NALIRIFOX Compared With FOLFIRINOX for Treatment-Naive Metastatic Pancreatic Ductal Adenocarcinoma: An Overall Survival External Control Arm Study
Abstract
The pivotal NAPOLI 3 trial reported superior overall survival (OS) for NALIRIFOX (liposomal irinotecan+oxaliplatin + 5-fluorouracil/leucovorin) vs gemcitabine + nab-paclitaxel (GnP) in patients with treatment-naive metastatic pancreatic ductal adenocarcinoma (mPDAC) (P = .036). No prospective studies have compared first-line NALIRIFOX vs FOLFIRINOX (irinotecan + oxaliplatin + 5-fluorouracil/leucovorin) for mPDAC. Using patient-level data from NAPOLI 3 and the Flatiron Health electronic health record database, we compared OS for first-line NALIRIFOX in NAPOLI 3 to external control arms treated with FOLFIRINOX or modified FOLFIRINOX (mFOLFIRINOX). The mFOLFIRINOX cohorts were aligned to NAPOLI 3 eligibility criteria and statistically matched (inverse probability of treatment weighting [IPTW]) on key baseline characteristics. The NALIRIFOX cohort included 383 patients; the FOLFIRINOX and mFOLFIRINOX cohorts included 219 and 154 patients, respectively. Measurable baseline characteristics for the comparator cohorts were generally well balanced following IPTW adjustments. IPTW-adjusted OS (HR; 95% CI) favored NALIRIFOX vs FOLFIRINOX (0.79; 0.64-0.96; P = .02) and mFOLFIRINOX (0.80; 0.64-0.99; P = .04). Pending evidence from clinical trials or real-world prospective studies, this analysis provides preliminary insights into the comparative survival benefit of NALIRIFOX vs mFOLFIRINOX in patients with untreated mPDAC.
Introduction
Pancreatic cancer is the third leading cause of cancer-related death in the US.1 Pancreatic ductal adenocarcinoma (PDAC) accounts for more than 90% of pancreatic cancer cases2 and has a poor prognosis owing to its aggressive nature, nonspecific early symptoms, and limited treatment options; 5-year survival for patients with an advanced disease diagnosis is approximately 3%.1,3,4
Historically, FOLFIRINOX (irinotecan + oxaliplatin + 5-fluorouracil/leucovorin [5-FU/LV]) and gemcitabine in combination with nab-paclitaxel (GnP) were considered first-line (1L) standard-of-care treatment options for patients with metastatic PDAC (mPDAC). Regulatory approval of GnP was based on significant overall survival (OS) benefits in patients with mPDAC treated with GnP compared with gemcitabine monotherapy in the international phase 3 MPACT trial (hazard ratio [HR] for OS, 0.72; 95% CI, 0.62-0.83; P < .001).5-7
Conversely, FOLFIRINOX does not have regulatory approval for use as a 1L regimen for mPDAC; its widespread adoption in clinical practice followed demonstration of significant OS benefit compared with gemcitabine monotherapy in the French phase 3 PRODIGE trial in patients with newly diagnosed mPDAC (HR for OS, 0.57; 95% CI, 0.45-0.73; P < .001).8 Comparative clinical trial data for FOLFIRINOX and GnP are limited, and optimal treatment selection has been complicated by frequent tolerability-related modifications to the FOLFIRINOX dosing regimen (mFOLFIRINOX), the absence of a standard mFOLFIRINOX regimen, and inconsistent results from real-world studies comparing FOLFIRINOX and GnP.9-15 The phase 2 PASS-01 study, conducted to inform biomarker-driven treatment selection in treatment-naïve patients without germline pathogenic variants BRCA or PALB2, reported numerically longer progression-free survival with GnP compared with mFOLFIRINOX (5.3 vs 4.0 months, respectively; HR, 1.37; 95% CI, 0.97-1.92; P = .069). However, median OS for both treatment arms suggested limited survival benefit with either regimen for patients with advanced disease.16
In 2024, liposomal irinotecan was added to 1L mPDAC treatment armamentarium for use in combination with 5-FU/LV + oxaliplatin (NALIRIFOX) in adult patients. Regulatory approval of 1L NALIRIFOX was based on the phase 3 NAPOLI 3 trial, which reported clinically meaningful and statistically significant improvements in OS and progression-free survival with NALIRIFOX vs GnP (HR for OS, 0.83; 95% CI, 0.70-0.99; P = .036).17 However, clinically informed treatment selection remains limited by the lack of direct head-to-head trials of the comparative efficacy–safety profiles of these quadruplet, triplet, and doublet regimens.
The comparator arm for the NAPOLI 3 trial was decided based on regulatory approval for 1L mPDAC treatments; while FOLFIRINOX and mFOLFIRINOX are widely used based on clinical trial data,8,19 GnP has been the only regimen approved by the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA).6,7 Pending the availability of prospective real-world comparative data, we utilized a robust US electronic health record (EHR) database to create a proxy control arm for the NAPOLI 3 NALIRIFOX treatment arm. This included patients treated with 1L FOLFIRINOX in real-world clinical practice who met the eligibility criteria for NAPOLI 3. Using this external control arm (ECA), an FDA-endorsed methodology in the absence of relevant trial evidence,20 we aimed to provide preliminary insights into the comparative OS for 1L NALIRIFOX and FOLFIRINOX in patients matched on key baseline characteristics.
Materials and Methods
Data Sources
The NALIRIFIOX cohort for the present analysis was the intention-to-treat (ITT) population of the NALIRIFOX cohort from the NAPOLI 3 trial. NAPOLI 3 was a phase 3, open-label, randomized trial in previously untreated adults with mPDAC. A total of 770 participants were randomized 1:1 to receive NALIRIFOX (n = 383) or GnP (n = 387) between February 19, 2020, and August 17, 2021. The ITT population included all patients who were randomized to receive the NALIRIFOX regimen of liposomal irinotecan plus oxaliplatin plus 5-FU/LV. Participants were followed every 2 months after the end of treatment until death or trial closure.
The FOLFIRINOX ECA for the present analysis was constructed from the Flatiron Health EHR database, which contains deidentified, patient-level, longitudinal, demographically and geographically diverse data, representing more than 2.5 million patients with active cancer who were treated at over 280 cancer clinics and academic centers across the US. The dataset was derived from structured (eg, items directly captured in the EHR) and unstructured (eg, physician notes and pathology reports) data elements.
Study Design
The index date for the NALIRIFOX cohort was defined as the date of randomization during the trial; participants’ baseline characteristics and medical history were assessed during screening (ie, within 28 days prior to randomization). For the FOLFIRINOX cohort, the index date was defined as the initiation of 1L treatment with FOLFIRINOX for mPDAC. The baseline period was defined as the 12-month period prior to the index date.
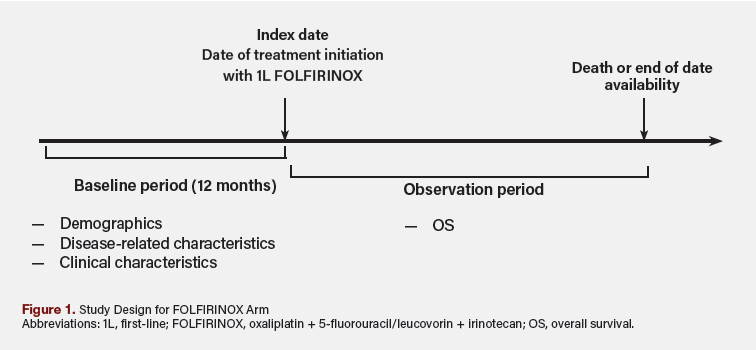 The observation period was defined as the duration from index date to the earliest of patient death or date of last confirmed activity based on the available data (ie, the last record of patient vitals, medication administrations, reported laboratory tests/results from structured data, or clinician confirmed receipt of treatments from unstructured data). An overview of the study design for the FOLFIRINOX cohort is shown in Figure 1.
The observation period was defined as the duration from index date to the earliest of patient death or date of last confirmed activity based on the available data (ie, the last record of patient vitals, medication administrations, reported laboratory tests/results from structured data, or clinician confirmed receipt of treatments from unstructured data). An overview of the study design for the FOLFIRINOX cohort is shown in Figure 1.
Study Population and Cohorts
To align the FOLFIRINOX cohort to the NALIRIFOX arm cohort, patients with mPDAC treated with 1L FOLFIRINOX were eligible if they initiated treatment between January 1, 2020, and July 31, 2022 (ie, during the NAPOLI 3 study period) and met key NAPOLI 3 eligibility criteria within the EHR data (Table S1).
Once constructed, a subgroup of the FOLFIRINOX cohort was identified that comprised patients who received a modified FOLFIRINOX (mFOLFIRINOX) regimen, defined as an initial dose of irinotecan of 150 mg/m2 or less or initial cumulative dose (bolus + infusion) of 5-FU of 2720 mg/m2 or less during the first cycle (within 30 days of the index date; Table S2, Figure S1). Sensitivity analyses were conducted to determine the impact on OS of application of NAPOLI 3 eligibility criteria to patients treated in real-world practice, and of aligning the real-world treatment window to that of the NAPOLI 3 study period: the first sensitivity analysis (the “all-comer cohort”) comprised all adults in the Flatiron Health EHR database with mPDAC who were treated with 1L FOLFIRINOX on or after January 1, 2014 (ie, start of data availability); the second sensitivity analysis (the “all-comer contemporary cohort”) comprised the subset of the all-comer cohort who were treated between January 1, 2020, and July 31, 2022.
Outcomes
Overall survival during the study period was assessed for all study cohorts and defined as the time from index date to death from any cause. Patients who did not experience an event during the study period were censored at the last known time that they were alive (for the NALIRIFOX cohort) or at their last confirmed activity date (for the mFOLFIRINOX cohorts).
No safety outcome evaluations were planned because of anticipated inconsistent documentation of adverse events in real-world practice, which would prevent meaningful comparison with per-protocol safety data collected during the NAPOLI 3 trial.
Statistical Analysis
Summary statistics were reported for baseline patient demographic, clinical, and disease-related characteristics, including frequencies and percentages for categorical variables and means, standard deviations, medians, and interquartile ranges for continuous variables. Differences between cohorts were calculated using standardized differences. Inverse probability of treatment weighting (IPTW) was used to control for potential confounding and reduce non-comparability between the 2 cohorts. IPTW is an established method used in observational studies where randomization is not feasible.21 The IPTW approach aims to “simulate” a randomized controlled trial where the distribution of measured baseline covariates is similar between treatment cohorts. Causal analysis methods, including IPTW, are recommended in FDA guidance as a means of addressing confounding and improving the rigor of non-randomized comparative analyses.21 Through IPTW, stabilized weights were calculated based on the estimated propensity scores. Propensity scores were derived using a logistic regression with NALIRIFOX treatment as the dependent variable and a set of measured baseline covariants that were clinically or statistically relevant (ie, variables showing imbalance between cohorts based on a standardized difference ≥10%) as independent variables: age category at index, sex, race, metastatic status at initial diagnosis of PDAC, European Cooperative Oncology Group (ECOG) performance status, surgery history, chemotherapy history, site of primary disease, time from metastatic diagnosis to index date, comorbidities (hypertension, thrombosis, ascites), and laboratory measures (alanine transaminase, creatinine clearance, platelet count, serum albumin, serum total bilirubin).
Median OS and corresponding 95% CI were calculated by Kaplan-Meier analysis for the unweighted and weighted NALIRIFOX and FOLFIRINOX cohorts, separately. Hazard ratios and 95% CI were estimated by Cox proportional hazards modeling with a robust variance estimator (level was set at 0.05).
Ethics
For the NAPOLI 3 trial data, anonymization and assessment of the risk of reidentification of trial participants was performed by a third party to protect clinical datasets against adversary attacks in compliance with EMA Policy 0070. The Flatiron Health EHR data were deidentified and complied with the Health Insurance Portability and Accountability Act (HIPAA). Therefore, ethical approval was not required.
Results
Patient Characteristics
In total, the NALIRIFOX cohort included the 383 patients from the NAPOLI 3 ITT arm, and the FOLFIRINOX cohort included 219 patients treated in real-world practice who met the key NAPOLI 3 eligibility criteria for the present analysis. Of the 219 patients in the overall FOLFIRINOX cohort, 70.3% (n = 154) received a regimen that met the study definition of mFOLFIRINOX and were included in the mFOLFIRINOX cohort (Figure S1). For the OS sensitivity analyses, the all-comer cohort included 3271 patients, and the contemporary all-comer cohort included 1000 patients.
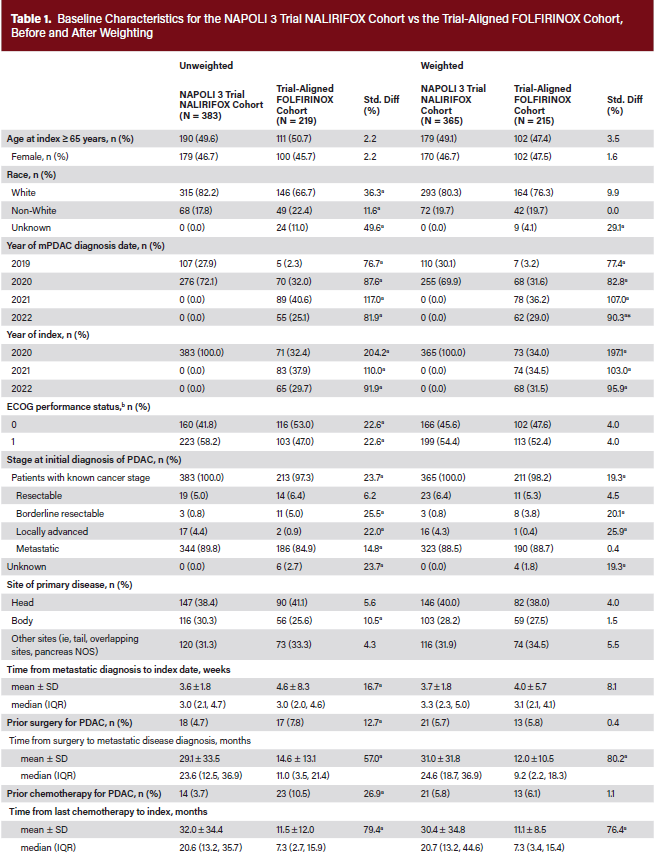
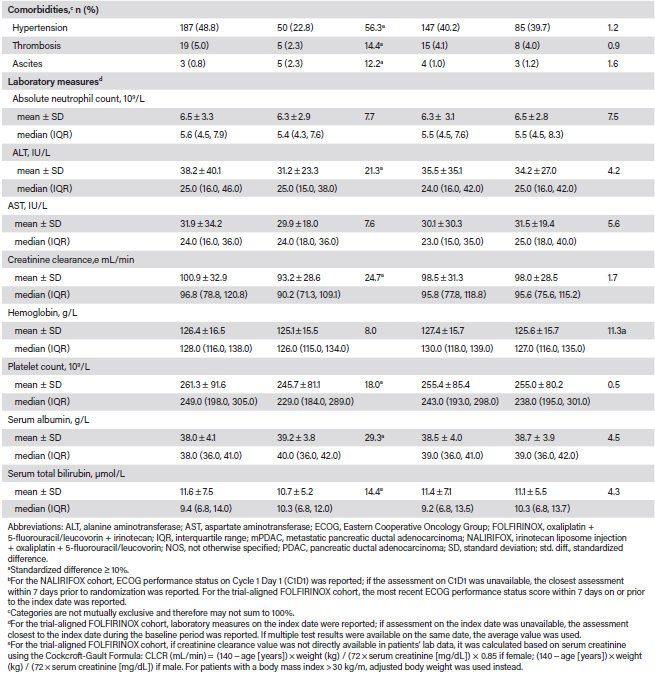
The characteristics of patients in the NALIRIFOX and FOLFIRINOX cohorts, before and after weighting, are detailed in Table 1. Prior to weighting, a higher proportion of patients in the NALIRIFOX cohort than the FOLFIRINOX cohort had ECOG performance status of 1 at baseline (58.2% vs 47.0%; standardized difference, 22.6%). Most patients in both cohorts had metastatic disease at diagnosis. The mean (± SD) time from metastatic diagnosis to index date was 3.6 ± 1.8 months in the NALIRIFOX cohort, and 4.6 ± 8.3 months in the FOLFIRINOX cohort (standardized difference, 16.7%). A greater proportion of patients in the NALIRIFOX cohort than the FOLFIRINOX cohort had primary disease in the body of the pancreas (30.3% vs 25.6%; standardized difference, 10.5%). Regarding prior treatments, approximately 5% of patients in the NALIRIFOX cohort and 8% in the FOLFIRINOX cohort had undergone surgery; 4% and 11%, respectively, had received prior chemotherapy for PDAC. Imbalances between the cohorts were also observed for various laboratory measures. After applying IPTW, key characteristics were generally well balanced between the NALIRIFOX cohort and the comparator FOLFIRINOX (Table 1), mFOLFIRINOX (Table S3), all-comer sensitivity (Table S4), and contemporary all-comer sensitivity (Table S5) cohorts.
Comparison of OS
The median duration of follow-up was 10.9 months for the NALIRIFOX cohort and 8.9 months for the FOLFIRINOX cohort. Following IPTW, median (95% CI) OS was longer for the NALIRIFOX cohort compared with the FOLFIRINOX cohort (11.7; 10.5-13.0) months vs 9.0 (7.3-11.1) months, respectively). The analysis demonstrated a statistically significant OS benefit favoring NALIRIFOX compared with FOLFIRINOX (HR, 0.79; 95% CI, 0.64-0.96; P = .02) (Table 2, Figure 2).
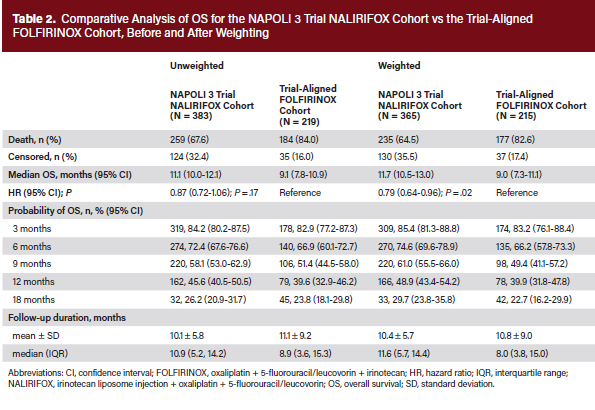
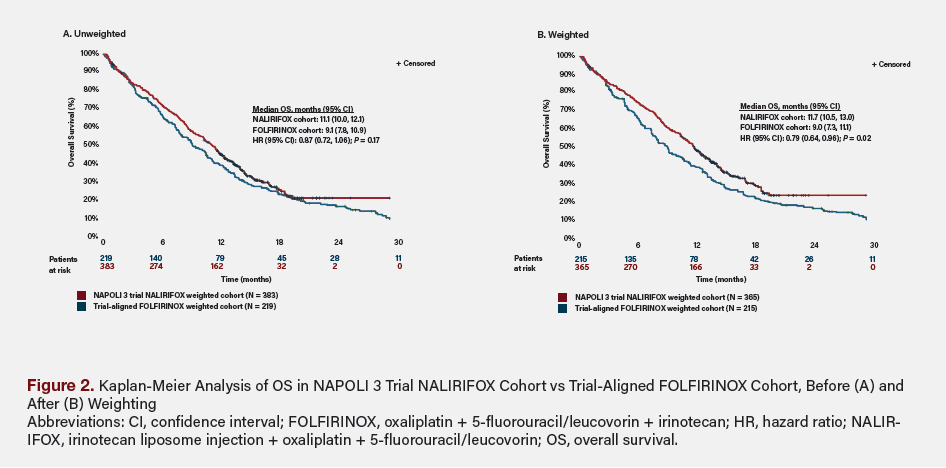 Subgroup and sensitivity analyses results were consistent with the main analysis. Median (95% CI) OS was longer for the weighted NALIRIFOX cohort (11.5; 10.3-12.5 months) than for the IPTW-adjusted mFOLFIRINOX cohort (8.0; 6.7-11.2 months) (HR, 0.80; 95% CI, 0.64-0.99; P = .04).
Subgroup and sensitivity analyses results were consistent with the main analysis. Median (95% CI) OS was longer for the weighted NALIRIFOX cohort (11.5; 10.3-12.5 months) than for the IPTW-adjusted mFOLFIRINOX cohort (8.0; 6.7-11.2 months) (HR, 0.80; 95% CI, 0.64-0.99; P = .04).
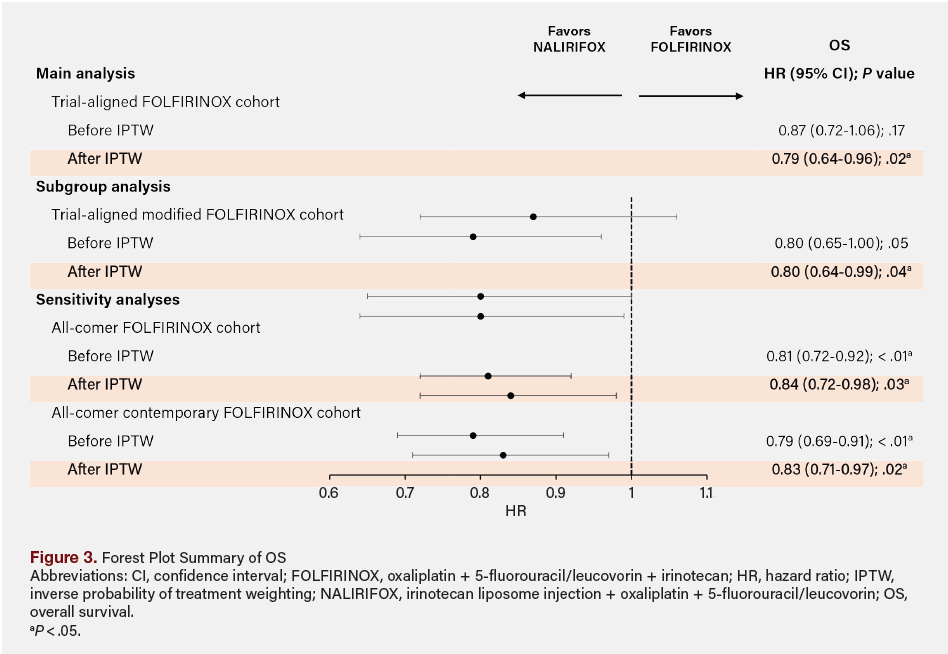 Similarly, sensitivity analyses comparing OS for the NALIRIFOX cohort with that for the FOLFIRINOX all-comer and all-comer contemporary cohorts yielded similar results to those of the main analysis. Overall, treatment with NALIRIFOX was associated with a statistically significant reduction in the hazard of death compared with FOLFIRINOX (main analysis, 21% reduction; sensitivity analyses, 16%-17% reduction) and compared with mFOLFIRINOX (20% reduction) (Figure 3).
Similarly, sensitivity analyses comparing OS for the NALIRIFOX cohort with that for the FOLFIRINOX all-comer and all-comer contemporary cohorts yielded similar results to those of the main analysis. Overall, treatment with NALIRIFOX was associated with a statistically significant reduction in the hazard of death compared with FOLFIRINOX (main analysis, 21% reduction; sensitivity analyses, 16%-17% reduction) and compared with mFOLFIRINOX (20% reduction) (Figure 3).
Discussion
The lack of direct comparative evidence for NALIRIFOX vs FOLFIRINOX from clinical trials or prospective real-world studies has created an important evidence gap for the management of previously untreated mPDAC. A meta-analysis of 7 clinical trials concluded that 1L NALIRIFOX and FOLFIRINOX may provide comparable efficacy and that both offer superior survival benefits over GnP.22
In the absence of prospective comparative study data, we constructed an ECA of patients who received full or modified FOLFIRINOX treatment in real-world clinical practice and who met key eligibility criteria for NAPOLI 3. We further aligned the real-world mFOLFIRINOX cohorts to the NAPOLI 3 NALIRIFOX cohort using statistical weighting to adjust for residual differences in the distribution of key baseline characteristics. After weighting, NALIRIFOX was associated with a statistically significant reduction in the probability of death compared with FOLFIRINOX (21% reduction) and compared with mFOLFIRINOX (20% reduction).
In the present analysis, mFOLFIRINOX accounted for approximately 70% of patients in the FOLFIRINOX cohort, suggesting that modification of FOLFIRINOX is common in real-world clinical practice. This finding aligns with existing evidence that mFOLFIRINOX may be better tolerated and preferred over the traditional regimen.19,23 A 2018 meta-analysis of 11 studies evaluating the efficacy of mFOLFIRINOX indicated that it had similar survival benefits but lower toxicity than full FOLFIRINOX.23 In the present analysis, NALIRIFOX consistently demonstrated improved OS compared with mFOLFIRINOX. However, it should be noted that the reasons for modifying FOLFIRINOX were not captured in this study and that, in the absence of a standardized definition of mFOLFIRINOX dosing, the definition used for the present analysis may not capture all modifications likely used in clinical practice.
The OS benefits observed for NALIRIFOX in the present analysis may, in part, reflect a key pharmacologic difference between the NALIRIFOX and mFOLFIRINOX regimens, specifically the inclusion of the liposomal vs non-liposomal irinotecan, respectively. Compared with the non-liposomal formulation, liposomal irinotecan has a longer half-life, and the liposomal encapsulation improves drug retention and tumor exposure, with potential benefits for enhanced efficacy.24-26
The results of this study should be interpreted while considering certain limitations. It is important to acknowledge that, although the baseline characteristics of the cohorts were aligned through application of key NAPOLI 3 eligibility criteria to all cohorts and subsequent statistical adjustments/weighting, some residual differences may have remained. In addition, it was not feasible to apply all NAPOLI 3 eligibility criteria to individuals in the mFOLFIRINOX cohorts owing to variability in data recorded in real-world practice. It is also relevant to note that heterogeneity in completeness of EHR data recording may have introduced selection bias owing to patients with insufficient data being unevaluable for inclusion in the mFOLFIRINOX study cohorts. Furthermore, comorbidities and concomitant medication use that existed prior to mPDAC diagnosis (or that are unrelated to the mPDAC treatment) may not be completely captured in the Flatiron Health EHR database, resulting in incomplete characterization (and potential under-representation) of the full comorbidity burden of the real-world mFOLFIRINOX cohorts. Further, the missingness or under-representation of certain variables that influence treatment selection or study endpoints may lead to potential residual or unmeasured confounding. However, key confounders were well balanced following IPTW adjustments, including ECOG performance score, which is recognized as an important prognostic factor in mPDAC populations.27,28
Of additional note are the minor differences in the defined index dates for the cohorts: date of randomization for the NALIRIFOX; date of first FOLFIRINOX administration for the mFOLFIRINOX. Although the differences in index date definitions introduced potential for bias among the cohorts, the mean time between date randomization and the date of first NALIRIFOX administration in NAPOLI 3 was only 3 days, resulting in minimal potential impact that was likely insufficient to account for the observed difference in OS between the cohorts. Another factor to consider is that the NALIRIFOX cohort comprised all randomized patients in the NAPOLI 3 ITT population, including 13 patients (3.4%) who did not receive any dose of the NALIRIFOX regimen. In contrast, all patients in the mFOLFIRINOX cohorts received at least 1 treatment cycle. This difference may underestimate the clinical benefit of the NALIRIFOX regimen.
Another consideration is the regional difference between the 2 study cohorts; the Flatiron Health EHR includes data from US clinics; in contrast, NAPOLI 3 was a global trial. However, findings from NAPOLI 3 indicate that median OS was comparable across regional subgroups (North America, 11.2 months; rest of the world, 11.1 months), suggesting that regional differences are unlikely to bias the results meaningfully.17 Nonetheless, future analyses that include a balanced distribution of regions may help further evaluate this potential source of bias.
Lastly, because this study focused on a specific population of patients with mPDAC treated with 1L FOLFIRINOX who met the inclusion/exclusion criteria of NAPOLI 3, the findings of the main analysis may not be generalizable to the broader mPDAC population receiving 1L mFOLFIRINOX regimens. However, sensitivity analyses involving the all-comer and contemporary all-comer FOLFIRINOX cohorts were consistent with the main analysis, providing reassurance as to the robustness and generalizability of the results.
Conclusion
In the absence of head-to-head data from clinical trials, this ECA study demonstrated that patients with mPDAC who were statistically matched on baseline characteristics survived for significantly longer on 1L NALIRIFOX treatment than on 1L mFOLFIRINOX. Acknowledging inherent differences in ecology of care between clinical trials and real-world clinical practice, these results provide preliminary insights into the comparative efficacy/effectiveness of 1L NALIRIFOX and mFOLFIRINOX in patients with mPDAC, paving the way for more conclusive findings from prospective real-world studies.
This article has supplementary material, which can be accessed here.
Key Clinical Summary
- NALIRIFOX demonstrated significantly improved OS vs FOLFIRINOX and modified FOLFIRINOX in treatment-naive metastatic PDAC using an external control arm with IPTW adjustment (HR ~0.79-0.80).
- Median OS was longer with NALIRIFOX (≈11.5-11.7 months) compared with FOLFIRINOX (≈9.0 months) and mFOLFIRINOX (≈8.0 months).
- Findings address the lack of head-to-head trials, supporting NALIRIFOX as a potentially more effective first-line option, though confirmation in prospective studies is needed.
Clinical Pathway Category: Treatment
This study informs the treatment domain of clinical pathways by providing comparative, evidence-based insights suggesting that first-line NALIRIFOX may offer superior overall survival vs FOLFIRINOX in metastatic PDAC, addressing a key gap where head-to-head trial data are lacking. By leveraging robust statistical adjustment of real-world and trial data, it aligns with evidence-based standards and supports pathway optimization toward more effective, data-driven regimen selection to improve oncology care delivery.
Author Information
Authors: Daniel A. King (MD, PhDw)1; Mei Sheng Duh (ScD)2; Rose Chang (ScD)2; Louise Yu (MS)2; Chunyi Xu (MS)2; Eric M. Maiese (PhD)3*; George Kim (MD)4
Affiliations:
1Northwell Health, New Hyde Park, NY; 2Analysis Group, Inc, Boston, MA; 3Ipsen Biopharmaceuticals, Inc, Cambridge, MA; 4Division of Hematology and Oncology, University of Florida, Gainesville, FL
*At the time the work was conducted.
Address correspondence to:
Daniel A. King, MD, PhD
Northwell Health, 450 Lakeville Road, New Hyde Park, NY 11042
Email: dking14@northwell.edu
Phone: +1 516-734-8900
Funding: This study was funded by Ipsen.
Disclosures: E.M. was an employee of Ipsen Biopharmaceuticals, Inc, at the time the work was conducted and holds stock/options for Ipsen Biopharmaceuticals, Inc. M.S.D., R.C., L.Y., and C.X. are employees of Analysis Group, Inc., which has received consulting fees from Ipsen Biopharmaceuticals, Inc. for the conduct of this study. D.K. has served on the advisory board for Ipsen and AstraZeneca and received honoraria from ClearView, The Kinetix Group, and Omni Health Media.
Author contributions: Mei Sheng Duh, Rose Chang, Louise Yu, Chunyi Xu, and Eric M. Maiese contributed to the conception and design of the study. Daniel A. King, Mei Sheng Duh, Rose Chang, Louise Yu, Chunyi Xu, Eric M. Maiese, and George Kim contributed to the analysis and/or interpretation of the data. Daniel A. King, Mei Sheng Duh, Rose Chang, Louise Yu, Chunyi Xu, Eric M. Maiese, and George Kim contributed to the writing of this manuscript.
Data availability: Restrictions apply to the availability of these data since the data underlying this publication were provided by Flatiron Health Inc under contract to Ipsen.
Acknowledgments: Medical writing was provided by Shelley Batts, PhD, an independent contractor of Analysis Group, Inc, and medical writing support by Alison Chisholm, MPH, of Oxford PharmaGenesis, Oxford, UK, which was sponsored by Ipsen in accordance with Good Publication Practice guidelines (GPP 2022).
References
1. Cancer stat facts: pancreatic cancer. National Cancer Institute. Accessed March 3, 2025. https://seer.cancer.gov/statfacts/html/pancreas.html
2. Kleeff J, Korc M, Apte M, et al. Pancreatic cancer. Nat Rev Dis Primers. 2016;2:16022. doi:10.1038/nrdp.2016.22
3. Lambert A, Schwarz L, Borbath I, et al. An update on treatment options for pancreatic adenocarcinoma. Ther Adv Med Oncol. 2019;11:1758835919875568. doi:10.1177/175883591987556
4. Martinez-Bosch N, Vinaixa J, Navarro P. Immune evasion in pancreatic cancer: from mechanisms to therapy. Cancers. 2018;10(1):6. doi:10.3390/cancers10010006
5. Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369(18):1691-1703. doi:10.1056/NEJMoa1304369
6. Abraxane. Prescribing information. Bristol Myers Squibb; 2022. Accessed April 3, 2026. https://packageinserts.bms.com/pi/pi_abraxane.pdf
7. Pazenir. Package insert. Teva; 2022. Accessed October 3, 2025. https://www.ema.europa.eu/en/documents/product-information/pazenir-epar-product-information_en.pdf
8. Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364(19):1817-1825. doi:10.1056/NEJMoa1011923
9. Braiteh F, Patel MB, Parisi M, Ni Q, Park S, Faria C. Comparative effectiveness and resource utilization of nab-paclitaxel plus gemcitabine vs FOLFIRINOX or gemcitabine for the first-line treatment of metastatic pancreatic adenocarcinoma in a US community setting. Cancer Manag Res. 2017;9:141-48. doi:10.2147/cmar.S126073
10. Cartwright TH, Parisi M, Espirito JL, et al. Clinical outcomes with first-line chemotherapy in a large retrospective study of patients with metastatic pancreatic cancer treated in a US community oncology setting. Drugs Real World Outcomes. 2018;5(3):149-159. doi:10.1007/s40801-018-0137-x
11. Kim S, Signorovitch JE, Yang H, et al. Comparative effectiveness of nab-paclitaxel plus gemcitabine vs FOLFIRINOX in metastatic pancreatic cancer: a retrospective nationwide chart review in the United States. Adv Ther. 2018;35(10):1564-1577. doi:10.1007/s12325-018-0784-z
12. Kim GP, Parisi MF, Patel MB, Pelletier CL, Belk KW. Comparison of treatment patterns, resource utilization, and cost of care in patients with metastatic pancreatic cancer treated with first-line nab-paclitaxel plus gemcitabine or FOLFIRINOX. Expert Rev Clin Pharmacol. 2017;10(5):559-565. doi:10.1080/17512433.2017.1302330
13. Patel T, Miccio J, Cecchini M, et al. Clinical outcomes of first line FOLFIRINOX vs. gemcitabine plus nab-paclitaxel in metastatic pancreatic cancer at the Yale Smilow Hospital System. J Gastrointest Oncol. 2021;12(6):2547-2556. doi:10.21037/jgo-21-202
14. Chan KKW, Guo H, Cheng S, et al. Real-world outcomes of FOLFIRINOX vs gemcitabine and nab-paclitaxel in advanced pancreatic cancer: a population-based propensity score-weighted analysis. Cancer Med. 2020;9(1):160-169. doi:10.1002/cam4.2705
15. Sigel K, Zhou M, Park YA, et al. Gemcitabine plus nab-paclitaxel versus FOLFIRINOX for unresected pancreatic cancer: comparative effectiveness and evaluation of tumor growth in veterans. Semin Oncol. 2021;48(1):69-75. doi:10.1053/j.seminoncol.2021.02.001
16. Knox JJ, O'Kane G, King D, et al. PASS-01: Randomized phase II trial of modified FOLFIRINOX versus gemcitabine/nab-paclitaxel and molecular correlatives for previously untreated metastatic pancreatic cancer. J Clin Oncol. 2025;43(31):3355-3368. doi:10.1200/jco-25-00436
17. Wainberg ZA, Melisi D, Macarulla T, et al. NALIRIFOXversus nab-paclitaxel and gemcitabine in treatment-naive patients with metastatic pancreatic ductal adenocarcinoma (NAPOLI 3): a randomised, open-label, phase 3 trial. Lancet. 2023;402(10409):1272-1281. doi:10.1016/S0140-6736(23)01366-1
18. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Pancreatic Adenocarcinoma V.2.2025. Accessed February 3, 2025. http://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf
19. Stein SM, James ES, Deng Y, et al. Final analysis of a phase II study of modified FOLFIRINOX in locally advanced and metastatic pancreatic cancer. Br J Cancer. 2016;114(7):737-743. doi:10.1038/bjc.2016.45
20. US Food and Drug Administration. considerations for the design and conduct of externally controlled trials for drug and biological products. February 2023. Accessed March 14, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/considerations-design-and-conduct-externally-controlled-trials-drug-and-biological-products
21. US Food and Drug Administration. Adjusting for covariates in randomized clinical trials for drugs and biological products. May 2023. Accessed March 14, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/adjusting-covariates-randomized-clinical-trials-drugs-and-biological-products
22. Nichetti F, Rota S, Ambrosini P, et al. NALIRIFOX, FOLFIRINOX, and gemcitabine with nab-paclitaxel as first-line chemotherapy for metastatic pancreatic cancer: a systematic review and meta-analysis. JAMA Netw Open. 2024;7(1):e2350756. doi:10.1001/jamanetworkopen.2023.50756
23. Tong H, Fan Z, Liu B, Lu T. The benefits of modified FOLFIRINOX for advanced pancreatic cancer and its induced adverse events: a systematic review and meta-analysis. Sci Rep. 2018;8(1):8666. doi:10.1038/s41598-018-26811-9
24. Drummond DC, Noble CO, Guo Z, Hong K, Park JW, Kirpotin DB. Development of a highly active nanoliposomal irinotecan using a novel intraliposomal stabilization strategy. Cancer Res. 2006;66(6):3271-3277. doi:10.1158/0008-5472.Can-05-4007
25. Brendel K, Bekaii-Saab T, Boland PM, et al. Population pharmacokinetics of liposomal irinotecan in patients with cancer and exposure-safety analyses in patients with metastatic pancreatic cancer. CPT Pharmacometrics Syst Pharmacol. 2021;10(12):1550-1563. doi:10.1002/psp4.12725
26. Adiwijaya BS, Kim J, Lang I, et al. Population pharmacokinetics of liposomal irinotecan in patients with cancer. Clin Pharmacol Ther. 2017;102(6):997-1005. doi:10.1002/cpt.720
27. Colloca G. Performance status as prognostic factor in phase III trials of first-line chemotherapy of unresectable or metastatic pancreatic cancer: a trial-level meta-analysis. Asia Pac J Clin Oncol. 2022;18(3):232-239. doi:10.1111/ajco.13598
28. Yu KH, Ozer M, Cockrum P, Surinach A, Wang S, Chu BC. Real-world prognostic factors for survival among treated patients with metastatic pancreatic ductal adenocarcinoma. Cancer Med. 2021;10(24):8934-8943. doi:10.1002/cam4.4415













