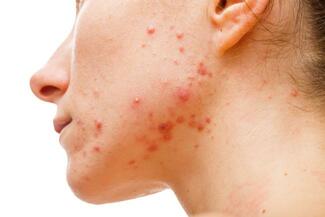
Ranbaxy Laboratories, Inc., a wholly-owned subsidiary of Ranbaxy Laboratories Limited, has announced the launch and sale of Absorica (isotretinoin) Capsules, for the treatment of severe, recalcitrant acne in patients 12 years of age and older.
Because isotretinoin has a high lipophilicity, oral absorption of the drug is enhanced when it is taken with a high-fat meal. However, the new Absorica is formulated with patented Lidose technology and can be taken without regard to meals. According to news reports on the release of Absorica, the fasted AUC0-t of the drug is approximately 83% greater than that of Accutane, although the products are bioequivalent under federal conditions. As a result of this equivalency, Absorica is not interchangeable and cannot be substituted with generic products of Accutane.
According to news reports, the approval of Absorica is based on a large, pivotal clinical trial that enrolled 925 patients.
"We are most pleased to make Absorica available as a valuable option for dermatologists and a subset of patients who suffer from severe recalcitrant nodular acne,” explains Dr. Ashish Anvekar, senior director of Ranbaxy Laboratories Inc. “Absorica will be the flagship brand of the Ranbaxy dermatology product portfolio in the US."