Differential Effect of Chronic Kidney Disease on Right-Sided Extravalvular Damage in Low-Flow, Low-Gradient Aortic Stenosis
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/26.00078. Epub May 14, 2026.
Abstract
Objectives. In the transcatheter aortic valve replacement (TAVR) era, the presence of low-flow, low-gradient aortic stenosis (LFLG AS) itself does not preclude intervention. However, chronic kidney disease (CKD) and right-sided extravalvular damage (RSED) complicate procedural risk assessment. Both their association with each other and their joint impact on post-TAVR mortality in LFLG AS and across its subtypes remain incompletely characterized.
Methods. The authors performed a retrospective study of patients with LFLG AS who underwent TAVR between 2019 and 2022, stratified by CKD stage (from no CKD to end-stage renal disease [ESRD]) and LFLG AS subtype (classical vs paradoxical). RSED was defined by greater than or equal to moderate pulmonary hypertension, greater than or equal to moderate tricuspid regurgitation, or right ventricular (RV) systolic dysfunction. Poisson regression models assessed associations between CKD stage and RSED, while multivariable Cox regression models and Wald tests compared mortality across cohorts.
Results. Among 402 patients (137 classical, 265 paradoxical), ESRD was independently associated with RSED and RV dysfunction, whereas earlier CKD stages were not. ESRD was associated with increased RV dysfunction in paradoxical LFLG AS but not in classical LFLG AS. Advanced CKD and RSED were each associated with increased post-TAVR mortality. In paradoxical LFLG AS, combined RSED and advanced CKD were associated with significantly higher mortality than either condition alone. This effect was not observed in classical LFLG AS patients.
Conclusions. Advanced renal dysfunction is associated with RSED in LFLG AS. In paradoxical LFLG AS, their combination has a possibly multiplicative association with mortality.
Introduction
In the modern transcatheter aortic valve replacement (TAVR) era, the presence of low-flow, low-gradient aortic stenosis (LFLG AS) itself does not preclude intervention.1 Nevertheless, patients with LFLG AS represent a higher risk population for whom careful risk stratification is necessary. Its 2 subtypes, classical LFLG (cLFLG; left ventricular ejection fraction [LVEF] <50%) and paradoxical LFLG AS (pLFLG; LVEF ≥50%), add further clinical complexity by virtue of their distinct pathophysiologies2 and, consequently, their differences in post-TAVR outcomes.3 Beyond left-sided hemodynamics, outcomes in LFLG AS may be critically influenced by systemic and right-sided factors that interact in a reciprocal and potentially self-perpetuating manner.
Among these, chronic kidney disease (CKD) and right-sided extravalvular damage (RSED) represent 2 interrelated determinants of prognosis. CKD is highly prevalent in patients with AS and has been associated with adverse myocardial remodeling, increased post-TAVR mortality, and worse outcomes in low-flow severe AS.4-7 Conversely, markers of RSED (namely, pulmonary hypertension [pHTN], significant tricuspid regurgitation [TR], and right ventricular [RV] dysfunction) are also independently associated with increased mortality after TAVR.8-13 Importantly, these processes are biologically intertwined: right-sided heart failure and RSED contribute to worsening renal function through venous congestion and impaired renal perfusion,14-17while CKD, in turn, worsens cardiac remodeling in heart failure in a reciprocal fashion.18 CKD and RSED may each therefore promote the development of the other in a deadly spiral, with important implications for patient risk stratification.
The purpose of this study is to examine the association between CKD and RSED within LFLG AS and across each stage of CKD. It also explores specifically the association between CKD and RV dysfunction for each of the LFLG AS subtypes, as differences between them may shed further light upon their distinct pathophysiologies.
Methods
Data collection
This retrospective study was approved by the institutional review board and utilized data from our center from January 2019 to December 2022. Clinical and echocardiographic data were reviewed and analyzed. Informed consent was waived given the retrospective nature of the study.
The investigation included patients with echocardiographic parameters consistent with severe LFLG AS (aortic valve area [AVA] ≤1.0 cm², stroke volume index ≤35 mL/m², and mean gradient <40 mm Hg) who ultimately underwent TAVR. RV dysfunction was determined from pre-TAVR echocardiography as a tricuspid annular plane systolic excursion (TAPSE) of less than 1.7 cm, tissue doppler S’ velocity of less than 9.5 cm/s, or RV fractional area change of less than 35%. Patients were determined to have RSED if they had at least moderate pHTN, at least moderate TR, or RV dysfunction, based on Généreux et al’s staging classification of AS.19 Our study defined moderate pHTN as a mean pulmonary artery pressure of greater than or equal to 40 mm Hg, a hemodynamically equivalent and physiologically more accurate metric for Généreux et al’s pHTN criteria of a systolic pulmonary artery pressure threshold of greater than or equal to 60 mm Hg.15,20 For patients who lacked right heart catheterization data up to 5 years before their TAVR dates, the systolic threshold of 60 mm Hg was used based on echocardiographic data.
Group stratification
Patients were stratified by LFLG subtype (cLFLG AS vs pLFLG AS) and by renal function: no CKD (glomerular filtration rate [GFR] > 90, no proteinuria), CKD 1-2 (GFR 90+ with proteinuria and GFR 60-90 with or without proteinuria), CKD 3a/3b (GFR 30-60), CKD 4-5 (GFR < 30 not on hemodialysis [HD]), and end-stage renal disease (ESRD) (on HD). The primary outcome was the composite measure of RSED, composed of at least moderate pHTN, at least moderate TR, and evidence of RV dysfunction on transthoracic echocardiogram. Additionally, the outcome of RV dysfunction was measured across each of the LFLG AS subtypes. Data on all-cause mortality (hereafter referred to as mortality) were collected up to January 2025.
Statistical analysis
Categorical variables are presented as counts and percentages and were compared across CKD stages using chi-squared tests. Comparisons between LFLG AS subtypes were performed using chi-squared tests or Fisher’s exact tests when expected cell counts were less than 5. Continuous variables are presented as medians with interquartile ranges and were compared across CKD stages using Kruskal-Wallis tests and between LFLG AS subtypes using Mann-Whitney U tests. The relative risk of having developed RSED and RV dysfunction at each stage of CKD was evaluated using Poisson regression models, adjusted for age, sex, coronary artery disease (CAD) requiring revascularization, prior stroke, and LFLG subtype. For each CKD stage, the relative risk of each of the outcomes was calculated in comparison to the patients without CKD.
Time-to-event analyses were performed using Kaplan-Meier methods, with mortality as the primary outcome. Survival was compared first across each of the CKD stages, then among those with neither CKD 3+ nor RSED or RV dysfunction, those either with CKD 3+ alone or with RSED or RV dysfunction alone, and those with both CKD 3+ and RSED or RV dysfunction. A global log-rank test was first performed, followed by multivariable Cox regression to estimate adjusted hazard ratios (HRs) and 95% confidence intervals (CIs). The first analysis used the group without CKD as the reference; the second analysis, the group with neither CKD 3+ nor RSED or RV dysfunction. Models were adjusted for age, sex, CAD requiring revascularization, prior stroke, and LFLG subtype. Wald tests were used to compare mortality risk between specific groups. Kaplan-Meier curves and number-at-risk tables were truncated at 40 months for visualization, but all statistical analyses used the full duration of follow-up.
All statistical tests were 2-sided, and a P-value of less than 0.05 was considered statistically significant. Statistical analyses were performed using Python (SciPy, lifelines, statsmodels, NumPy, and pandas libraries).
Results
Baseline characteristics
Four hundred-two patients met the inclusion criteria of our study; 176 (43.8%) had right heart catheterization data available within 5 years of their TAVR dates. Clinical and echocardiographic baseline characteristics as stratified by renal function and LFLG AS subtypes are summarized in Tables 1 and 2, respectively. The CKD 3a/3b, CKD 4-5, and ESRD groups had higher proportions of patients with congestive heart failure (CHF) and prior coronary artery bypass grafting (CABG), myocardial infarction (MI), and cerebrovascular accidents (CVA) (Table 1).
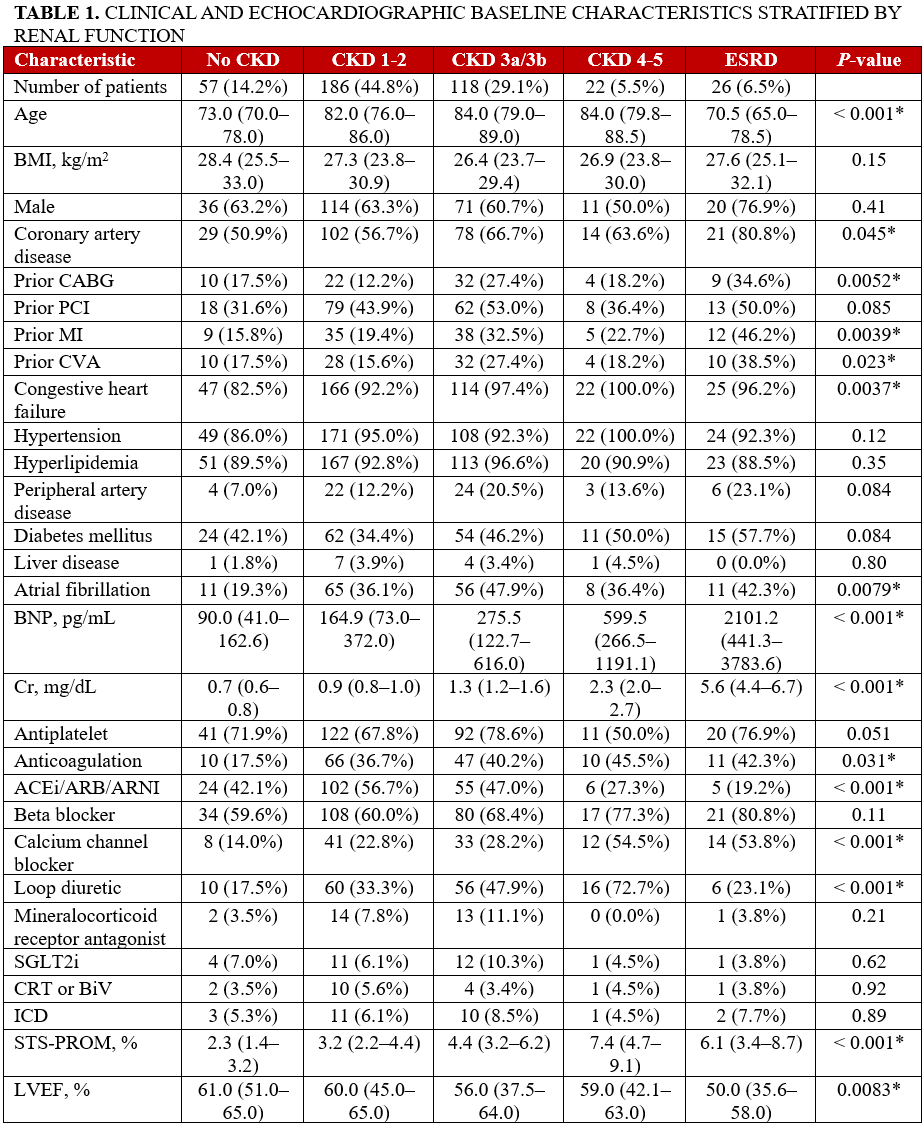
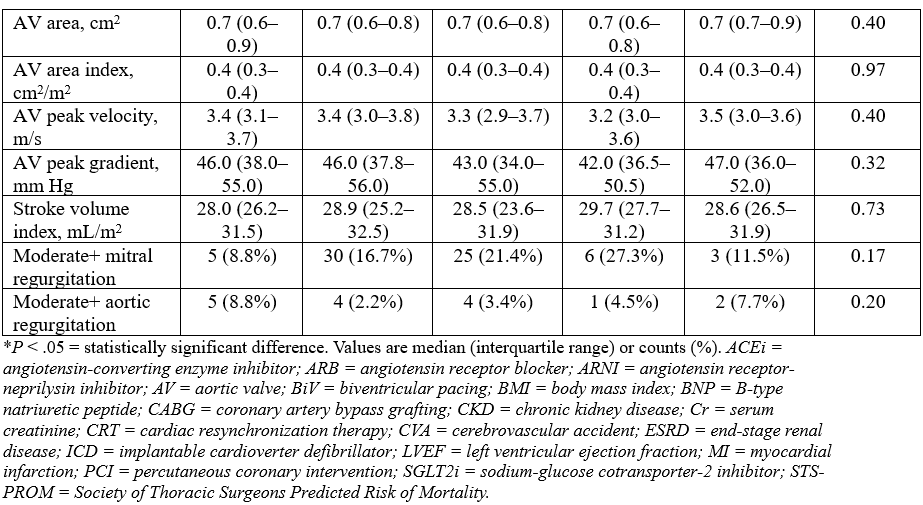
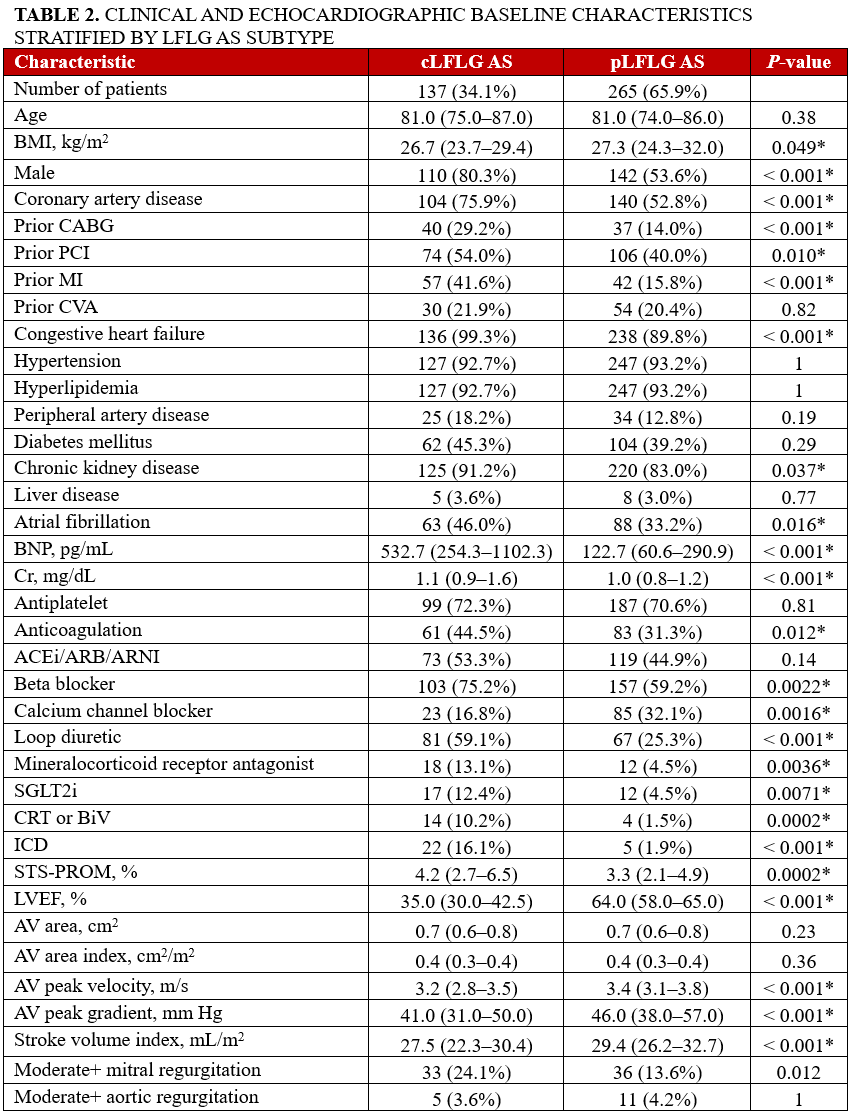

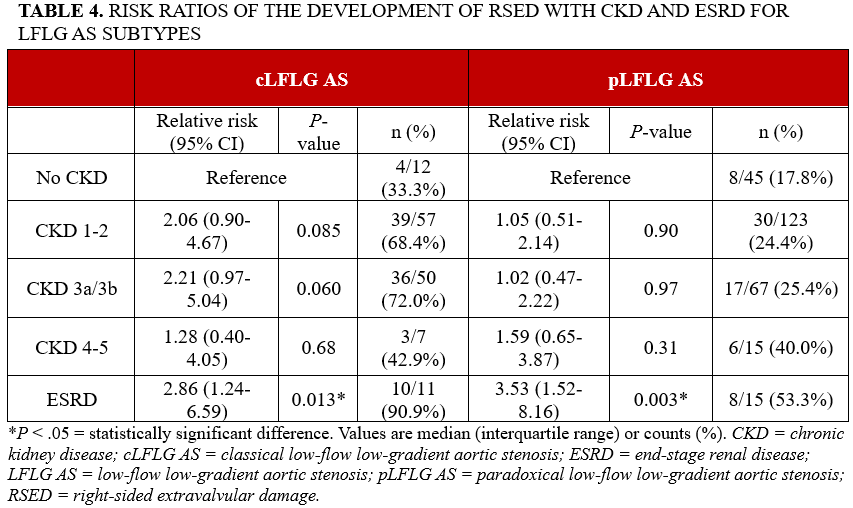
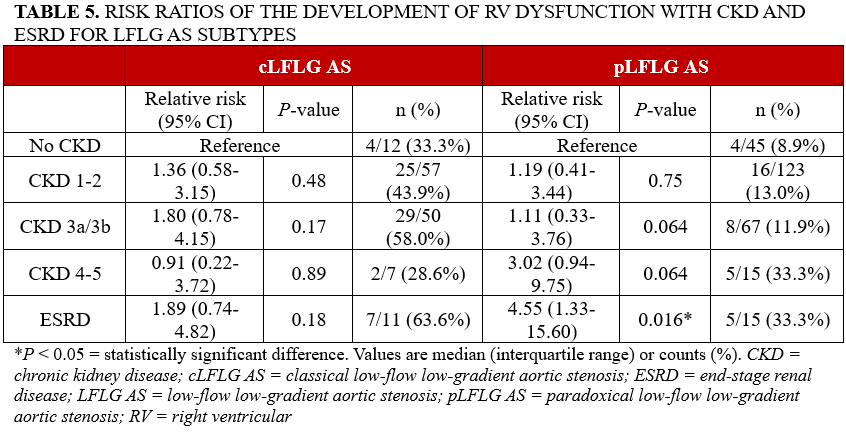
Of the patients with cLFLG AS, 12 (8.8%) had no CKD, 57 (41.6%) had CKD 1-2, 50 (36.5%) had CKD 3a/3b, 7 (5.1%) had CKD 4-5, and 11 (8.0%) had ESRD. Of the patients with pLFLG AS, 45 (17.0%) had no CKD, 123 (46.4%) had CKD 1-2, 67 (25.3%) had CKD 3a/3b, 15 (5.6%) had CKD 4-5, and 15 (5.6%) had ESRD. Overall, the cLFLG AS group had a higher proportion of patients with CKD, male sex, CAD, and CHF, along with prior CABG, percutaneous coronary intervention, and MI (Table 2).
CKD’s association with RSED and RV dysfunction
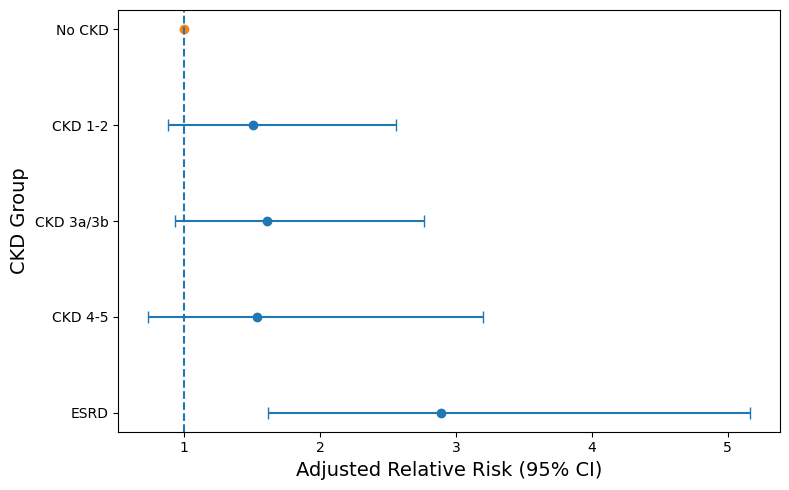
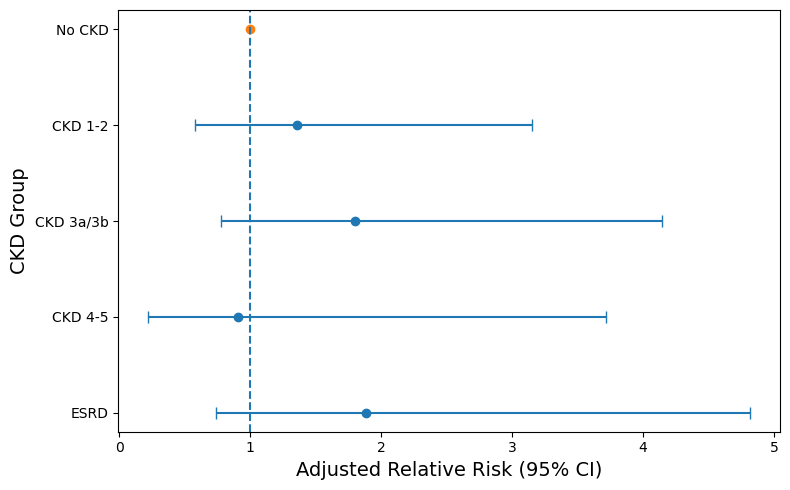
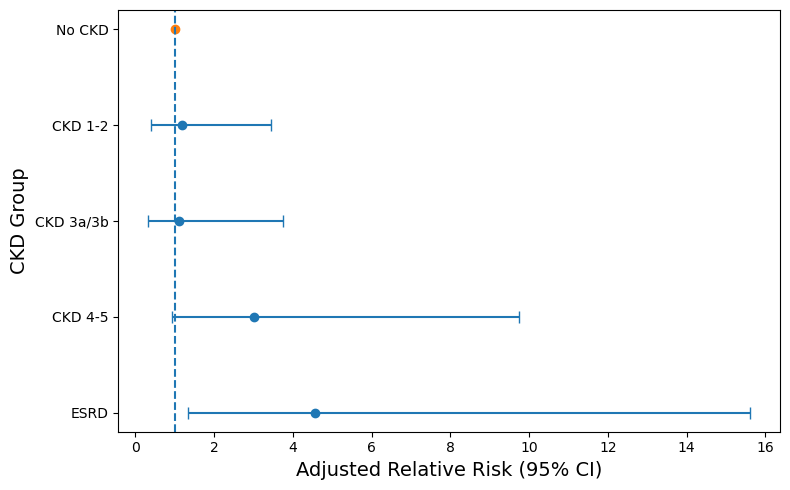
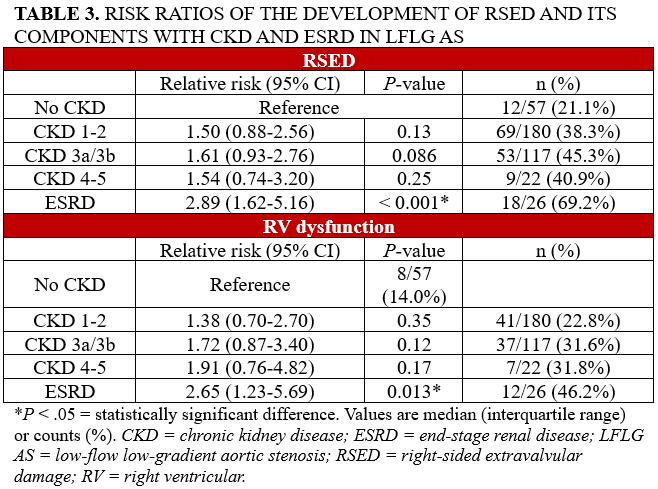
The analysis of all patients with LFLG AS revealed a significant association between ESRD and RSED (RR: 2.89; 95% CI, 1.62-5.16; P < .001) (Figure 1), as well as between ESRD and RV dysfunction (RR: 2.65; 95% CI, 1.23-5.69; P = .013) (Table 3). The association between ESRD and RSED persisted for both cLFLG AS (RR: 2.86; 95% CI, 1.24-6.59; P = .013) and pLFLG AS (RR: 3.53; 95% CI, 1.52-8.16; P = .003) (Table 4). However, in cLFLG AS, RV dysfunction was not associated with any of the CKD stages (Figure 2). Contrastingly, in pLFLG AS, ESRD was associated with RV dysfunction (RR: 4.55; 95% CI, 1.33-15.60; P = .016) (Figure 3, Table 5). For LFLG AS overall and each of its subtypes, CKD 1-2, CKD 3a/3b, and CKD 4-5 were not associated with either RSED or RV dysfunction.
Post-TAVR survival analysis by CKD stage
During a maximum follow-up of 71 months, multivariate Cox regression revealed increased post-TAVR mortality for all LFLG AS in patients with CKD 4-5 (HR: 4.24; 95% CI, 1.80-9.96; P < .001) and ESRD (HR: 4.26; 95% CI, 1.86-9.75; P < .001) compared with those without CKD (Figure 4). Similarly, ESRD was associated with increased mortality in both cLFLG AS (HR: 4.89; 95% CI, 1.20-19.86; P = .026) and pLFLG AS (HR: 5.46; 95% CI, 1.86-16.04; P = .0020). However, only cLFLG AS also demonstrated increased mortality among patients with CKD 4-5 (HR: 6.34; 95% CI, 1.50-26.84; P = .012). For LFLG AS overall and its subtypes, mortality was not associated with CKD 1-2 or CKD 3a/3b.
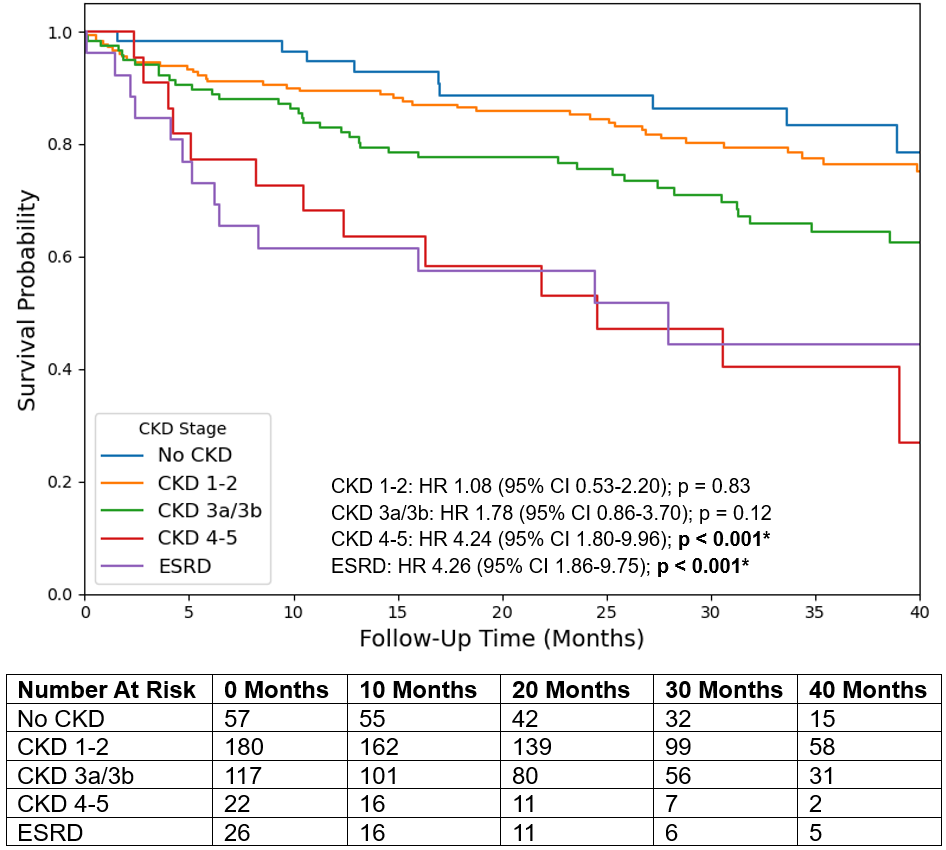
Post-TAVR survival analysis by CKD status, RSED, and RV dysfunction
Across the overall LFLG AS cohort, CKD 3+ and RSED were independently associated with increased mortality. Mortality was highest with RSED and CKD 3+ together (HR: 4.29; 95% CI, 2.49-7.39; P < .001), demonstrating significantly higher mortality than RSED alone (HR: 1.90; 95% CI, 1.17-3.07; P = .0094) and CKD 3+ alone (HR: 1.83; 95% CI, 1.08-3.11; P = .025) (Figure 5). Likewise, RV dysfunction and CKD 3+ were independently associated with increased mortality for all LFLG AS, with their combination at the highest risk (HR: 3.39; 95% CI, 1.99-5.77; P < .001). However, while combined RV dysfunction and CKD 3+ had significantly higher mortality than RV dysfunction alone (HR: 1.80; 95% CI, 1.00-3.24; P = .049), no difference in mortality was found when compared with CKD 3+ alone.
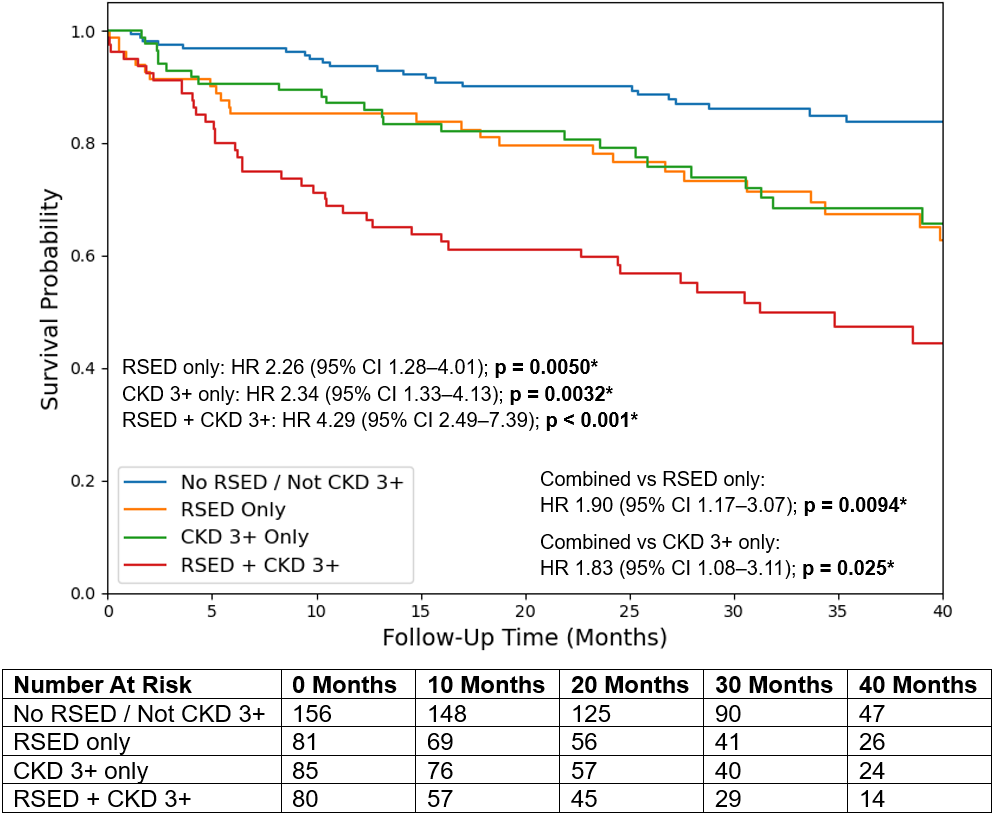
Subtype analysis of cLFLG AS demonstrated a renal-driven pattern of increased mortality, which was associated with CKD 3+ alone (HR: 4.25; 95% CI, 1.38-13.08; P = .0116) and RSED and CKD 3+ together (HR: 4.04; 95% CI, 1.55-10.56; P = .0044). Moreover, RSED and CKD 3+ together were associated with a significantly higher mortality risk than RSED alone (HR: 1.90; 95% CI, 1.03-3.53; P = .041) but not compared with CKD 3+ alone (Figure 6). Survival analysis by RV dysfunction yielded parallel findings: mortality was associated with CKD 3+ alone (HR: 2.75; 95% CI, 1.26-6.03; P = .011) and with RV dysfunction and CKD 3+ together (HR: 2.36; 95% CI, 1.15-4.81; P = .019), which had a significantly higher mortality risk than RV dysfunction alone (HR: 2.19; 95% CI, 1.02-4.73; P = .045) but not compared with CKD 3+ alone. Notably, neither RSED alone nor RV dysfunction alone was associated with mortality.
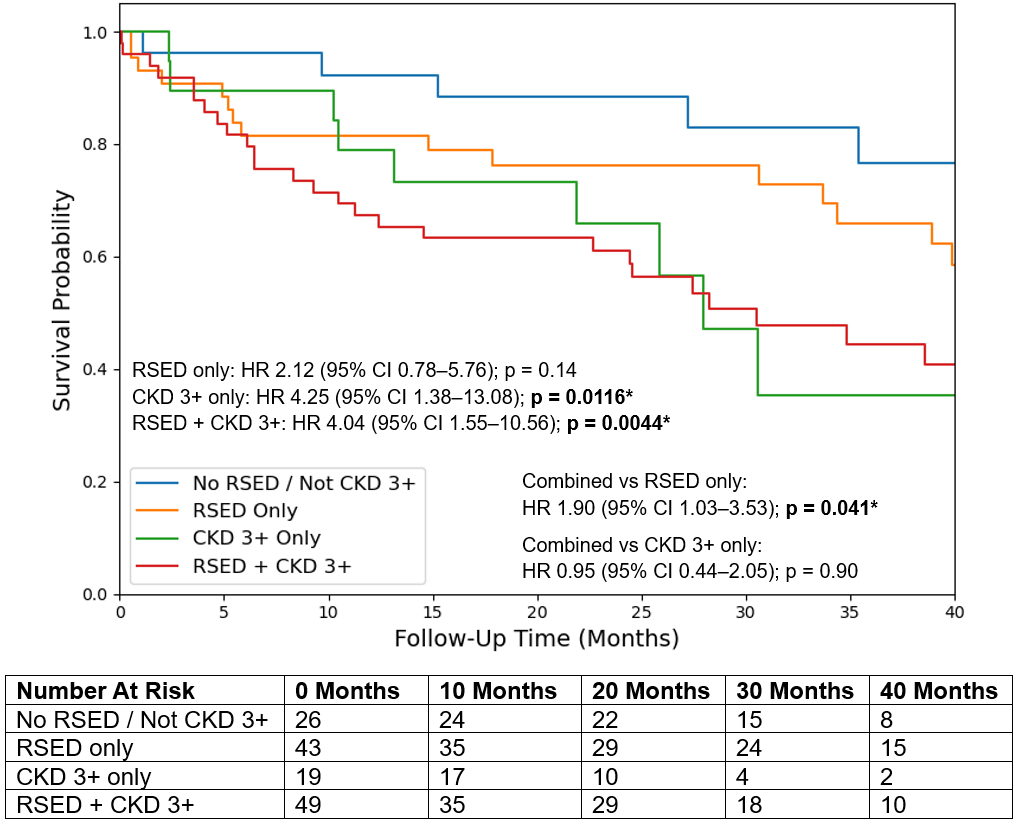
For pLFLG AS, RSED alone was associated with increased mortality (HR: 2.24; 95% CI, 1.06-4.73; P = .035). CKD 3+ was not independently associated with mortality, but there was significantly higher mortality risk among the group with RSED and CKD 3+ together (HR: 5.04; 95% CI, 2.51-10.11; P < .001), with higher mortality than RSED alone (HR: 2.25; 95% CI, 1.03-4.94; P = .043) and CKD 3+ alone (HR: 2.74; 95% CI, 1.34-5.59; P = .0057) (Figure 7). In the RV dysfunction analysis, both RV dysfunction and CKD 3+ were independently associated with mortality, with the highest risk found in their combination (HR: 6.04; 95% CI, 2.78-13.12; P < .001). The combination of RV dysfunction and CKD 3+ did not differ significantly in mortality from RV dysfunction alone; it did, however, differ from CKD 3+ alone (HR: 2.95; 95% CI, 1.35-6.47; P = .0067).
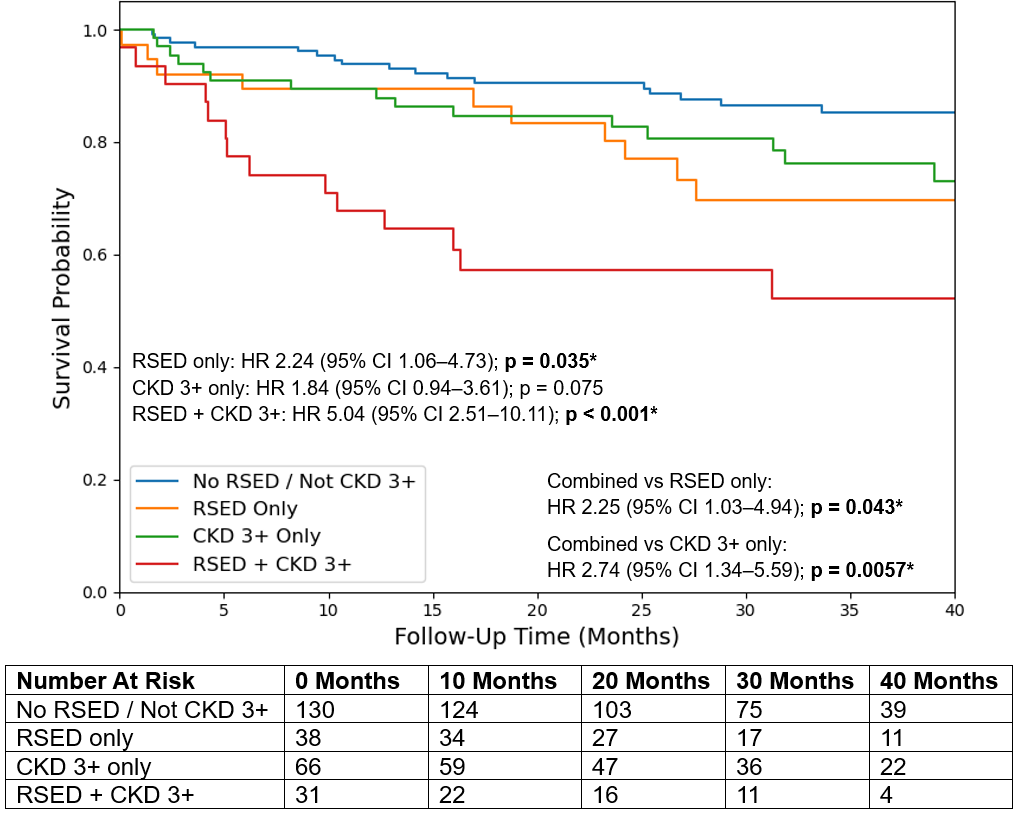
Discussion
Severe renal dysfunction as a risk factor for RSED and RV dysfunction
In this retrospective cohort of patients with LFLG AS who were selected to undergo TAVR, we found that advanced renal dysfunction, particularly ESRD, was strongly associated with RSED and RV dysfunction. Importantly, this association was not observed in earlier stages of CKD. Our findings extend prior work by demonstrating that, among patients with LFLG AS, the burden of RSED is primarily concentrated among those with ESRD rather than across the full spectrum of CKD severity. This suggests a threshold effect, whereby the association with right-sided cardiac dysfunction is observed only with the hemodynamic, inflammatory, and neurohormonal dysfunction seen in advanced renal dysfunction.18
A particularly novel finding of this study is the differential relationship between CKD and RV dysfunction across LFLG AS subtypes. Among patients with pLFLG AS, ESRD was associated with a markedly increased risk of developing RV dysfunction, whereas no such association was observed in cLFLG AS. pLFLG AS is characterized by concentric remodeling, small ventricular cavity size, and LV diastolic dysfunction,21,22 all of which may render the RV susceptible to dysfunction when compounded with the adverse loading conditions and inflammatory stress imposed by renal dysfunction. Importantly, though, the absence of a similar association in cLFLG AS should be interpreted cautiously given the smaller sample size of this subgroup, which increases the risk of type II error.
CKD, RSED, and RV dysfunction as risk factors for post-TAVR mortality
Across the overall LFLG cohort, both CKD 3+ and RSED were independently associated with increased mortality after TAVR, and the presence of both conditions was associated with the highest mortality risk. Notably, the combined RSED and CKD 3+ group was found to have significantly higher mortality compared with either condition alone, possibly suggesting multiplicative adverse prognostic effects. Similar findings were observed when examining RV dysfunction specifically. These findings align with the known post-TAVR risks that both renal dysfunction and right-sided cardiac damage impose. Together, these conditions likely represent advanced cardiorenal syndrome and systemic disease progression, which explains the markedly elevated mortality observed when both are present. Importantly, though, residual confounding remains possible. The association between advanced renal dysfunction and mortality may arise from the former’s role as a signal of global systemic illness or frailty, rather than as an independently associated risk factor.
A key finding of this study is the differential prognostic significance of renal dysfunction and right-sided cardiac damage between cLFLG AS and pLFLG AS. In cLFLG AS, advanced renal dysfunction emerged as the dominant predictor of mortality, potentially reflecting more advanced systemic disease and reduced physiologic reserve in this population. In contrast, pLFLG AS demonstrated a multiplicative interaction between right-sided damage and renal dysfunction, with patients bearing both comorbidities having particularly poor post-TAVR prognoses. As previously noted, the pLFLG AS phenotype arises from LV concentric remodeling and diastolic dysfunction typical of heart failure with preserved ejection fraction.21,22 RV systolic dysfunction in this context may signal either a superimposed myopathic process involving the RV, characterized by cardiomyocyte loss and contractile impairment similar to that observed in heart failure with reduced ejection fraction,23 or progression to more advanced stages of heart failure with preserved ejection fraction.24 The convergence of impaired LV filling and intrinsic RV systolic dysfunction may therefore explain why RSED specifically identifies a higher-risk phenotype in paradoxical LFLG AS.
Key points
These findings have significant clinical implications. First, they suggest that patients with LFLG AS and advanced renal disease represent a particularly high-risk subgroup with respect to right-sided cardiac disease, which can further worsen post-TAVR outcomes. Second, the heightened vulnerability of patients with pLFLG AS with advanced CKD developing RV dysfunction underscores the importance of careful right-heart assessment in this population during pre-TAVR evaluation. Indeed, these findings suggest that patients with pLFLG AS with advanced CKD may be at higher risk of developing RSED. The combination of these 2 risk factors, in turn, may be associated with significantly increased post-TAVR mortality. Ultimately, more cautious evaluation may be warranted among those patients with pLFLG AS who have already developed both comorbidities. Likewise, patients who have not yet developed RSED may benefit from intervention before they do so. Finally, greater weight should be placed on the presence of advanced CKD among patients with cLFLG AS, irrespective of extravalvular damage, as renal dysfunction alone is associated with their post-TAVR mortality risk.
Limitations
Several limitations of this study warrant consideration. The retrospective design introduces the potential for residual confounding despite multivariable adjustment. For example, advanced renal dysfunction may be a signal of global systemic illness and frailty, rather than an independent risk factor for mortality. Echocardiographic assessment of RV dysfunction and pulmonary hypertension was based on clinically reported parameters rather than standardized right heart catheterization measurements. The relatively small number of patients with CKD 4-5 and ESRD may limit statistical power for certain subgroup analyses and necessitates cautious interpretation of effect estimates. Additionally, the relatively small number of patients with cLFLG AS increases the risk of a type II error occurring during statistical analysis. Thus, the differences observed between the LFLG AS subtypes, particularly regarding the association between CKD and right-sided cardiac damage, should be interpreted cautiously. Finally, this study focused on pre-TAVR associations with RSED and did not evaluate longitudinal changes in right-sided cardiac function.
Conclusions
Among patients with LFLG AS who were selected to undergo TAVR, advanced renal dysfunction, particularly ESRD, was associated with an increased burden of RSED and RV dysfunction. This association is most pronounced among patients with pLFLG AS, in whom ESRD is strongly linked to RV dysfunction, but not so in cLFLG AS. Importantly, both advanced CKD and the presence of RSED are associated with higher post-TAVR mortality; CKD is the predominant risk factor in cLFLG AS, while the 2 comorbidities possibly have a multiplicative association with mortality in pLFLG AS. These findings highlight a possible subtype-specific interaction between renal dysfunction, right-sided cardiac remodeling, and clinical outcomes, and underscore the importance of comprehensive right-heart evaluation in patients with LFLG AS and advanced CKD. Further study is warranted to verify these findings, particularly for patients with LFLG AS.
Affiliations and Disclosures
Eric J. Kim, MD, MBE1; Yash Prakash, MD1; Hassan Beesley, MD1; Grigory Manyak, MD1; Mihir Prakash2; Lakshay Chopra, MD3; Akarsh Sharma, MD1; Carlo Mannina, MD3; George D. Dangas, MD, PhD4; Lucy M. Safi, DO4; Sahil Khera, MD, MPH4; Samin K. Sharma, MD4; Annapoorna S. Kini, MD4; Gilbert H.L. Tang, MD, MSc, MBA4; Stamatios Lerakis, MD, PhD4
From the 1Samuel Bronfman Department of Medicine at Mount Sinai Hospital, New York, New York; 2College of Arts and Sciences at Case Western Reserve University, Cleveland, Ohio; 3Department of Cardiology at Mount Sinai Morningside and West, New York, New York; 4Mount Sinai Fuster Heart Hospital, New York, New York.
Disclosures: Dr Khera is a consultant and proctor for Medtronic, consultant and proctor for Abbott Structural Heart, consultant and proctor for W. L. Gore & Associates, consultant and advisory board member of EastEnd Medical, and serves on the speaker’s bureau for Zoll Medical and Edwards Lifesciences. Dr Tang has received speaker’s honoraria and served as a physician proctor, consultant, advisory board member, TAVR publications committee member, RESTORE study steering and screening committee member, APOLLO trial screening committee member, and IMPACT MR steering committee member for Medtronic; has received speaker’s honoraria and served as a physician proctor, consultant, advisory board member, ENVISION trial screening committee member, and TRILUMINATE trial anatomic eligibility and publications committee member for Abbott Structural Heart; and has served as an advisory board member for Boston Scientific, Anteris, Philips, Edwards Lifesciences, Peija Medical, and Shenqi Medical Technology. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Declaration of generative artificial intelligence (AI) and AI-assisted technologies: During the preparation of this work, the authors used ChatGPT (OpenAI) for both statistical software coding and language editing of the manuscript. All AI-generated content and code were reviewed and edited by the authors, who take full responsibility for the accuracy and integrity of the final manuscript.
Address for correspondence: Stamatios Lerakis, MD, PhD, Icahn School of Medicine at Mount Sinai, 1 Gustave L. Levy Place, New York, NY 10029, USA. Email: stamatios.lerakis@mountsinai.org
References
1. Beerkens FJ, Tang GHL, Kini AS, et al. Transcatheter aortic valve replacement beyond severe aortic stenosis: JACC state-of-the-art review. J Am Coll Cardiol. 2025;85(9):944-964. doi:10.1016/j.jacc.2024.11.051
2. Garbi M, MacCarthy P, Shah AM, Chambers JB. Classical and paradoxical low-flow low-gradient aortic stenosis: a heart failure perspective. Struct Heart. 2018;2(1):3-9. doi:10.1080/24748706.2017.1384876
3. Prakash Y, Chopra L, Mannina C, et al. Comparative outcomes of transcatheter aortic valve replacement and conservative management in patients with low-flow, low-gradient aortic stenosis. Am J Cardiol. 2025;252:30-39. doi:10.1016/j.amjcard.2025.05.018
4. Lee HJ, Kim K, Gwak SY, et al. Impact of renal function on myocardial remodeling and outcomes in patients with moderate aortic stenosis. J Am Soc Echocardiogr. 2025;38(8):658-667. doi:10.1016/j.echo.2025.04.008
5. Makki N, Lilly SM. Advanced chronic kidney disease: relationship to outcomes post-TAVR, a meta-analysis. Clin Cardiol. 2018;41(8):1091-1096. doi:10.1002/clc.22993
6. Eleid MF, Michelena HI, Nkomo VT, et al. Causes of death and predictors of survival after aortic valve replacement in low flow vs. normal flow severe aortic stenosis with preserved ejection fraction. Eur Heart J Cardiovasc Imaging. 2015;16(11):1270-1275. doi:10.1093/ehjci/jev091
7. Dalsania AK, Chopra L, Ueyama H, et al. Effects of chronic kidney disease on transcatheter aortic valve replacement outcomes in low-flow low-gradient aortic stenosis. Am J Cardiol. 2023;206:86-88. doi:10.1016/j.amjcard.2023.08.096
8. Kansakar S, Qureshi WT, Sharma NR, et al. Impact of right heart failure on outcomes of transcatheter aortic valve implantation: insights from the national inpatient sample. J Clin Med. 2025;14(3):841. doi:10.3390/jcm14030841
9. Prakash Y, Sharma A, Chopra L, et al. Right-Sided Extravalvular Damage: An Overlooked Driver of Risk Model Failure in Low-Flow, Low-Gradient Aortic Stenosis. J Soc Cardiovasc Angiogr Interv. Published online December 30, 2025:104116. doi:10.1016/j.jscai.2025.104116
10. Chopra L, Ueyama H, Dalsania A, et al. Prognostic implication of pulmonary hypertension in low-flow low-gradient aortic stenosis after transcatheter aortic valve replacement. Am J Cardiol. 2023;208:6-12. doi:10.1016/j.amjcard.2023.09.054
11. Fan J, Liu X, Yu L, et al. Impact of tricuspid regurgitation and right ventricular dysfunction on outcomes after transcatheter aortic valve replacement: a systematic review and meta-analysis. Clin Cardiol. 2019;42(1):206-212. doi:10.1002/clc.23126
12. Schwartz LA, Rozenbaum Z, Ghantous E, et al. Impact of right ventricular dysfunction and tricuspid regurgitation on outcomes in patients undergoing transcatheter aortic valve replacement. J Am Soc Echocardiogr. 2017;30(1):36-46. doi:10.1016/j.echo.2016.08.016
13. Lindman BR, Maniar HS, Jaber WA, et al. Effect of tricuspid regurgitation and the right heart on survival after transcatheter aortic valve replacement: insights from the Placement of Aortic Transcatheter Valves II inoperable cohort. Circ Cardiovasc Interv. 2015;8(4):e002073. doi:10.1161/CIRCINTERVENTIONS.114.002073
14. Dini FL, Demmer RT, Simioniuc A, et al. Right ventricular dysfunction is associated with chronic kidney disease and predicts survival in patients with chronic systolic heart failure. Eur J Heart Fail. 2012;14(3):287-294. doi:10.1093/eurjhf/hfr176
15. Konstam MA, Kiernan MS, Bernstein D, et alAmerican Heart Association Council on Clinical Cardiology; Council on Cardiovascular Disease in the Young; Council on Cardiovascular Surgery and Anesthesia. Evaluation and management of right-sided heart failure: a scientific statement from the American Heart Association. Circulation. 2018;137(20):e578-e622. doi:10.1161/CIR.0000000000000560
16. Reinecke A, Dißmann P, Frey N, et al. In heart failure, echocardiographic parameters of right ventricular function are powerful tools to predict renal failure. ESC Heart Fail. 2025;12(3):2310-2320. doi:10.1002/ehf2.15176
17. Guazzi M, Gatto P, Giusti G, et al. Pathophysiology of cardiorenal syndrome in decompensated heart failure: role of lung-right heart-kidney interaction. Int J Cardiol. 2013;169(6):379-384. doi:10.1016/j.ijcard.2013.09.014
18. Noels H, van der Vorst EPC, Rubin S, et al. Renal-cardiac crosstalk in the pathogenesis and progression of heart failure. Circ Res. 2025;136(11):1306-1334. doi:10.1161/CIRCRESAHA.124.325488
19. Généreux P, Pibarot P, Redfors B, et al. Staging classification of aortic stenosis based on the extent of cardiac damage. Eur Heart J. 2017;38(45):3351-3358. doi:10.1093/eurheartj/ehx381
20. Chemla D, Humbert M, Sitbon O, Montani D, Hervé P. Systolic and mean pulmonary artery pressures: are they interchangeable in patients with pulmonary hypertension? Chest. 2015;147(4):943-950. doi:10.1378/chest.14-1755
21. Hachicha Z, Dumesnil JG, Bogaty P, Pibarot P. Paradoxical low-flow, low-gradient severe aortic stenosis despite preserved ejection fraction is associated with higher afterload and reduced survival. Circulation. 2007;115(22):2856-2864. doi:10.1161/CIRCULATIONAHA.106.668681
22. Pibarot P, Dumesnil JG. Low-flow, low-gradient aortic stenosis with normal and depressed left ventricular ejection fraction. J Am Coll Cardiol. 2012;60(19):1845-1853. doi:10.1016/j.jacc.2012.06.051
23. Schiattarella GG, Tong D, Hill JA. Can HFpEF and HFrEF coexist? Circulation. 2020;141(9):709-711. doi:10.1161/CIRCULATIONAHA.119.045171
24. Borlaug BA, Sharma K, Shah SJ, Ho JE. Heart failure with preserved ejection fraction: JACC scientific statement. J Am Coll Cardiol. 2023;81(18):1810-1834. doi:10.1016/j.jacc.2023.01.049