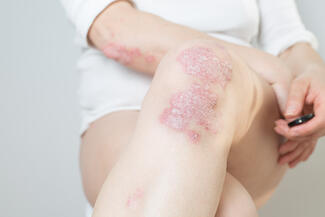
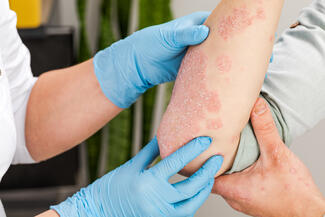
Oral Icotrokinra Demonstrates High Efficacy and Favorable Safety in Plaque Psoriasis
Icotrokinra, an oral interleukin-23 (IL-23) receptor antagonist peptide, demonstrated robust efficacy and a safety profile comparable to placebo in patients with moderate-to-severe plaque psoriasis, according to a systematic review and meta-analysis of randomized controlled trials. The findings suggest a potential oral alternative to injectable biologics.
Investigators analyzed 5 randomized trials including 1951 patients receiving icotrokinra 200 mg once daily or placebo. The primary endpoint was achievement of a 75% reduction in Psoriasis Area and Severity Index (PASI 75) at week 16.
Clinical responses were observed early and sustained over time. At week 4, PASI 75 response rates were higher with icotrokinra compared with placebo (15% vs 2%). By week 16, 73% of patients receiving icotrokinra achieved PASI 75 compared with 11% in the placebo group. Higher clearance thresholds also favored icotrokinra, with PASI 90 achieved in 54% vs 4% and PASI 100 in 30% vs 1%.
The authors reported that “icotrokinra 200 mg once daily significantly increased PASI 75 response rates compared with placebo,” and concluded that the therapy offers “rapid and sustained clinical benefit with a safety profile comparable to placebo.”
Safety outcomes were similar between treatment groups. Rates of adverse events, serious adverse events, and infections did not differ significantly, suggesting good tolerability.
These results are notable given the limitations of existing oral therapies for psoriasis, which often show modest efficacy or safety concerns. Icotrokinra’s mechanism—selective inhibition of the IL-23 receptor—targets a central inflammatory pathway in psoriasis while offering the convenience of oral administration.
Reference
Altayf A, Lateiresh M, Marwan L, Alfeetouri MY, Zouaoui Y, Elhadi M. Efficacy and safety of oral icotrokinra in moderate-to-severe plaque psoriasis: a systematic review, meta-analysis, and trial sequential analysis. Int J Dermatol. Published online March 17, 2026. doi:10.1111/ijd.70380