The Prevalence of Cardiac Symptoms in Ehlers-Danlos Syndrome and Hypermobility Spectrum Disorder: A Pilot Study
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/25.00395. Epub April 21, 2026.
Abstract
Objectives. The authors compared palpitations, presyncope, syncope, and postural orthostatic tachycardia syndrome (POTS) prevalence among patients with hypermobile Ehlers-Danlos syndrome (hEDS) and hypermobility spectrum disorder (HSD).
Methods. A retrospective analysis of patients referred for cardiac evaluation at the New York Institute of Technology Ehlers-Danlos Syndrome/Hypermobility Treatment Center between January 2019 and November 2023 was conducted. hEDS and HSD patients underwent cardiac evaluation and were stratified by median age into younger (<35 years) and older (≥35 years) groups. Symptom prevalence was compared between hypermobile patients overall and by age cohort. Data were presented as mean ± SD; P < .05 was considered statistically significant.
Results. One hundred-nine patients (75 hEDS, 34 HSD) underwent cardiac evaluation: 75 hEDS (71 females/4 males; age 33.9 + 11.6 years) and 34 HSD (29 females/ 5 males; age 40.0 + 13.8 years). POTS (48% vs 23.5%, P = .016) was significantly more prevalent in the hEDS group; palpitations, presyncope,
Conclusions. Patients with hEDS exhibited greater symptoms than HSD patients, and symptoms were particularly more pronounced with age. POTS prevalence decreased with age in both groups. Larger prospective studies are needed to explore underlying mechanisms.
Introduction
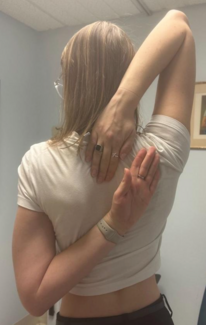
Ehlers-Danlos syndrome (EDS) is a connective tissue disorder characterized by defects in collagen synthesis. Categorized into 13 subtypes, EDS has an estimated prevalence of 1 in 5000, with the most common subtype being hypermobile EDS (hEDS).1,2 Other common EDS subtypes include classical-like and vascular EDS, which may be diagnosed through genetic testing. Patients with hEDS typically present with complaints of musculoskeletal pain and symptoms of skin elasticity and joint laxity (Figure 1).2 The diagnosis of hEDS is clinical and was established based on the 2017 International Classification of Ehlers-Danlos syndromes, which requires the following criteria to be met: (1) generalized joint hypermobility, (2) generalized systemic manifestations of connective tissue disorder, musculoskeletal complaints, and/or family history in a first degree relative, and (3) exclusion of alternative and rheumatologic diagnoses.3,4 Those who do not meet the diagnostic criteria may subsequently be diagnosed with hypermobility spectrum disorder (HSD).
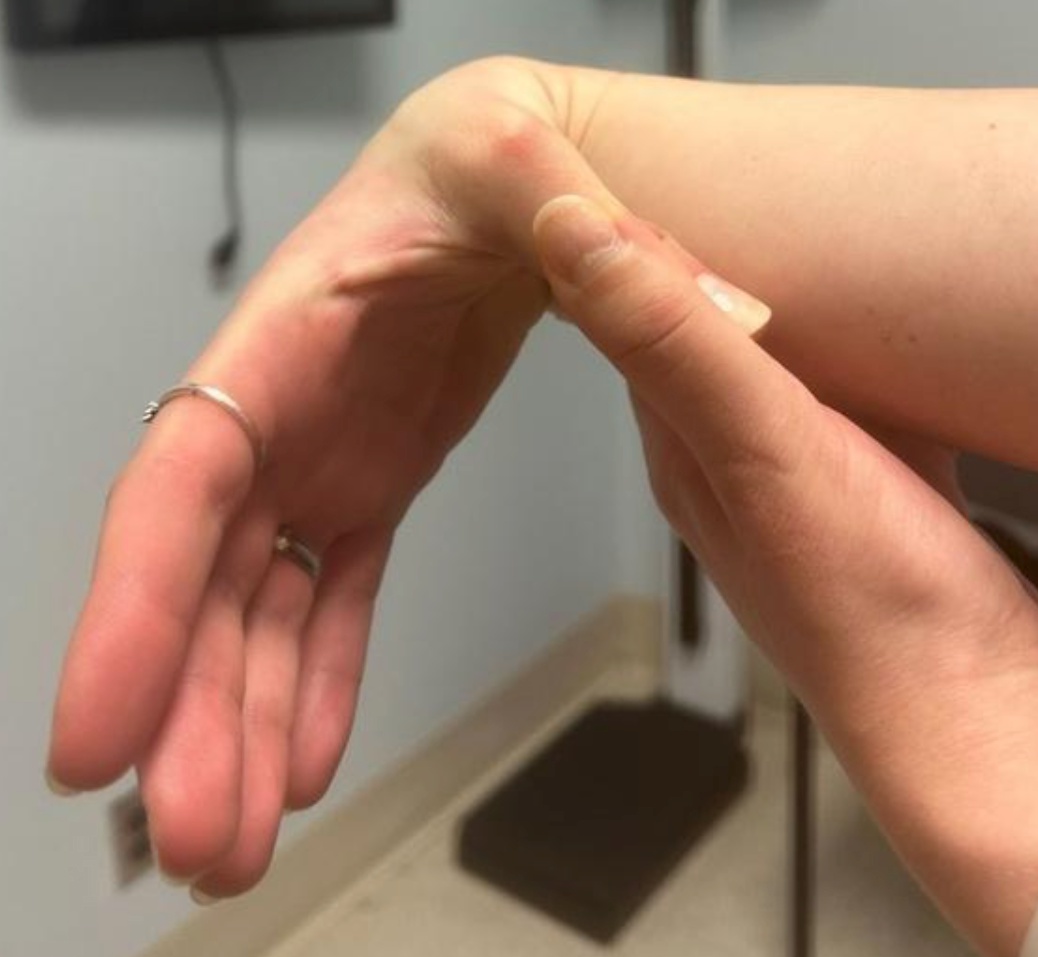
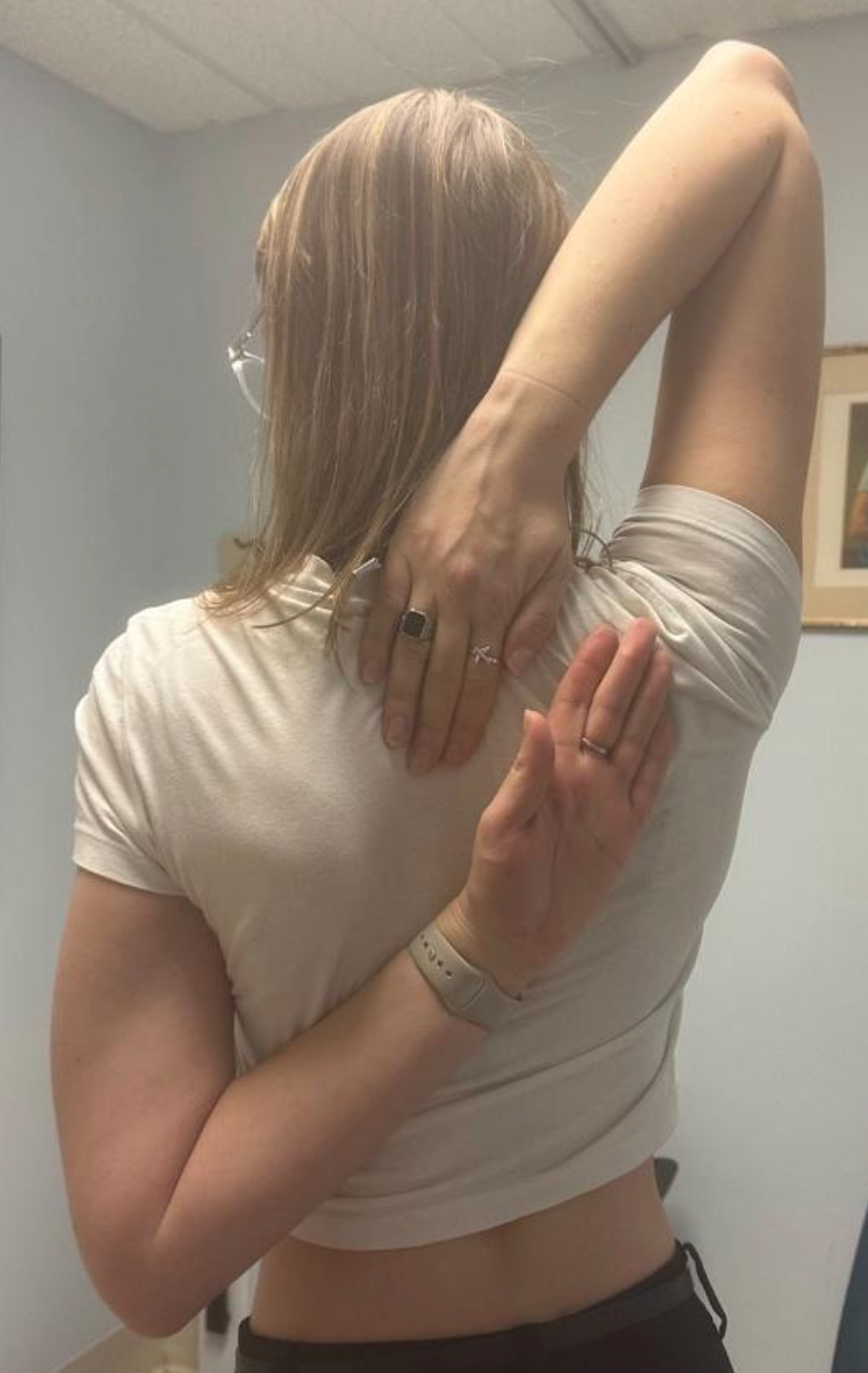
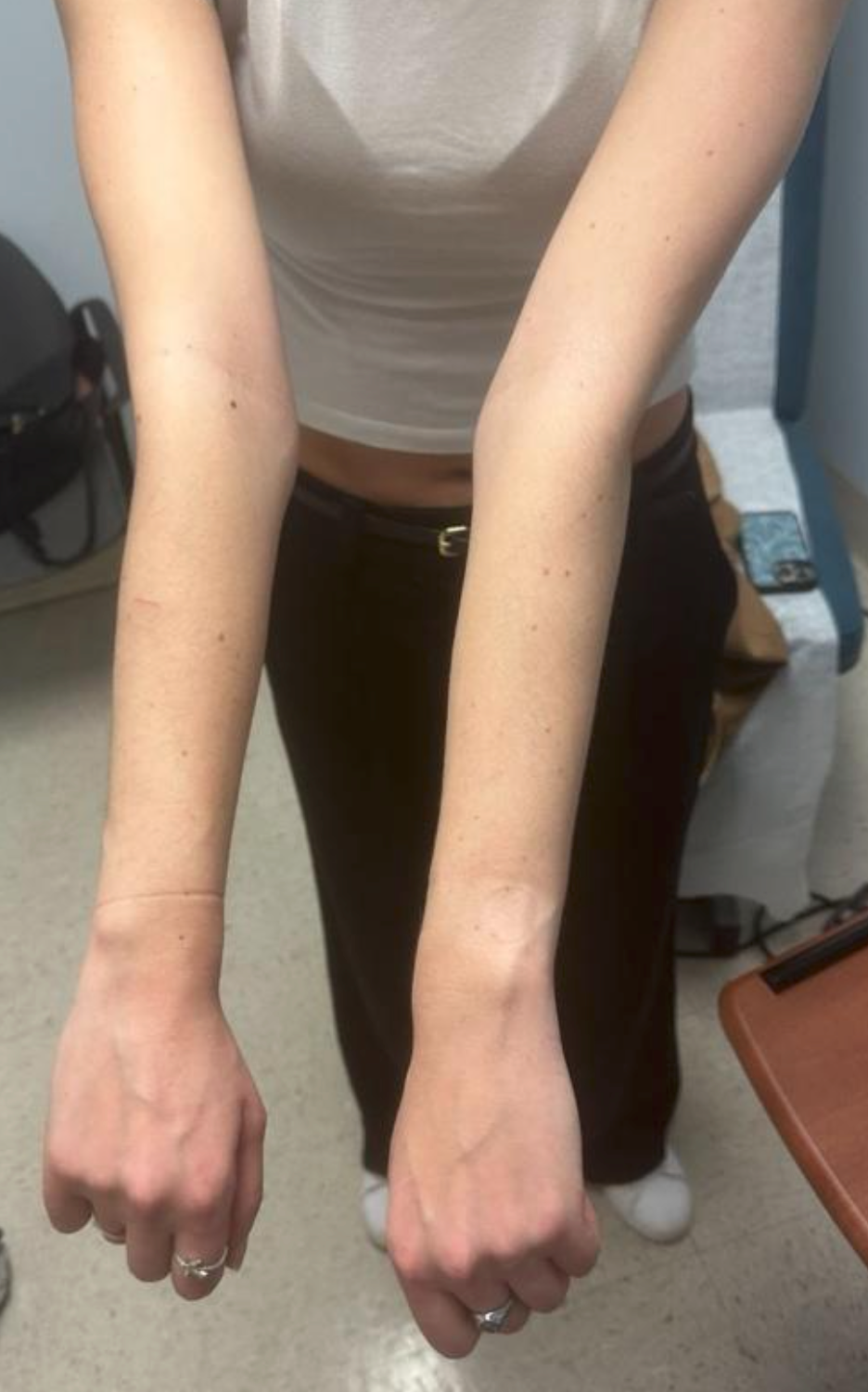
Hypermobile patients, such as those with hEDS and HSD, may experience cardiac symptoms such as palpitations, presyncope, and/or syncope. Some of these symptoms may be attributed to a comorbid condition known as postural tachycardia syndrome (POTS).3 Patients with POTS, a common form of dysautonomia in hypermobile patients, often experience orthostatic intolerance related to impaired venous return and abnormal autonomic regulation upon assuming upright posture that results in compensatory tachycardia and presyncopal symptoms.5 The diagnosis of POTS is based on a combination of clinical history and objective assessment of orthostatic heart rate response. Diagnostic criteria include a sustained increase in heart rate of at least 30 beats per minute within 10 minutes of upright posture in the absence of orthostatic hypotension. While tilt table testing (TTT) may be used to support diagnosis, alternative assessments such as active standing test are also commonly utilized, and formal autonomic testing is not required in all cases.5,6
The propensity to develop cardiac symptoms in hypermobile patients emphasizes the multisystem involvement of these connective tissue disorders. The objective of this study was to investigate and compare the prevalence of palpitations, presyncope/syncope, and POTS in patients with hEDS and HSD referred from a hypermobility specialty clinic.
Methods
Between January 2019 and November 2023, patients diagnosed with hypermobile EDS (hEDS) or HSD were referred from the New York Institute of Technology (NYIT) Ehlers-Danlos Syndrome/Hypermobility Treatment Center to the Long Island Heart Rhythm Center (LIHRC) for cardiac evaluation. Patients with genetically defined EDS subtypes, including classical-like EDS and vascular EDS, were excluded from the present analysis because of their distinct genetic, structural, and cardiovascular risk profiles. Retrospective observational studies from the LIHRC are granted exempt status, and informed consent was waived by the NYITCOM Institutional Review Board (BHS-1465 - Diagnostic and Therapeutic Outcomes from the Long Island Heart Rhythm Center).
Diagnoses of hEDS and HSD were established clinically by providers specializing in hypermobility disorders using the 2017 International Classification of the Ehlers-Danlos Syndromes.4 Diagnostic assessment incorporated Beighton scoring for generalized joint hypermobility, evaluation for system manifestations of connective tissue disorder, musculoskeletal features, exclusion of alternative rheumatologic diagnoses, and review of family history when available. Currently, a molecular diagnostic marker for hEDS is unavailable; therefore, classification is based on clinical criteria and expert assessment. Patients who demonstrated symptomatic joint hypermobility but did not fully meet criteria for hEDS were classified as having HSD. All patients underwent cardiac consultation by a cardiologist who works regularly with hypermobile patients as part of the NYIT EDS/Hypermobility Treatment Center’s multidisciplinary team, and underwent a history, physical exam, and 12-lead electrocardiogram if possible.
If indicated, some symptomatic patients were referred for external cardiac monitoring, echocardiography, and, if necessary, an electrophysiology study. Patients with positional symptoms were considered for further evaluation, including TTT. Diagnosis of POTS was determined based on characteristic clinical symptoms (eg, tachycardia and presyncope upon standing) in conjunction with objective hemodynamic findings defined as an increase in heart rate of 30 or more beats per minute within 10 minutes of standing or head-up tilt or a sustained heart rate of 120 or more beats per minute, assessed via TTT or active standing test. Presyncope, syncope, palpitations, and postural orthostatic tachycardia syndrome were selected as outcomes of interest, as these symptoms commonly prompted cardiology referral and were consistently documented during cardiac evaluation in hypermobile patients.
Symptoms were ascertained through retrospective review of cardiology consultation notes and longitudinal electronic health record documentation, based on patient-reported history documented during evaluation. While patients with hEDS and HSD commonly experience a broad range of multisystem manifestations, including fatigue, chronic pain, and gastrointestinal dysmotility, these features were outside the scope of the present cardiac-focused analysis. The prevalence of palpitations, POTS, and presyncope/syncope was analyzed and compared between patients with hEDS and HSD using chi-squared analysis. The median age of the entire population was determined and used as the cutoff point between the younger (Cohort 1) and older cohorts (Cohort 2). The prevalence of cardiac symptoms (palpitations, presyncope/syncope, and POTS) among both cohorts, together and separately, was analyzed using chi-squared analysis. All statistical analyses were performed using Excel version 16.87 (Microsoft Corporation). Data is reported as mean ± standard deviation, and a P-value of less than or equal to 0.05 was considered statistically significant.
Results
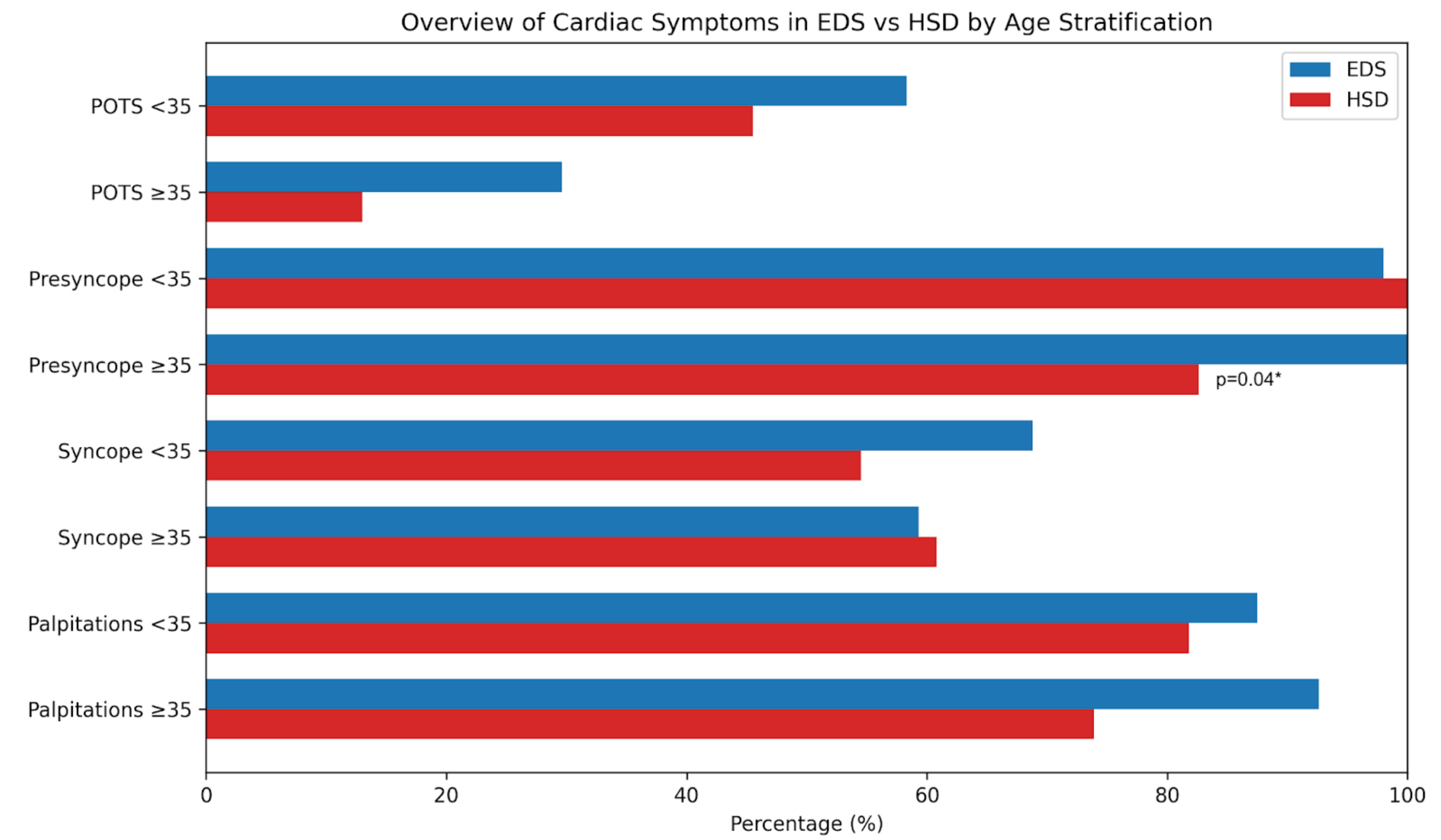
There were 109 hypermobile patients (75 hEDS/34 HSD) with a median age of 35.9 + 12.6 years who underwent cardiac evaluation. The hEDS cohort consisted of 75 patients (71 females and 4 males) with an average age of 33.9 + 11.6 years. The HSD cohort consisted of 34 patients (29 females and 5 males) with an average age of 40.3 + 13.8 years. Presyncope was the most commonly reported symptom among both patient cohorts and was present in 98.6% of those with hEDS and 94.1% of those with HSD (P = .58). Syncope was present in 65.3% of patients with hEDS and 58.8% of patients with HSD (P = .50. Palpitations were noted in 90.0% of patients with hEDS and 76.5% of patients with HSD (P = .11). The prevalence of POTS was significantly higher in patients with hEDS compared with HSD (48% vs 23.5%, P = .016; Table 1).

Age-stratified analysis
Further analysis was conducted by stratifying patients into 2 age groups based on the mean and data distribution: the age range cutoff was younger than 35 years and 35 years or older. Of the 109 hypermobile patients, 59 (48 hEDS/11 HSD) were younger than 35 years (Cohort 1), and 50 (27 hEDS/23 HSD) were 35 years or older (Cohort 2).
In Cohort 1, 48 patients had hEDS (46 females and 2 males), with an average age of 26.2 ± 4.4 years. Eleven patients (10 females and 1 male) had HSD, with an average age of 25.6 ± 4.7 years. Almost all patients with hEDS (98%) and HSD (100%) had symptoms of presyncope (P = 1.0). Syncope was present in the majority of both patient populations (68.8% vs 54.5, P = .35). Palpitations were noted in 87.5% of patients with hEDS and 81.2% of patients with HSD (P = .60). Additionally, POTS was present in 58.3% of patients with hEDS and 45.5% of patients with HSD (P = .44) (Table 2).
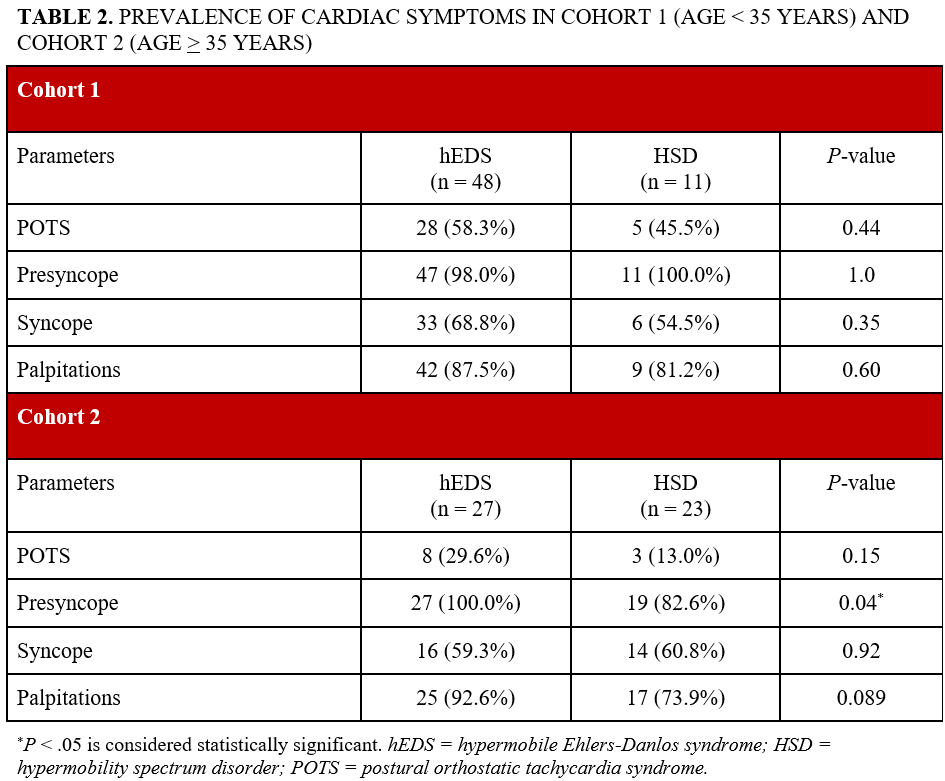
In Cohort 2, 27 patients had hEDS (25 females and 2 males), with an average age of 47.5 ± 8.9 years. Twenty-three patients (19 females and 4 males) had HSD, with an average age of 47.4 ± 10.8 years. Presyncope was prevalent in 100% of patients with hEDS and 82.6% of patients with HSD (P = .04). Palpitations were noted in 92.6% of patients with hEDS and 73.9% of patients with HSD (P = .089). Additionally, POTS was present in 29.6% of patients with hEDS and 13.0% of patients with HSD (P = .15) (Table 2).
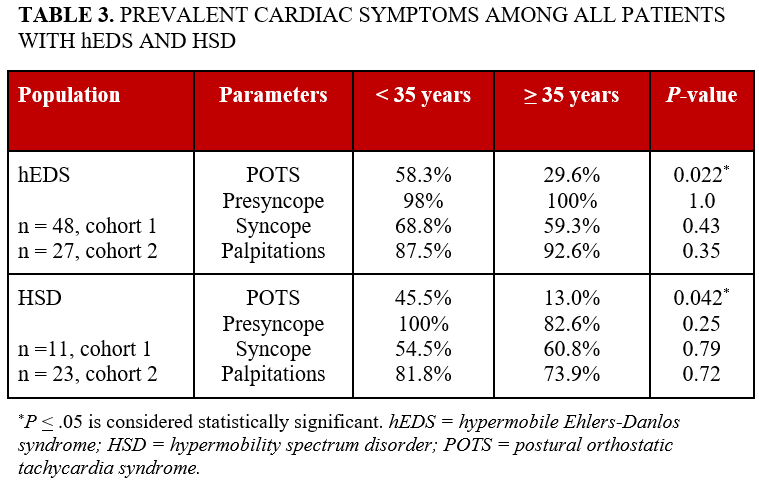
Among all patients with hEDS, POTS was significantly more prevalent in the younger cohort when compared with their older counterparts (58.3% vs 29.6%, P = .022; Table 3). Presyncope was present in 98% of those younger than 35 years and 100% of those 35 years or older (P = 1.0). Palpitations were noted in 87.5% of those younger than 35 years and 92.6% of those 35 years or older (P = .35). Similarly, among all patients with HSD, POTS was significantly more common in the younger cohort (45.5% vs 13.0%, P = .042). Presyncope/syncope was present in 100% of those younger than 35 years and in 82.6% of those 35 years or older (P = .25). Palpitations were noted in 81.8% of those younger than 35 years and 73.9% of those 35 years or older (P = .72).
Diagnostic testing utilization
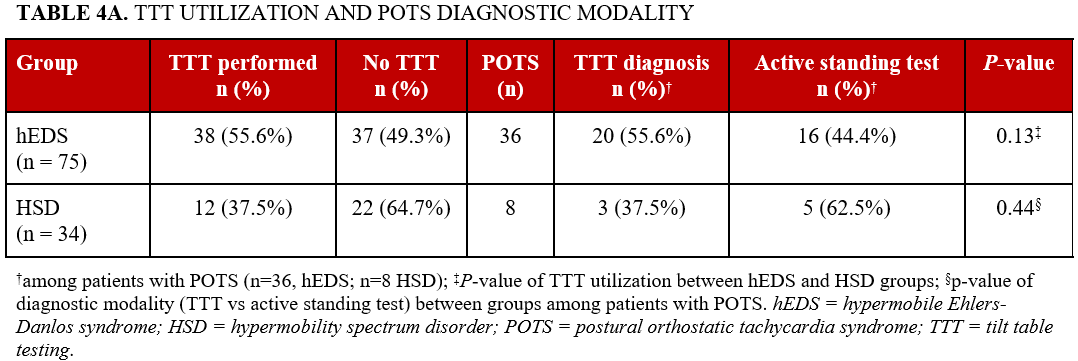
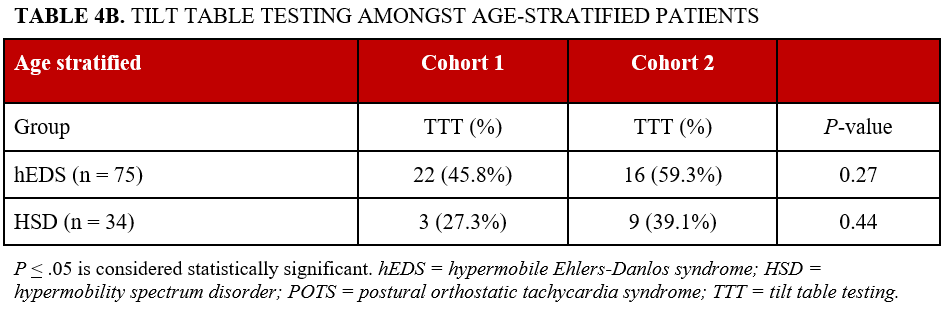
POTS diagnosis was assessed by TTT and active standing test. TTT was performed in 50 of 109 patients (45.9%) across all patients, including 38 of 75 patients with hEDS (50.7%) and 12 of 34 patients with HSD (35.3%) (P = .13). Among all patients with POTS, diagnosis was assessed with active standing test in 21 of 44 patients (47.7%) and TTT in 23 of 44 patients (52.2%). Diagnostic modality did not differ significantly between groups (P = .44, Table 4A). TTT utilization also did not differ significantly between age cohorts (Table 4B). In the hEDS cohort, TTT was performed in 22 of 48 patients younger than 35 years and 16 of 27 patients 35 years or older (P = .27); in the HSD cohort, TTT was performed in 3 of 11 patients younger than 35 years and 9 of 23 patients 35 years or older (P = .44).
Discussion
Hypermobile patients may demonstrate mild structural cardiac findings such as mitral valve prolapse or non-progressive aortic root dilation; however, major structural cardiovascular complications are uncommon.7,8 Morbidity in these populations more frequently arises from autonomic dysfunction and multisystem burden rather than progressive structural heart disease.8,9 hEDS and HSD groups are frequently associated with cardiac-related symptoms, particularly palpitations, presyncope/syncope, and POTS. The comparison of these symptoms between patients with hEDS and HSD has not been thoroughly investigated in previous literature. Orthostatic intolerance, a key feature of dysautonomia, has been reported to be more prevalent in patients with hEDS and HSD compared with control groups, indicating a higher incidence of autonomic dysfunction in these populations.9 In 1 study, 77.6% of patients with hEDS reported palpitations and 44.8% reported syncope,10 further emphasizing the need for a thorough understanding of the cardiac symptoms in these patients. However, there are limited studies on the prevalence of presyncope and palpitations in patients with HSD.
Our study further investigated differences in the prevalence of these symptoms between patients with hEDS and HDS. Cohort sizes in this population remain limited, with much of the literature including cohort studies of less than 100 adult patients, signifying the need for more insight into clinical characteristics and management in this population.10,11 Among the hEDS and HSD groups, we found a significantly greater prevalence of POTS in the hEDS group when compared with the HSD group (P = .016; Table 1). In contrast, palpitations, presyncope, and syncope were relatively common in both the hEDS and HSD groups, suggesting commonality of symptoms but non-discrimination across hypermobile phenotypes. These findings support the concept that autonomic dysfunction may represent a distinguishing cardiovascular feature of EDS, rather than a generalized increase in symptom burden. While prior studies have demonstrated a high prevalence of orthostatic symptoms and abnormalities on TTT in hypermobile populations,12 our results extend these observations by directly comparing hEDS and HSD, highlighting heterogeneity in dysautonomia across the hypermobility spectrum.
Age-stratified analysis further refines these relationships. There is currently limited data on long-term, age-related cardiac symptom progression in hypermobile patients, and the timing of symptom progression is unclear. It is important to note the significantly increased prevalence of presyncope within the older hEDS cohort when compared with the HSD cohort (Table 2). POTS was more frequently observed in younger patients with hEDS and HSD compared with their older counterparts (Table 3), suggesting that autonomic dysfunction may be more predominant earlier in the disease course. Importantly, TTT utilization did not differ significantly between groups or across age cohorts (Table 4) and was performed more frequently in patients with EDS compared with HSD. However, this difference was not significant, suggesting that referral bias is unlikely to fully explain the observed differences in the prevalence of POTS, although it cannot be excluded. Additionally, diagnostic modality did not differ significantly between groups, suggesting that differences in POTS prevalence are less likely to be attributable to variations in diagnostic approach (Table 4). Overall, the difference in the prevalence of cardiac symptoms between cohorts may suggest age-related vascular and autonomic changes in both patients with hEDS and patients with HSD (Figure 2).
This pilot study highlights the need for larger-scale studies to investigate differences in cardiac symptomatology across hypermobile populations. Our findings suggest that there may be an increased prevalence of POTS and presyncope/syncope in patients with hEDS. While previous studies have highlighted the prevalence of autonomic dysfunction in patients with EDS and HSD,10,11 there is a lack of specific screening and management guidelines in these populations. Despite increasing recognition of autonomic dysfunction, the 2017 American College of Cardiology, American Heart Association, and Heart Rhythm Society guidelines for the evaluation and management of syncope include recommendations for the general population but not specifically for hypermobile patients.13 Further investigation regarding the evaluation and management of syncope in hypermobile patients is warranted.
Limitations
This study is limited by its observational retrospective design, small sample size, and underrepresentation of male participants. EDS and HSD are relatively uncommon connective tissue diseases that can have an impact on patient recruitment. In addition, diagnostic distinction between hEDS and HSD remains challenging, given considerable phenotypic overlap. Misclassification or change of classification is possible, and physiological differences between these groups may be subtle. Our findings should be interpreted within this diagnostic context. Future studies should incorporate a prospective design with a larger patient population.
Conclusions
Patients with EDS and HSD often experience cardiac-related symptoms, including palpitations, presyncope, syncope, and POTS. While presyncope, syncope, and palpitations were highly prevalent in both groups, POTS was significantly more common in patients with hEDS. Additionally, POTS was observed more frequently in younger patients compared with older patients in both the hEDS and HSD groups.
Affiliations and Disclosures
Ermin Tale, DO1,2; Riya Kaushal, BA2; Bilal Niazi, BS2; Katie Nikishina, BA2; Amy Chen, MS2; Bernadette Riley, DO2; Todd J. Cohen,
From the 1Jacobi Medical Center/Albert Einstein College of Medicine, Department of Medicine, Bronx, New York; 2New York Institute of Technology College of Osteopathic Medicine, Department of Clinical Specialties, Old Westbury, New York.
The abstract forms of this publication have been presented and published in Heart Rhythm Society (https://www.heartrhythmjournal.com/article/S1547-5271(24)00752-5/fulltext) and the Journal of Osteopathic Medicine (https://www.degruyterbrill.com/document/doi/10.1515/jom-2025-2000/html).
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Address for correspondence: Todd Cohen, MD, New York Institute of Technology College of Osteopathic Medicine, 101 Northern Boulevard, P.O. Box 8000, Old Westbury, NY 11568. Email:
References
1. Blackburn PR, Xu Z, Tumelty KE, et al. Bi-allelic alterations in AEBP1 lead to defective collagen assembly and connective tissue structure resulting in a variant of Ehlers-Danlos syndrome. Am J Hum Genet. 2018;102(2):696-705. doi:10.1016/j.ajhg.2018.02.018
2. Aubry-Rozier B, Schwitzguebel A, Valerio F, et al. Are patients with hypermobile Ehlers–Danlos syndrome or hypermobility spectrum disorder so different? Rheumatol Int. 2021;41(10):1785-1794. doi:10.1007/s00296-021-04968-3
3. Hakim A. Hypermobile Ehlers-Danlos syndrome. 2004 Oct 22 [Updated 2024 Feb 22]. In: Adam MP, Bick S, Mirzaa GM, et al, eds. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2026. Accessed May 18, 2013.
4. Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet C Semin Med Genet. 2017;175(1):8-26. doi:10.1002/ajmg.c.31552
5. Sheldon RS, Grubb BP II, Olshansky B, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12(6):e41-e63. doi:10.1016/j.hrthm.2015.03.029
6. Goodman BP. Evaluation of postural tachycardia syndrome (POTS). Auton Neurosci. 2018;215:12-19. doi:10.1016/j.autneu.2018.04.004
7. Knight DRT, Bruno KA, Singh A, et al. Cardiac defects of hypermobile Ehlers-Danlos syndrome and hypermobility spectrum disorders: a retrospective cohort study. Front Cardiovasc Med. 2024;11:1332508. doi:10.3389/fcvm.2024.1332508
8. Rashed ER, Ruiz Maya T, Black J, et al. Cardiovascular manifestations of hypermobile Ehlers-Danlos syndrome and hypermobility spectrum disorders. Vasc Med. 2022 Jun;27(3):283-289. doi:10.1177/1358863X211067566
9. Peebles KC, Tan I, Butlin M, et al. The prevalence and impact of orthostatic intolerance in young women across the hypermobility spectrum. Am J Med Genet A. 2022;188(6):1761-1776. doi:10.1002/ajmg.a.62705
10. Pietri-Toro JM, Gardner OK, Leuchter JD, DiBartolomeo G, Hunter JA, Forghani I. Prevalence of cardiovascular manifestations in patients with hypermobile Ehlers-Danlos syndrome at the University of Miami. Am J Med Genet A. 2023;191(6):1502-1507. doi:10.1002/ajmg.a.63168
11. Kozyra M, Kostyun R, Strecker S. The prevalence of multisystem diagnoses among young patients with hypermobile Ehlers-Danlos syndrome and hypermobility spectrum disorder: a retrospective analysis using a large healthcare claims database. Medicine (Baltimore). 2024;103(41):e39212. doi:10.1097/MD.0000000000039212
12. Abu Orabi Z, Thompson SE, van Vliet J, Gee K, Roy A, Townend JN. Joint hypermobility: an under-recognised cause of palpitations, dizziness, and syncope in young females. J Clin Med. 2025;14(20):7373. doi:10.3390/jcm14207373
13. Shen WK, Sheldon RS, Benditt DG, et al. 017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2017;14(8):e155-e217. doi:10.1016/j.hrthm.2017.03.004