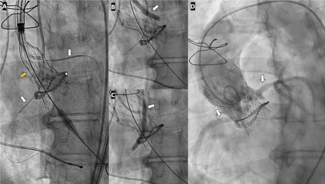
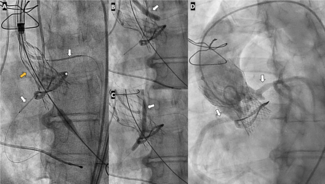
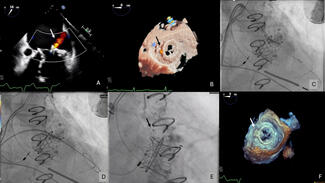
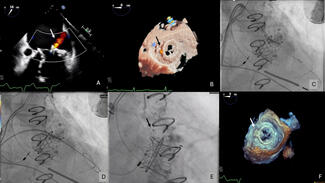
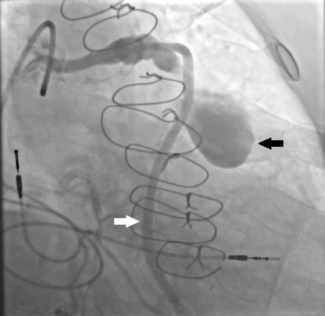
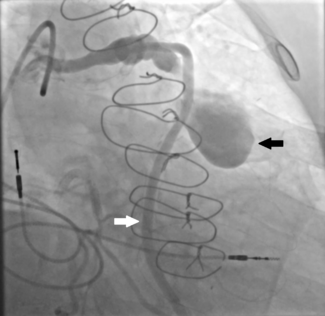
Protect the H2H Trial: Randomized Comparison of Emboliner Embolic Protection Catheter vs Sentinel Cerebral Protection System During Transcatheter Aortic Valve Replacement
In this interview with JIC, Dr Adam Greenbaum of Emory University shares insights on his late-breaking study, "Protect the H2H Trial: Randomized Comparison of Emboliner Embolic Protection Catheter vs Sentinel Cerebral Protection System During Transcatheter Aortic Valve Replacement," presented at the 2026 American College of Cardiology (ACC) Conference.
Transcript:
Hi, my name is Adam Greenbaum. I’m one of the co-directors of the Structural Heart and Valve Center at Emory University in Atlanta, Georgia, and I’m here to discuss the Protect the Head-to-Head (H2H) study, recently released at the American College of Cardiology (ACC) Conference in New Orleans.
Results showed similar 30-day outcomes for Emboliner and Sentinel, higher technical success for Emboliner, but also longer procedure times for Emboliner. How should clinicians weigh those tradeoffs in practice—and what will matter most for broader adoption: reproducibility outside experienced centers, cost-effectiveness, or clear patient benefit?
First, you're talking about operators that were very experienced with Sentinel, and they were actually using Emboliner for the first time—centers were allowed up to 2 roll-ins but, for the most part, this was a brand new device. So, of course they're going to take a little bit more time placing the device. You're also in a trial where there was a core lab that wanted to look at, for instance, whether all the vessels were covered at the time of valve deployment, so there were additional fluoroscopic views that people were asked to obtain. So, when you look at a continuous variable like fluoroscopy time, sure, they're going to be statistically significant, but there really wasn't much clinically significant difference in terms of contrast volume, fluoroscopy time. Overall procedural lengths were the same, the device dwell times were about the same. So, I wouldn't necessarily think that's going to factor in too much for a structuralist, as there's always a learning curve to a new device.
But what was interesting was the technical success—meaning, were the intended vessels covered at the time of valve deployment? It was much higher with the Emboliner because the device just drapes across the arch, you don't actually have to get the baskets in the great vessels. Plus, it comes from the legs, you don't have to deal with tortuosity in the arms. So technical success was much higher.
With Emboliner capturing substantially more and smaller debris particles, yet having similar results to the Sentinel, is there a threshold of debris burden that you believe is clinically relevant, or are we still lacking the tools to make that determination?
That's a tough one. Clearly it captured more debris, and that's not surprising, because there's also a basket in the descending aorta. So not only is it protecting the great vessels, but it actually is preventing downstream emboli from what might go to the gut or the kidneys, for instance. I think debris capture is going to be greater with a device that captures all particles.
Now, why didn’t that necessarily translate into a lower stroke rate? The trial was designed as a non-inferiority trial, right? So, it was designed to just show a non-inferiority. And to me, the issue here is that historically we've used this 30-day endpoint; a 30-day composite endpoint was stroke, death, and acute kidney injury (AKI) because that was the endpoint that was used, for instance, when the Sentinel trial was first done.
To me, the problem with that is that although endpoints accrue so that your composite endpoint is higher so that it's easier to make comparisons with less patients in the study, you introduce noise into the system because you can't ask a device that's placed during the time of a procedure to protect against a stroke at day 7 or day 17. There was 1 patient in the Emboliner arm that had a stroke at day 5 who has atrial fibrillation, wasn't on anticoagulation. It was a weird thing that happened. One of the patients in the Emboliner arm actually came back with an MI at day 17, had fibrolytic therapy, and a hemorrhagic stroke. So, to me, that adds noise to the system. But if you look at what happened on day 1, there was 1 stroke in the Emboliner arm and 4 in the Sentinel arm. So not at the secondary endpoint, but when you start to look early on, it's intriguing to me that there may be a signal there that these devices, if they're easy to place and technically successful, that you actually can reduce stroke.
You’ve said that with the right endpoint, these devices would show a significant benefit in terms of strokes. If you were designing the next trial, what would the right stroke endpoint be? And what else would you change in the design of the trial?
First of all, my primary endpoint would be 72 hours max; I'd have a secondary endpoint at 24. You know, there were no MRIs in this study, and that's controversial, right? Because if there's an MRI-positive stroke, but not a clinical endpoint, then a clinical stroke, then someone's going to say, “well, what did it really mean?” But I think those are better timepoints. And I think then increase the number of patients, and once you do that you might actually be able to show superiority or, in this case, a statistical difference between the two.
The Emboliner is designed to provide full-body protection, whereas Sentinel protects only selected vessels—Do you think future trials should shift focus beyond stroke to put more weight on systemic outcomes like renal or peripheral complications? And adding to that, what result do you think would have the most immediate clinical impact in terms of shifting the scale towards wide Emoliner use?
I think the kidney thing is fascinating because we see a lot of patients' creatinine go up after the procedure. We assume it's contrast; sometimes it goes down a few days later and sometimes it doesn't. It's a well-known phenomenon, at least with transfemoral diagnostic catheterization and PCI, that there is cholesterol emboli right to the kidneys during those procedures. Interestingly, when you look at what's captured in these baskets, there’s debris captured in almost 100% of the cases. It's thrombus, it's arterial wall, it's some valve tissue, right? So, to me, being able to protect the kidneys is interesting.
There was a lot of interest in cognitive decline, right? Maybe these MRI hits weren't leading to clinical strokes, but it was leading to cognitive decline and dementia down the road. Well, how do we know that embolite to the kidneys, while it may not cause acute renal failure, doesn't lead to renal failure sooner, albeit later in life, but sooner than it would have been had they not had X number of nephrons taken out by embolic debris? So, the AKI thing is interesting. Again, this trial was not powered to statistically look at any differences, but if you look at the acute Stage 3 AKI in the trial, it was 1 in the Emboliner, 3 in the Sentinel arm. So again, I just look at some of these numbers that the trial wasn't powered to make any definitive conclusions about, but I’m intrigued that there may be something there as well.
The transcript has been lightly edited for clarity.
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.