Peer Reviewed
Bilateral Surgical Bypass With Prosthetic Conduit Incorporating Deep Venous Arterialization for Limb Preservation
Abstract
This case report describes bilateral surgical bypass in a patient with “no-option” chronic limb-threatening ischemia. Both bypasses incorporated deep venous arterialization for limb preservation. Details of the case are illustrated along with some of the nuances involved in surgical bypass with prosthetic conduit incorporating deep venous arterialization.
J CRIT LIMB ISCHEM 2026:6(2):E46-E50. doi: 10.25270/jcli/CLIG-2500004
Key words: chronic limb-threatening ischemia, deep venous arterialization, bypass, prosthetic graft
Introduction
Chronic limb-threatening ischemia (CLTI) represents approximately 10% to 15% of the growing population of patients with peripheral arterial disease. Approximately 20% of patients with CLTI are considered “no option”, lacking a suitable target for traditional revascularization by endovascular or open surgical techniques.1 These no-option patients are at increased risk for nonhealing of the lower extremity with amputation rates as high as 40% at 1 year.2 Deep venous arterialization (DVA) offers revascularization in an attempt to obtain limb preservation in this challenging group of patients. DVA delivers oxygenated blood to the capillary bed in a retrograde manner by way of the venous system and has been described with surgical bypass as well as percutaneous endovascular techniques. This case report describes bilateral surgical bypass incorporating DVA for healing and limb preservation.
Case Report
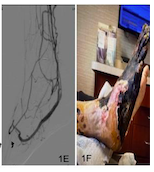
A 70-year-old African American woman presented with CLTI of the lower extremities. The primary complaint involved left-foot rest pain with progressive tissue loss at the base of the fifth metatarsal head and gangrenous changes of the forefoot. The patient had a medical history of congestive heart failure, hypertension, hyperlipidemia, pancreatitis, atrial fibrillation, and poorly controlled type 2 diabetes mellitus. Prior attempts at revascularization at an outside facility included endovascular intervention (angioplasty of the superficial femoral, popliteal, and anterior tibial arteries) as well as 2 failed surgical bypass procedures (femoral to below-knee popliteal with polytetrafluoroethylene [PTFE] and femoral to posterior tibial artery with saphenous vein). Both bypasses had occluded within 12 months. Physical exam was notable for an ulceration of the left forefoot with gangrene of the first and fifth digits. There were no palpable pedal pulses. Noninvasive studies in the vascular laboratory revealed an ankle-brachial index (ABI) of 0.27 on the left and 0.53 on the right. The toe-brachial index (TBI) on the left was unmeasurable, with a TBI on the right of 0.41. The patient was admitted and scheduled for lower extremity arteriography to determine options for possible revascularization.
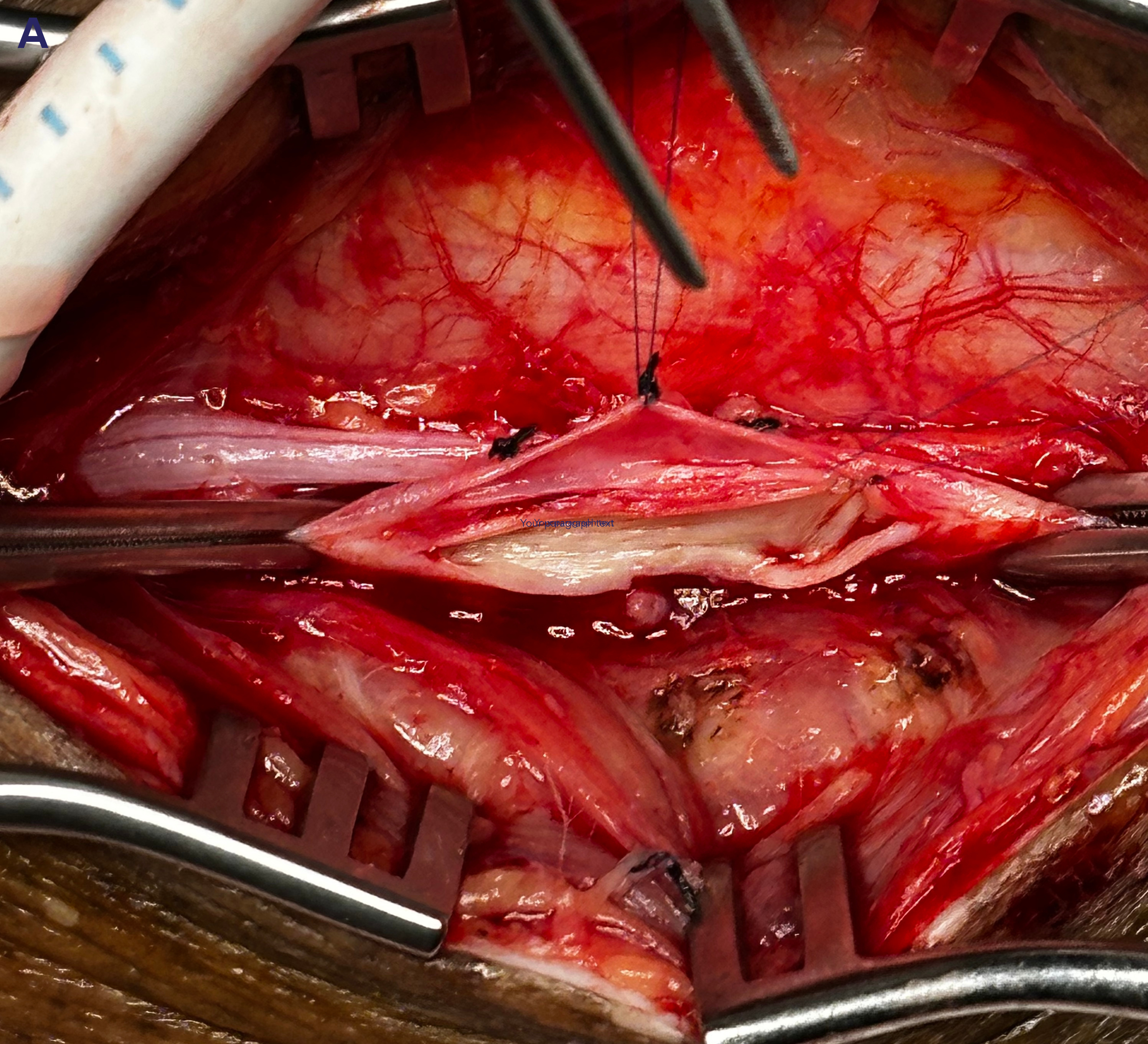
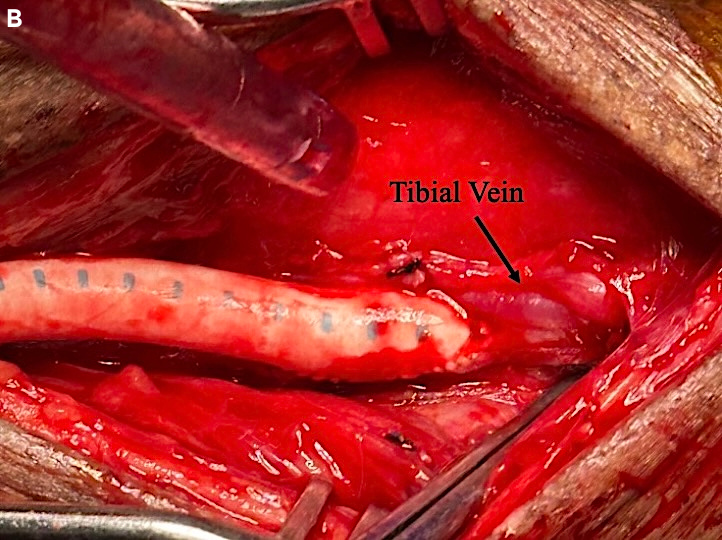
Arteriography showed a chronically occluded left superficial femoral artery with reconstitution of a segment of the above-knee popliteal artery, stenosis of the P2 segment of the distal popliteal artery, stenosis of the tibioperoneal trunk, and occlusion of the tibial arteries with reconstitution of a small, isolated posterior tibial segment at the ankle. A bypass was performed from the left external iliac artery to the posterior tibial vascular bundle incorporating DVA of the posterior tibial vein. Due to prior procedures and a hostile left groin, a limited retroperitoneal incision was made in the left lower quadrant to expose the external iliac artery, which was found to have a strong pulse. A second incision was made in the medial aspect of the lower left leg just superior to the ankle to expose the posterior tibial vascular bundle. On examination, the posterior tibial artery was a diseased, calcified isolated segment not suitable for arterial bypass. The bypass conduit was a 6-mm heparin-bonded expanded PTFE (HePTFE) graft (Propaten, W.L. Gore) tunneled between the proximal and distal incisions. The patient received intravenous heparin, and the proximal anastomosis was performed in an end-to-side manner using 6-0 Prolene suture (Ethicon) in the parachute technique. The distal anastomosis was performed using a common ostium technique with 7-0 Prolene suture and the parachute technique. The arterial segment was included to facilitate suturing the prosthetic conduit to the tibial vein by essentially stenting open the anastomosis with the arterial wall and allowing increased luminal diameter of the anastomosis to the tibial vein at this location (Figure 1). Vein valve disruption or lysis was performed prior to completion of the anastomosis using Cooley coronary dilators to physically disrupt the vein valves intraluminally. Valve lysis of 4 tibial vein valves was performed between the anastomosis and the plantar surface of the foot. The anastomosis was closed, and anterograde flow in the vein segment was strong with a palpable pulse and a triphasic Doppler signal in the distal vein. The wounds were irrigated and closed, and the patient returned to recovery in stable condition. Postoperatively, the patient was maintained on aspirin and 24 hours of intravenous heparin prior to conversion to apixaban (5 mg) to maintain graft patency. Podiatric wound care was continued with 72 hours of intravenous antibiotics prior to discharge.
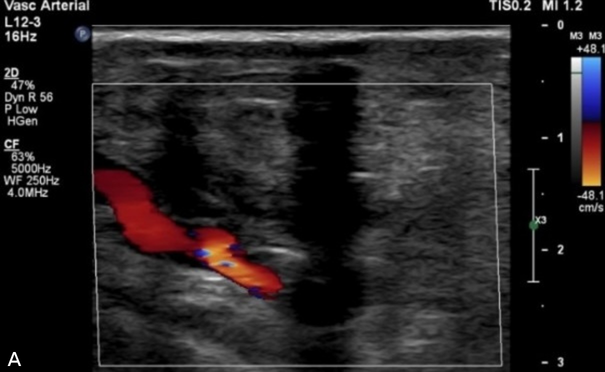
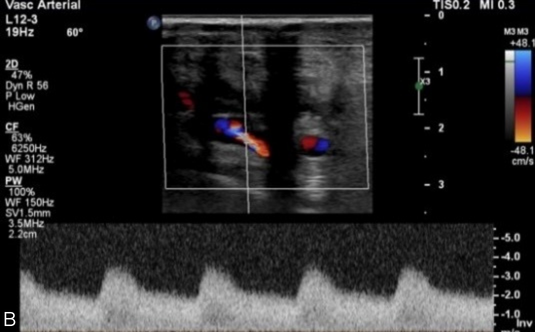
Two weeks after discharge, the patient was admitted to the medical service for an acute-on-chronic heart failure exacerbation. Computed tomography angiography of the abdominal aorta and bilateral runoff to rule out graft infection found the graft to be patent without fluid accumulation or other radiographic signs of infection. After discharge, the patient followed our standard surveillance protocol with outpatient evaluation at 3, 6, and 12 months. Vascular lab studies at 3 months showed a left ABI of 1.05 and TBI of 0.66 (initial TBI was 0.12). At 6 months, the ABI was not obtained due to concern regarding compression of the distal anastomosis, and the TBI was 0.54 with triphasic digital waveforms. At the patient’s yearly follow-up visit, the TBI remained 0.5. Duplex ultrasounds obtained at these time intervals confirmed graft patency (Figure 2). The left foot wounds healed in 6 months with podiatric wound care.
During follow-up, a right calcaneal lesion extended to subcutaneous tissue with exposure of a portion of the Achilles tendon despite wound care. The patient had undergone failed bypass on the right in the past, from the femoral artery to the proximal anterior tibial artery, using a PTFE conduit without any anastomotic adjuncts. A right lower extremity arteriogram was obtained, and the patient’s right lower extremity arterial anatomy was deemed nonamenable to conventional revascularization. Realizing DVA is not the ideal option for heel ulceration, there was no other alternative for the right foot. Because lower extremity bypass with DVA had maintained the left limb, it was decided to attempt a similar intervention on the right side. A right-leg bypass was performed using a similar technique as described above but using the femoral artery as inflow and the anterior tibial vascular bundle as outflow. The patient was discharged with bilateral multiphasic pedal Doppler signals for follow-up with vascular surgery and podiatric in one of our wound centers. The patient returned to home out of the area but has a patent bypass and limb preservation at 6 months.
Discussion
Several no-option patients are presenting for revascularization to achieve healing and amputation prevention, especially in the context of a limb preservation program. These patients may have undergone prior endovascular or open surgical procedures and continue to face a lack of healing and limb loss. DVA is a technique for consideration in such patients, meant to deliver oxygenated blood in a retrograde manner through the venous system to the tissue by way of the capillary bed. This concept was described by Alexis Carrel during his work with anastomotic technique in a canine femoral model. At that time, it was noted that although venous valves prevent such reverse circulatory patterns, after several hours flow did pass through the capillaries and into the arterial tree, filling in a retrograde manner, with dark blood demonstrating such flow reversal. Clinical utility was reported in the surgical literature with a report in 2010 describing 18 patients having an in situ vein bypass to perfuse the deep venous arch of the foot. The authors reported 55% limb salvage in this challenging patient group.3 In 2011, Mutirangura reported on 26 patients with unreconstructable distal arteries undergoing vein bypass to the paramalleolar deep veins with destruction of distal valve competency. This experience resulted in 49% patency and 75% limb salvage at 2 years.4 More recently, endovascular DVA has proven to be relatively safe and effective.5 Off-the-shelf devices have been used for the endovascular approach, as well as a standardized commercial product (the LimFlow system).
This case report describes our early experience with surgical bypass incorporating DVA. The technique built upon our previously reported anastomotic adjunctive techniques.6-8 The patient required bilateral procedures to avoid major bilateral amputation after failure of other surgical and endovascular attempts at revascularization. Due to prior bypass attempts, the patient did not have saphenous vein available as the DVA bypass conduit. The lack of a suitable target for arterial bypass also precluded a fistulous construction, leading to the decision to incorporate DVA. A direct tibial venous anastomosis was not chosen to avoid a prosthetic conduit sutured directly to a small tibial vein. The need for such a prosthetic bypass is a situation we face in a significant number of our bypass cohort due to prior usage for limb bypass, coronary artery bypass grafting, varicose vein procedures, and prior phlebitis.9 The use of HePTFE with anastomotic adjuncts has increased prosthetic graft utilization and outcomes.9,10 In this case, the anastomotic configuration optimized technique with flow directed into the distal tibial venous circulation (Figure 1). There was no anticipation of flow into the corresponding tibial artery. Healing related directly to DVA is somewhat speculative; however, we believe there is not a component of arterial revascularization in this case. A sustained improvement in TBI did occur with duplex imaging to demonstrate graft patency and with flow into the tibial venous circulation. The final arbiter is healing, and this did occur.
Patients should be counseled that edema can occur initially, and the patient must be willing to embark upon a postoperative course that requires time and aggressive wound care. Definitive wound closure is often delayed, allowing for a decrease in such edema and optimization of retrograde perfusion with any angiogenesis that may occur. In regards to postoperative medical therapy, initial experience with the distal vein patch technique indicated that anticoagulation resulted in enhanced graft patency, although this did not reach statistical significance.11 Perioperative intravenous heparin with transition to oral anticoagulation had been standard; however, because of the rapid onset of the direct oral anticoagulant (DOAC) medications and a reduction in the risk of hematoma and incisional bleeding with intravenous heparin, this practice has been reconsidered. Currently, patients undergoing surgical DVA are administered an antiplatelet agent (often clopidogrel) and a DOAC (exact medication based on patient insurance and wishes). Dual antiplatelet therapy is used for a history of bleeding with anticoagulation or inability to obtain a DOAC.
Follow-up of these patients includes duplex ultrasound evaluation of the conduit as well as distal venous architecture. We have not routinely performed follow-up arteriography nor secondary intervention, as is often the case with the endovascular technique.12 Follow-up arteriography was not considered as important as for endovascular DVA because surgical DVA has a more focused and distal venous target. However, such routine follow-up arteriography with secondary intervention is being considered after surgical DVA, as well as a more aggressive approach to secondary venous intervention to enhance outcomes.
Conclusion
This case report involves bilateral revascularization by surgical bypass incorporating DVA for limb preservation. Surgical DVA will continue to play a role in the armamentarium for limb preservation; however, as standardized endovascular devices become refined and more available, the endovascular approach may take precedence. Regardless, DVA should be considered for patients considered “no option” to obtain limb preservation. This procedure requires effort, close follow-up, and unique wound care to obtain optimal outcomes. This requires commitment both by the surgeon and patient.
Affiliations and Disclosures
Grant Killian, MS, is from the University of Virginia School of Medicine, Charlottesville, Virginia; Richard F. Neville, MD, FACS, DFSVS, is from Inova Schar Heart and Vascular, Falls Church, Virginia.
Dr Neville serves on the Scientific Advisory Board of W. L. Gore. Mr Killian reports no financial relationships or conflicts of interest regarding the content herein.
Manuscript accepted April 29, 2026.
Address for Correspondence: Richard F. Neville, MD, FACS, DFSVS, Inova Schar Heart and Vascular, 3300 Gallows Rd, Falls Church, VA 22042. Email: Richard.neville@inova.org
References
1. Shishehbor MH, Powell RJ, Montero-Baker MF, et al; PROMISE II Investigators. Transcatheter arterialization of deep veins in chronic limb-threatening ischemia. N Eng J Med. 2023;388(13):1171-1180. doi:10.1056/NEJMoa2212754
2. Varu VN, Hogg ME, Kibbe MR. Critical limb ischemia. J Vasc Surg. 2010;51(1):230-241. doi:10.1016/j.jvs.2009.08.073
3. Busato CR, Utrabo CAL, Gomes RZ, et al. J The great saphenous vein in situ for the arterialization of the venous arch of the foot. J Vasc Bras. 2010;9(3):119-123.
4. Mutirangura P, Ruangsetakit C, Wongwanit C, Sermsathanasawadi N, Chinsakchai K. Pedal bypass with deep venous arterialization: the therapeutic option in critical limb ischemia and unreconstructable distal arteries. Vascular. 2011;19(6):313-319. doi:10.1258/vasc.2010.oa0278
5. Saini N, Marrone L, Desai S, Herman K, Rundback JH. Comparison of outcomes in deep venous arterialization in multiple practice settings. J Vasc Surg. 2024;80(5):1507-1514. doi:10.1016/j.jvs.2024.05.051
6. Neville RF, Lidsky M, Capone A, Babrowicz J, Rahbar R, Sidawy AN. An expanded series of distal bypass using the distal vein patch technique to improve prosthetic graft performance in critical limb ischemia. Eur J Vasc Endovasc Surg. 2012; 44(2):177-182. doi:10.1016/j.ejvs.2012.04.014
7. Neville RF, Dy B, Singh N, DeZee KJ. Distal vein patch with an arteriovenous fistula: a viable option for the patient without autogenous conduit and severe distal occlusive disease. J Vasc Surg. 2009; 50(1):83-88. doi:10.1016/j.jvs.2008.12.052
8. Lauria AL, Propper BW, Neville RF. Surgical deep vein arterialization; adding to the armamentarium of complex limb salvage. Ann Vasc Surg. 2022;87:198-204. doi:10.1016/j.avsg.2022.03.036
9. Gerling KA, Gabel JA, Neville RF. The current pattern of surgical bypass in the context of a limb preservation practice. J Crit Limb Ischem. 2024;4(1):E29-33. doi:10.25270/jcli/CLIG23-00017
10. Dexter, DJ, O’Neill F, Neville RF. Systematic literature review comparing clinical utility of heparin-bonded expanded polytetrafluoroethylene graft to autologous saphenous vein graft for management of below-knee peripheral arterial disease. J Vasc Surg. 2024;80(6):1863-1870.e6. doi:10.1016/j.jvs.2024.07.023
11. Neville RF, Capone A, BS, Amdur R, Lidsky M, Babrowicz J, Sidawy AN. A comparison of tibial artery bypass performed with heparin bonded expanded polytetrafluoroethylene and greater saphenous vein to treat critical limb ischemia. J Vasc Surg. 2012;56(4):1008-1014. doi:10.1016/j.jvs.2012.03.020
12. Zaman NS, Shackles C, Moriarty KT, Herman K, Rundback JH. Patterns of failure in deep venous arterialization and implications for management. J Crit Limb Ischem. 2022;2(3):E58-E63. doi:10.25270/jcli/CLIG22-00008