 2004;27(5):1137-1142. doi:10.2337/diacare.27.5.1137
18. Darwazah AK, Islim I, Hanbali B, Shama RA, Aloul J. Emergency coronary artery bypass surgery after failed percutaneous coronary intervention. J Cardiovasc Surg (Torino). 2009;50(6):795-800.
19. Shimony A, Joseph L, Mottillo S, Eisenberg MJ. Coronary artery perforation during percutaneous coronary intervention: a systematic review and meta-analysis. Can J Cardiol. 2011;27(6):843-850. doi:10.1016/j.cjca.2011.04.014
20. Ford TJ, Adamson C, Morrow AJ, et al. Coronary artery perforations: Glasgow Natural History Study of Covered Stent Coronary Interventions (GNOCCI) study. J Am Heart Assoc. 2022;11(19):e024492. doi:10.1161/JAHA.121.024492
21. Thielmann M, Wendt D, Slottosch I, et al. Coronary artery bypass graft surgery in patients with acute coronary syndromes after primary percutaneous coronary intervention: a current report from the North-Rhine Westphalia Surgical Myocardial Infarction registry. J Am Heart Assoc. 2021;10(18):e021182. doi:10.1161/JAHA.121.021182
22. Noaman S, Vogrin S, Dinh D, et al; VCOR Investigators. Percutaneous coronary intervention volume and cardiac surgery availability effect on acute coronary syndrome-related cardiogenic shock. JACC Cardiovasc Interv. 2022;15(8):876-886. doi:10.1016/j.jcin.2022.01.283
23. Li K, Kalwani NM, Heidenreich PA, Fearon WF. Elective percutaneous coronary intervention in ambulatory surgery centers. JACC Cardiovasc Interv. 2021;14(3):292-300. doi:10.1016/j.jcin.2020.10.015