As the population ages, the desire for cosmetic procedures to restore a more youthful appearance will continue to rise. Multiple components contribute to volume loss in the aging face including extrinsic factors such as ultraviolet and oxidative damage as well as intrinsic factors such as bone and soft tissue remodeling, which are both hormonally and genetically regulated.1,2
The loss of volume in the mid-face results in flattening and furrowing of the central area of the mid-cheek. Additionally, displacement of tissues downward and medially cause the depth of the nasolabial fold to be exaggerated.3 Restoring this volume loss has become an integral part of the aesthetic treatment plans for the aging face.
Hyaluronic acid (HA) is a naturally occurring glycosaminoglycan that is a component of the extracellular matrix in connective tissue. HA is naturally non-immunogenic.3,4 HA has a high affinity for water, and thus it plays an integral role in maintaining dermal volume and compressibility.4 Unfortunately, in its natural state, HA has a half-life of only 1 to 2 days due to aqueous dilution and enzymatic degredation.4 In order for HA to be a viable filler, manufacturers had to develop methods to increase resistance to degradation.5
In 2013, the FDA approved a new HA filler specifically for the treatment of mid-face volume loss.6 The product, VYC-20L (Juvéderm Voluma XC, Allergan Inc.), consists of a 20-mg/mL mixture of low molecular weight (<1 mDa) and high molecular weight (>1 mDa) HA resulting in a more mobile network facilitating efficient cross-linking.3,7 Due to the efficient cross-linking, VYC-20L is a highly cohesive gel with greater hardness than other HA products resulting in increased longevity as well as greater lift capability.7
Complications with HA fillers are rare. One rare complication that has been reported with HA fillers is the development of blue papules and nodules in the area treated due to the Tyndall effect. The Tyndall effect occurs when HA fillers are inappropriately implanted into the superficial dermis or epidermis.8 The lesions appear blue because blue light waves have a higher frequency than red light waves and are more easily scattered.8 When light hits the surface of the skin with superficially placed HA particles, the particles cause the light waves to be reflected, and due to increased scatter, blue is the predominant color that emerges.9
Fortunately, the discoloration caused by the Tyndall effect can be treated. The most common treatment of this complication is the use of hyaluronidase, which is an off-label treatment for this condition.8 No cases of discoloration caused by the Tyndall effect occurring with injection of VYC-20L have been reported.
In this article, we present a case study of a patient developing a blue nodule under her right eye after being treated with VYC-20L. The complication was successfully treated with hyaluronidase.
Therapeutic Alternatives
Most HA fillers are known to resorb in 6 to 18 months, although the more superficial the placement of the material, the longer the duration of the discoloration.8 Due to the transient nature of HA fillers, treatment of the blue lesions caused by the Tyndall effect may not be necessary in cases where the lesions are small and imperceptible. However, VYC-20L has been shown in studies to retain its effectiveness for 2 years or longer; therefore, waiting for the product to resorb is not a viable option.3,7
Treatment of the discoloration with a QS Nd:YAG laser at 1,064 nm has been reported.9 In this report, discoloration resolved after 1 treatment with the VersaPulse (Lumenis) 1,064 QS Nd:YAG 3 mm spot, 5.5 J/cm2 and the laser side effects were not elicudated.9 The authors did not know the mechanism of action or the target chromophore, but they theorized that the laser increased HA turnover by some unknown mechanism.
There have been some reports of the discoloration from HA being treated by nicking the skin with a small-gauge needle (30 gauge) or surgical scalpel (#11 blade) and expressing the contents.10 This can be done immediately after injection and up to 12 months or more after placement.10 This procedure is usually done as a last resort after all other treatment options have failed.8
Case Study
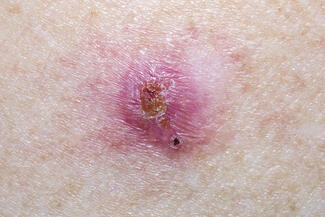
Our 61-year-old female patient presented for evaluation of a small blue papule of 3 months duration after injection of VYC-20L under her right eye (Figure 1). Shortly after the injection of VYC-20L and correction of the moderate mid-facial volume defect bilaterally, she noticed development of a blue discoloration in the lower eyelid, which was still present 3 months later. At this time, the lesions were felt to be due to superficially placed HA resulting in the Tyndall effect.
The patient desired to have the color changes treated. Therapeutic options were discussed with the patient and we recommended treatment with hyaluronidase. She was informed of the potential side effects of hyaluronidase and that despite the lack of FDA approval for the treatment of unwanted HA deposits, hyaluronidase has been used efficiently to treat these deposits caused by other HA fillers. After informed consent was obtained, a 150 unit/mL vile of hyaluronidase human injection (Hylenex recombinant, Halozyme Therapeutics) was diluted with 0.9% saline (1 mL). Approximately, 0.1 mL of the mixture was injected into the lesion. About 1 week later the patient received a second injection of 0.1 mL of hyaluronidase into the area. The procedure was well-tolerated by the patient.
Discussion
VYC-20L is the only FDA-approved filler for treatment of moderate-to-severe mid-face volume loss. The clinical study conducted to gain FDA approval showed that 85.6% of patients in the treatment group improved by 1 point or more on the Mid-Face Volume Deficit Scale (MFVDS) at 6 months, and there was a statistically significant difference (P<0.001) between the treatment and control groups.7 Additionally, duration of effectiveness calculated using Kaplan-Meier analysis based on evaluating investigators’ assessments of overall MFVDS showed that 73.9% of subjects were still responders at 1 year and 44.6% were responders at 2 years.7 The results were achieved with the initial treatment and a touch-up procedure 30 days after the initial procedure.
The study also showed that the VYC-20L was a safe product. The majority of the adverse events were treatment site reactions typical of HA injections including tenderness, swelling, firmness and lumps or bumps.7 The researchers reported that the majority of these effects lasted 2 weeks or less and resolved without treatment. Two patients had severe delayed-onset adverse events (1 was lumps in the treatment site that developed at 7 months and the other was cellulitis in the area after an injury that resulted in nodularity) that were treated with antibiotics, anti-inflammatories and hyaluronidase successfully without long-term sequella.7 Thus, VYC-20L is an effective treatment with a good safety profile.
These findings are corroborated by several other studies. In 1 study from Australia, 103 patients received VYC-20L to the mid-face. Of the patients enrolled, 96% were documented to be MFVDS responders at 8 weeks and 88.2% and 81.5% were still responders at weeks 52 and 78, respectively, after the initial treatment and 1 touch-up procedure at week 4.3. Patient satisfaction with the product was high with 70 of 72 patients that completed the study saying they would recommend the product.3
In this study, adverse events were transient and infrequent. The most common adverse events reported were injection site bruising and swelling.3 Several other studies from Europe with shorter follow-up times also showed high patient satisfaction and low occurrence of adverse events.5,11 None of the studies reported development of any skin discoloration from the Tyndall effect.
Despite being used off-label, several clinical trials and many case reports show the effectiveness of hyaluronidase in the treatment of HA filler related side effects.12 In addition to discoloration caused by the Tyndall effect, hyaluronidase has been used to treat nodules, granulomatous reactions, impending necrosis and overcorrection caused by HA fillers.8
Hyaluronidase is a soluble protein enzyme that acts at the site of location injection to break down and hydrolyze HA.13 It is FDA approved as an adjuvant in subcutaneous fluid administration for achieving hydration. It comes as a white powder that needs to be diluted with saline before injection. HA fillers with a high degree of cross-linking, like VYC-20L, is more resistant to treatment with hyaluronidase and may require multiple treatments.8 Despite this hurdle, this case study shows that complications from VYC-20L can be successfully treated with hyaluronidase.
Side effects from hyaluronidase are rare and usually local injection site reactions.13 However, allergic and even a few anaphylactic reactions have been reported.12 Most of these were based on ophthalmologic or other non-cosmetic uses of hyaluronidase, but there are cases of angioedema after the use of hyaluronidase for the treatment of HA side effects.12,14 Despite these risks, a meta-analysis of the literature performed in 2009 does not recommend skin testing prior to using hyaluronidase because the allergic reactions are rare and cannot be totally excluded by the skin test.12 This article determined that hyaluronidase is a safe and effective treatment for HA filler adverse events.12
Patient Outcome
Our patient denied side effects from the hyaluronidase. Specifically, the patient denied any post-injection pruritus or edema, which are the most common reported side effects.12 A week after the second injection the patient noticed marked decrease in the discoloration in the area treated (Figure 2). The patient was happy with the improvement.
Conclusion
Correcting mid-facial volume loss is becoming an important treatment of the aging face. As the use of the newly FDA-approved HA filler, VYC-20L increases, the likelihood of adverse events occurring will continue to rise. Hyaluronidase is an effective option for treating adverse events with HA fillers that has been used for many years. This case study demonstrates that hyaluronidase seems to be the most promising treatment of the Tyndall effect caused by this new filler. n
Dr. Torres is a practicing dermatologist at Affiliated Dermatologists in Morristown, NJ, as well as a Mohs and cosmetic surgeon. He also teaches Mohs and cosmetic surgery in a Procedural Dermatology Fellowship accredited by the Accreditation Council for Graduate Medical Education (ACGME).
Dr. Pilcher is a fellow at Affiliated Dermatologists in Morristown, NJ.
Dr. Rogachefsky is a practicing dermatologist and the Program Director of the ACGME-approved Procedural Dermatology Fellowship at Affiliated Dermatologists & Dermatologist Surgeons in Morristown, NJ.
Disclosure: The authors report no relevant financial relationships.
References
1. Donofrio LM. Evaluation and management of the aging face. In: Robinson, JK, Hanke CW, Siegel DM, Fatila A, eds. Surgery of the Skin. Philadelphia, PA: Elsevier, Inc; 2010:351-361.
2. Rohrick RJ, Pessa JE. The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg. 2007;119(7):2219-2227.
3. Calllan P, Goodman GJ, Charlisle I, et al. Efficacy and safety of a hyaluronic acid filler in subjects treated for correction of midface volume deficiency: a 24 month study. Clin Cosmet Investig Dermatol. 2013;6:81-89.
4. Gold M. The science and art of hyaluronic acid dermal filler use in esthetic applications. J Cosmet Dermatol. 2009;8(4):301-307.
5. Hoffmann K. Volumizing effects of smooth, highly cohesive, viscous 20 mg/mL hyaluronic acid volumizing filler: prospective European study. BMC Dermatol. 2009;9:9.
6. Recently approved devices. FDA website. https://www.fda.gov/medicaldevices/productsandmedicalprocedures/deviceapprovalsandclearances/recently-approveddevices/ucm374231.htm. Accessed September 22, 2014.
7. Jones D, Murphy DK. Volumizing hyaluronic acid filler for midface volume deficit: 2-year results from a pivotal single-blind randomized controlled Study. Dermatol Surg. 2013;39(11):1602-1612.
8. Funt D, Pavicic T. Dermal fillers in aesthetics: an overview of adverse events and treatment approaches. Clin Cosmet Investig Dermatol. 2013;6:295-316.
9. Hirsch RJ. Narurkar N, Carruthers J. Management of injected hyaluronic acid induced Tyndall effects. Lasers Surg Med. 2006;38(3):202-204.
10. Douse-Dean T, Jacob CI. Fast and easy treatment for reduction of Tyndall effect secondary to cosmetic use of hyaluronic acid. J Drugs Dermatol. 2008;7(3): 281-283.
11. Philipp-Dormston WG, Eccleston D, De Boulle K, Hilton S, van den Elzen H, Nathan M. A prospective, observational study of the volumizing effect of open-label aesthetic use of Juvéderm® Voluma® with lidocaine in mid-face area. J Cosmet Laser Ther. 2014;16(4):171-179.
12. Rzany B, Becker-Wegerich P, Bachmann F, Erdmann R, Wollina U, Hyaluronidase in the correction of hyaluronic acid-based fillers: a review and recommendation for use. J Cosmet Dermatol. 2009;8(4):317-323.
13. Brody HJ. Use of hyaluronidase in the treatment of granulomatous hyaluronic acid reactions or unwanted hyaluronic acid misplacement. Dermatol Surg. 2005;31(8 Pt 1):893-897.
14. Andre P, Flechet ML. Angioedema after ovine hyaluronidase injection for treating hyaluronic acid overcorrection. J Cosmet Dermatol. 2008;7(2):136-138.
As the population ages, the desire for cosmetic procedures to restore a more youthful appearance will continue to rise. Multiple components contribute to volume loss in the aging face including extrinsic factors such as ultraviolet and oxidative damage as well as intrinsic factors such as bone and soft tissue remodeling, which are both hormonally and genetically regulated.1,2
The loss of volume in the mid-face results in flattening and furrowing of the central area of the mid-cheek. Additionally, displacement of tissues downward and medially cause the depth of the nasolabial fold to be exaggerated.3 Restoring this volume loss has become an integral part of the aesthetic treatment plans for the aging face.
Hyaluronic acid (HA) is a naturally occurring glycosaminoglycan that is a component of the extracellular matrix in connective tissue. HA is naturally non-immunogenic.3,4 HA has a high affinity for water, and thus it plays an integral role in maintaining dermal volume and compressibility.4 Unfortunately, in its natural state, HA has a half-life of only 1 to 2 days due to aqueous dilution and enzymatic degredation.4 In order for HA to be a viable filler, manufacturers had to develop methods to increase resistance to degradation.5
In 2013, the FDA approved a new HA filler specifically for the treatment of mid-face volume loss.6 The product, VYC-20L (Juvéderm Voluma XC, Allergan Inc.), consists of a 20-mg/mL mixture of low molecular weight (<1 mDa) and high molecular weight (>1 mDa) HA resulting in a more mobile network facilitating efficient cross-linking.3,7 Due to the efficient cross-linking, VYC-20L is a highly cohesive gel with greater hardness than other HA products resulting in increased longevity as well as greater lift capability.7
Complications with HA fillers are rare. One rare complication that has been reported with HA fillers is the development of blue papules and nodules in the area treated due to the Tyndall effect. The Tyndall effect occurs when HA fillers are inappropriately implanted into the superficial dermis or epidermis.8 The lesions appear blue because blue light waves have a higher frequency than red light waves and are more easily scattered.8 When light hits the surface of the skin with superficially placed HA particles, the particles cause the light waves to be reflected, and due to increased scatter, blue is the predominant color that emerges.9
Fortunately, the discoloration caused by the Tyndall effect can be treated. The most common treatment of this complication is the use of hyaluronidase, which is an off-label treatment for this condition.8 No cases of discoloration caused by the Tyndall effect occurring with injection of VYC-20L have been reported.
In this article, we present a case study of a patient developing a blue nodule under her right eye after being treated with VYC-20L. The complication was successfully treated with hyaluronidase.
Therapeutic Alternatives
Most HA fillers are known to resorb in 6 to 18 months, although the more superficial the placement of the material, the longer the duration of the discoloration.8 Due to the transient nature of HA fillers, treatment of the blue lesions caused by the Tyndall effect may not be necessary in cases where the lesions are small and imperceptible. However, VYC-20L has been shown in studies to retain its effectiveness for 2 years or longer; therefore, waiting for the product to resorb is not a viable option.3,7
Treatment of the discoloration with a QS Nd:YAG laser at 1,064 nm has been reported.9 In this report, discoloration resolved after 1 treatment with the VersaPulse (Lumenis) 1,064 QS Nd:YAG 3 mm spot, 5.5 J/cm2 and the laser side effects were not elicudated.9 The authors did not know the mechanism of action or the target chromophore, but they theorized that the laser increased HA turnover by some unknown mechanism.
There have been some reports of the discoloration from HA being treated by nicking the skin with a small-gauge needle (30 gauge) or surgical scalpel (#11 blade) and expressing the contents.10 This can be done immediately after injection and up to 12 months or more after placement.10 This procedure is usually done as a last resort after all other treatment options have failed.8
Case Study
Our 61-year-old female patient presented for evaluation of a small blue papule of 3 months duration after injection of VYC-20L under her right eye (Figure 1). Shortly after the injection of VYC-20L and correction of the moderate mid-facial volume defect bilaterally, she noticed development of a blue discoloration in the lower eyelid, which was still present 3 months later. At this time, the lesions were felt to be due to superficially placed HA resulting in the Tyndall effect.
The patient desired to have the color changes treated. Therapeutic options were discussed with the patient and we recommended treatment with hyaluronidase. She was informed of the potential side effects of hyaluronidase and that despite the lack of FDA approval for the treatment of unwanted HA deposits, hyaluronidase has been used efficiently to treat these deposits caused by other HA fillers. After informed consent was obtained, a 150 unit/mL vile of hyaluronidase human injection (Hylenex recombinant, Halozyme Therapeutics) was diluted with 0.9% saline (1 mL). Approximately, 0.1 mL of the mixture was injected into the lesion. About 1 week later the patient received a second injection of 0.1 mL of hyaluronidase into the area. The procedure was well-tolerated by the patient.
Discussion
VYC-20L is the only FDA-approved filler for treatment of moderate-to-severe mid-face volume loss. The clinical study conducted to gain FDA approval showed that 85.6% of patients in the treatment group improved by 1 point or more on the Mid-Face Volume Deficit Scale (MFVDS) at 6 months, and there was a statistically significant difference (P<0.001) between the treatment and control groups.7 Additionally, duration of effectiveness calculated using Kaplan-Meier analysis based on evaluating investigators’ assessments of overall MFVDS showed that 73.9% of subjects were still responders at 1 year and 44.6% were responders at 2 years.7 The results were achieved with the initial treatment and a touch-up procedure 30 days after the initial procedure.
The study also showed that the VYC-20L was a safe product. The majority of the adverse events were treatment site reactions typical of HA injections including tenderness, swelling, firmness and lumps or bumps.7 The researchers reported that the majority of these effects lasted 2 weeks or less and resolved without treatment. Two patients had severe delayed-onset adverse events (1 was lumps in the treatment site that developed at 7 months and the other was cellulitis in the area after an injury that resulted in nodularity) that were treated with antibiotics, anti-inflammatories and hyaluronidase successfully without long-term sequella.7 Thus, VYC-20L is an effective treatment with a good safety profile.
These findings are corroborated by several other studies. In 1 study from Australia, 103 patients received VYC-20L to the mid-face. Of the patients enrolled, 96% were documented to be MFVDS responders at 8 weeks and 88.2% and 81.5% were still responders at weeks 52 and 78, respectively, after the initial treatment and 1 touch-up procedure at week 4.3. Patient satisfaction with the product was high with 70 of 72 patients that completed the study saying they would recommend the product.3
In this study, adverse events were transient and infrequent. The most common adverse events reported were injection site bruising and swelling.3 Several other studies from Europe with shorter follow-up times also showed high patient satisfaction and low occurrence of adverse events.5,11 None of the studies reported development of any skin discoloration from the Tyndall effect.
Despite being used off-label, several clinical trials and many case reports show the effectiveness of hyaluronidase in the treatment of HA filler related side effects.12 In addition to discoloration caused by the Tyndall effect, hyaluronidase has been used to treat nodules, granulomatous reactions, impending necrosis and overcorrection caused by HA fillers.8
Hyaluronidase is a soluble protein enzyme that acts at the site of location injection to break down and hydrolyze HA.13 It is FDA approved as an adjuvant in subcutaneous fluid administration for achieving hydration. It comes as a white powder that needs to be diluted with saline before injection. HA fillers with a high degree of cross-linking, like VYC-20L, is more resistant to treatment with hyaluronidase and may require multiple treatments.8 Despite this hurdle, this case study shows that complications from VYC-20L can be successfully treated with hyaluronidase.
Side effects from hyaluronidase are rare and usually local injection site reactions.13 However, allergic and even a few anaphylactic reactions have been reported.12 Most of these were based on ophthalmologic or other non-cosmetic uses of hyaluronidase, but there are cases of angioedema after the use of hyaluronidase for the treatment of HA side effects.12,14 Despite these risks, a meta-analysis of the literature performed in 2009 does not recommend skin testing prior to using hyaluronidase because the allergic reactions are rare and cannot be totally excluded by the skin test.12 This article determined that hyaluronidase is a safe and effective treatment for HA filler adverse events.12
Patient Outcome
Our patient denied side effects from the hyaluronidase. Specifically, the patient denied any post-injection pruritus or edema, which are the most common reported side effects.12 A week after the second injection the patient noticed marked decrease in the discoloration in the area treated (Figure 2). The patient was happy with the improvement.
Conclusion
Correcting mid-facial volume loss is becoming an important treatment of the aging face. As the use of the newly FDA-approved HA filler, VYC-20L increases, the likelihood of adverse events occurring will continue to rise. Hyaluronidase is an effective option for treating adverse events with HA fillers that has been used for many years. This case study demonstrates that hyaluronidase seems to be the most promising treatment of the Tyndall effect caused by this new filler. n
Dr. Torres is a practicing dermatologist at Affiliated Dermatologists in Morristown, NJ, as well as a Mohs and cosmetic surgeon. He also teaches Mohs and cosmetic surgery in a Procedural Dermatology Fellowship accredited by the Accreditation Council for Graduate Medical Education (ACGME).
Dr. Pilcher is a fellow at Affiliated Dermatologists in Morristown, NJ.
Dr. Rogachefsky is a practicing dermatologist and the Program Director of the ACGME-approved Procedural Dermatology Fellowship at Affiliated Dermatologists & Dermatologist Surgeons in Morristown, NJ.
Disclosure: The authors report no relevant financial relationships.
References
1. Donofrio LM. Evaluation and management of the aging face. In: Robinson, JK, Hanke CW, Siegel DM, Fatila A, eds. Surgery of the Skin. Philadelphia, PA: Elsevier, Inc; 2010:351-361.
2. Rohrick RJ, Pessa JE. The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg. 2007;119(7):2219-2227.
3. Calllan P, Goodman GJ, Charlisle I, et al. Efficacy and safety of a hyaluronic acid filler in subjects treated for correction of midface volume deficiency: a 24 month study. Clin Cosmet Investig Dermatol. 2013;6:81-89.
4. Gold M. The science and art of hyaluronic acid dermal filler use in esthetic applications. J Cosmet Dermatol. 2009;8(4):301-307.
5. Hoffmann K. Volumizing effects of smooth, highly cohesive, viscous 20 mg/mL hyaluronic acid volumizing filler: prospective European study. BMC Dermatol. 2009;9:9.
6. Recently approved devices. FDA website. https://www.fda.gov/medicaldevices/productsandmedicalprocedures/deviceapprovalsandclearances/recently-approveddevices/ucm374231.htm. Accessed September 22, 2014.
7. Jones D, Murphy DK. Volumizing hyaluronic acid filler for midface volume deficit: 2-year results from a pivotal single-blind randomized controlled Study. Dermatol Surg. 2013;39(11):1602-1612.
8. Funt D, Pavicic T. Dermal fillers in aesthetics: an overview of adverse events and treatment approaches. Clin Cosmet Investig Dermatol. 2013;6:295-316.
9. Hirsch RJ. Narurkar N, Carruthers J. Management of injected hyaluronic acid induced Tyndall effects. Lasers Surg Med. 2006;38(3):202-204.
10. Douse-Dean T, Jacob CI. Fast and easy treatment for reduction of Tyndall effect secondary to cosmetic use of hyaluronic acid. J Drugs Dermatol. 2008;7(3): 281-283.
11. Philipp-Dormston WG, Eccleston D, De Boulle K, Hilton S, van den Elzen H, Nathan M. A prospective, observational study of the volumizing effect of open-label aesthetic use of Juvéderm® Voluma® with lidocaine in mid-face area. J Cosmet Laser Ther. 2014;16(4):171-179.
12. Rzany B, Becker-Wegerich P, Bachmann F, Erdmann R, Wollina U, Hyaluronidase in the correction of hyaluronic acid-based fillers: a review and recommendation for use. J Cosmet Dermatol. 2009;8(4):317-323.
13. Brody HJ. Use of hyaluronidase in the treatment of granulomatous hyaluronic acid reactions or unwanted hyaluronic acid misplacement. Dermatol Surg. 2005;31(8 Pt 1):893-897.
14. Andre P, Flechet ML. Angioedema after ovine hyaluronidase injection for treating hyaluronic acid overcorrection. J Cosmet Dermatol. 2008;7(2):136-138.
As the population ages, the desire for cosmetic procedures to restore a more youthful appearance will continue to rise. Multiple components contribute to volume loss in the aging face including extrinsic factors such as ultraviolet and oxidative damage as well as intrinsic factors such as bone and soft tissue remodeling, which are both hormonally and genetically regulated.1,2
The loss of volume in the mid-face results in flattening and furrowing of the central area of the mid-cheek. Additionally, displacement of tissues downward and medially cause the depth of the nasolabial fold to be exaggerated.3 Restoring this volume loss has become an integral part of the aesthetic treatment plans for the aging face.
Hyaluronic acid (HA) is a naturally occurring glycosaminoglycan that is a component of the extracellular matrix in connective tissue. HA is naturally non-immunogenic.3,4 HA has a high affinity for water, and thus it plays an integral role in maintaining dermal volume and compressibility.4 Unfortunately, in its natural state, HA has a half-life of only 1 to 2 days due to aqueous dilution and enzymatic degredation.4 In order for HA to be a viable filler, manufacturers had to develop methods to increase resistance to degradation.5
In 2013, the FDA approved a new HA filler specifically for the treatment of mid-face volume loss.6 The product, VYC-20L (Juvéderm Voluma XC, Allergan Inc.), consists of a 20-mg/mL mixture of low molecular weight (<1 mDa) and high molecular weight (>1 mDa) HA resulting in a more mobile network facilitating efficient cross-linking.3,7 Due to the efficient cross-linking, VYC-20L is a highly cohesive gel with greater hardness than other HA products resulting in increased longevity as well as greater lift capability.7
Complications with HA fillers are rare. One rare complication that has been reported with HA fillers is the development of blue papules and nodules in the area treated due to the Tyndall effect. The Tyndall effect occurs when HA fillers are inappropriately implanted into the superficial dermis or epidermis.8 The lesions appear blue because blue light waves have a higher frequency than red light waves and are more easily scattered.8 When light hits the surface of the skin with superficially placed HA particles, the particles cause the light waves to be reflected, and due to increased scatter, blue is the predominant color that emerges.9
Fortunately, the discoloration caused by the Tyndall effect can be treated. The most common treatment of this complication is the use of hyaluronidase, which is an off-label treatment for this condition.8 No cases of discoloration caused by the Tyndall effect occurring with injection of VYC-20L have been reported.
In this article, we present a case study of a patient developing a blue nodule under her right eye after being treated with VYC-20L. The complication was successfully treated with hyaluronidase.
Therapeutic Alternatives
Most HA fillers are known to resorb in 6 to 18 months, although the more superficial the placement of the material, the longer the duration of the discoloration.8 Due to the transient nature of HA fillers, treatment of the blue lesions caused by the Tyndall effect may not be necessary in cases where the lesions are small and imperceptible. However, VYC-20L has been shown in studies to retain its effectiveness for 2 years or longer; therefore, waiting for the product to resorb is not a viable option.3,7
Treatment of the discoloration with a QS Nd:YAG laser at 1,064 nm has been reported.9 In this report, discoloration resolved after 1 treatment with the VersaPulse (Lumenis) 1,064 QS Nd:YAG 3 mm spot, 5.5 J/cm2 and the laser side effects were not elicudated.9 The authors did not know the mechanism of action or the target chromophore, but they theorized that the laser increased HA turnover by some unknown mechanism.
There have been some reports of the discoloration from HA being treated by nicking the skin with a small-gauge needle (30 gauge) or surgical scalpel (#11 blade) and expressing the contents.10 This can be done immediately after injection and up to 12 months or more after placement.10 This procedure is usually done as a last resort after all other treatment options have failed.8
Case Study
Our 61-year-old female patient presented for evaluation of a small blue papule of 3 months duration after injection of VYC-20L under her right eye (Figure 1). Shortly after the injection of VYC-20L and correction of the moderate mid-facial volume defect bilaterally, she noticed development of a blue discoloration in the lower eyelid, which was still present 3 months later. At this time, the lesions were felt to be due to superficially placed HA resulting in the Tyndall effect.
The patient desired to have the color changes treated. Therapeutic options were discussed with the patient and we recommended treatment with hyaluronidase. She was informed of the potential side effects of hyaluronidase and that despite the lack of FDA approval for the treatment of unwanted HA deposits, hyaluronidase has been used efficiently to treat these deposits caused by other HA fillers. After informed consent was obtained, a 150 unit/mL vile of hyaluronidase human injection (Hylenex recombinant, Halozyme Therapeutics) was diluted with 0.9% saline (1 mL). Approximately, 0.1 mL of the mixture was injected into the lesion. About 1 week later the patient received a second injection of 0.1 mL of hyaluronidase into the area. The procedure was well-tolerated by the patient.
Discussion
VYC-20L is the only FDA-approved filler for treatment of moderate-to-severe mid-face volume loss. The clinical study conducted to gain FDA approval showed that 85.6% of patients in the treatment group improved by 1 point or more on the Mid-Face Volume Deficit Scale (MFVDS) at 6 months, and there was a statistically significant difference (P<0.001) between the treatment and control groups.7 Additionally, duration of effectiveness calculated using Kaplan-Meier analysis based on evaluating investigators’ assessments of overall MFVDS showed that 73.9% of subjects were still responders at 1 year and 44.6% were responders at 2 years.7 The results were achieved with the initial treatment and a touch-up procedure 30 days after the initial procedure.
The study also showed that the VYC-20L was a safe product. The majority of the adverse events were treatment site reactions typical of HA injections including tenderness, swelling, firmness and lumps or bumps.7 The researchers reported that the majority of these effects lasted 2 weeks or less and resolved without treatment. Two patients had severe delayed-onset adverse events (1 was lumps in the treatment site that developed at 7 months and the other was cellulitis in the area after an injury that resulted in nodularity) that were treated with antibiotics, anti-inflammatories and hyaluronidase successfully without long-term sequella.7 Thus, VYC-20L is an effective treatment with a good safety profile.
These findings are corroborated by several other studies. In 1 study from Australia, 103 patients received VYC-20L to the mid-face. Of the patients enrolled, 96% were documented to be MFVDS responders at 8 weeks and 88.2% and 81.5% were still responders at weeks 52 and 78, respectively, after the initial treatment and 1 touch-up procedure at week 4.3. Patient satisfaction with the product was high with 70 of 72 patients that completed the study saying they would recommend the product.3
In this study, adverse events were transient and infrequent. The most common adverse events reported were injection site bruising and swelling.3 Several other studies from Europe with shorter follow-up times also showed high patient satisfaction and low occurrence of adverse events.5,11 None of the studies reported development of any skin discoloration from the Tyndall effect.
Despite being used off-label, several clinical trials and many case reports show the effectiveness of hyaluronidase in the treatment of HA filler related side effects.12 In addition to discoloration caused by the Tyndall effect, hyaluronidase has been used to treat nodules, granulomatous reactions, impending necrosis and overcorrection caused by HA fillers.8
Hyaluronidase is a soluble protein enzyme that acts at the site of location injection to break down and hydrolyze HA.13 It is FDA approved as an adjuvant in subcutaneous fluid administration for achieving hydration. It comes as a white powder that needs to be diluted with saline before injection. HA fillers with a high degree of cross-linking, like VYC-20L, is more resistant to treatment with hyaluronidase and may require multiple treatments.8 Despite this hurdle, this case study shows that complications from VYC-20L can be successfully treated with hyaluronidase.
Side effects from hyaluronidase are rare and usually local injection site reactions.13 However, allergic and even a few anaphylactic reactions have been reported.12 Most of these were based on ophthalmologic or other non-cosmetic uses of hyaluronidase, but there are cases of angioedema after the use of hyaluronidase for the treatment of HA side effects.12,14 Despite these risks, a meta-analysis of the literature performed in 2009 does not recommend skin testing prior to using hyaluronidase because the allergic reactions are rare and cannot be totally excluded by the skin test.12 This article determined that hyaluronidase is a safe and effective treatment for HA filler adverse events.12
Patient Outcome
Our patient denied side effects from the hyaluronidase. Specifically, the patient denied any post-injection pruritus or edema, which are the most common reported side effects.12 A week after the second injection the patient noticed marked decrease in the discoloration in the area treated (Figure 2). The patient was happy with the improvement.
Conclusion
Correcting mid-facial volume loss is becoming an important treatment of the aging face. As the use of the newly FDA-approved HA filler, VYC-20L increases, the likelihood of adverse events occurring will continue to rise. Hyaluronidase is an effective option for treating adverse events with HA fillers that has been used for many years. This case study demonstrates that hyaluronidase seems to be the most promising treatment of the Tyndall effect caused by this new filler. n
Dr. Torres is a practicing dermatologist at Affiliated Dermatologists in Morristown, NJ, as well as a Mohs and cosmetic surgeon. He also teaches Mohs and cosmetic surgery in a Procedural Dermatology Fellowship accredited by the Accreditation Council for Graduate Medical Education (ACGME).
Dr. Pilcher is a fellow at Affiliated Dermatologists in Morristown, NJ.
Dr. Rogachefsky is a practicing dermatologist and the Program Director of the ACGME-approved Procedural Dermatology Fellowship at Affiliated Dermatologists & Dermatologist Surgeons in Morristown, NJ.
Disclosure: The authors report no relevant financial relationships.
References
1. Donofrio LM. Evaluation and management of the aging face. In: Robinson, JK, Hanke CW, Siegel DM, Fatila A, eds. Surgery of the Skin. Philadelphia, PA: Elsevier, Inc; 2010:351-361.
2. Rohrick RJ, Pessa JE. The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg. 2007;119(7):2219-2227.
3. Calllan P, Goodman GJ, Charlisle I, et al. Efficacy and safety of a hyaluronic acid filler in subjects treated for correction of midface volume deficiency: a 24 month study. Clin Cosmet Investig Dermatol. 2013;6:81-89.
4. Gold M. The science and art of hyaluronic acid dermal filler use in esthetic applications. J Cosmet Dermatol. 2009;8(4):301-307.
5. Hoffmann K. Volumizing effects of smooth, highly cohesive, viscous 20 mg/mL hyaluronic acid volumizing filler: prospective European study. BMC Dermatol. 2009;9:9.
6. Recently approved devices. FDA website. https://www.fda.gov/medicaldevices/productsandmedicalprocedures/deviceapprovalsandclearances/recently-approveddevices/ucm374231.htm. Accessed September 22, 2014.
7. Jones D, Murphy DK. Volumizing hyaluronic acid filler for midface volume deficit: 2-year results from a pivotal single-blind randomized controlled Study. Dermatol Surg. 2013;39(11):1602-1612.
8. Funt D, Pavicic T. Dermal fillers in aesthetics: an overview of adverse events and treatment approaches. Clin Cosmet Investig Dermatol. 2013;6:295-316.
9. Hirsch RJ. Narurkar N, Carruthers J. Management of injected hyaluronic acid induced Tyndall effects. Lasers Surg Med. 2006;38(3):202-204.
10. Douse-Dean T, Jacob CI. Fast and easy treatment for reduction of Tyndall effect secondary to cosmetic use of hyaluronic acid. J Drugs Dermatol. 2008;7(3): 281-283.
11. Philipp-Dormston WG, Eccleston D, De Boulle K, Hilton S, van den Elzen H, Nathan M. A prospective, observational study of the volumizing effect of open-label aesthetic use of Juvéderm® Voluma® with lidocaine in mid-face area. J Cosmet Laser Ther. 2014;16(4):171-179.
12. Rzany B, Becker-Wegerich P, Bachmann F, Erdmann R, Wollina U, Hyaluronidase in the correction of hyaluronic acid-based fillers: a review and recommendation for use. J Cosmet Dermatol. 2009;8(4):317-323.
13. Brody HJ. Use of hyaluronidase in the treatment of granulomatous hyaluronic acid reactions or unwanted hyaluronic acid misplacement. Dermatol Surg. 2005;31(8 Pt 1):893-897.
14. Andre P, Flechet ML. Angioedema after ovine hyaluronidase injection for treating hyaluronic acid overcorrection. J Cosmet Dermatol. 2008;7(2):136-138.