Extracorporeal Versus Conventional Cardiopulmonary Resuscitation for Out-of-Hospital Cardiac Arrests
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/25.00405. Epub April 29, 2026.
Abstract
Objectives. It remains unclear whether extracorporeal cardiopulmonary resuscitation (ECPR) is more efficacious than conventional cardiopulmonary resuscitation (CPR) at improving survival with favorable neurological outcomes among patients with out-of-hospital cardiac arrests (OHCA). The authors sought to determine the efficacy of ECPR vs conventional CPR among patients with OHCA in the pre-hospital setting.
Methods. The authors searched MEDLINE, EMBASE, and the Cochrane Library for randomized controlled trials (RCTs) comparing ECPR with conventional CPR for patients with OHCA. The primary outcome was survival with a favorable neurological outcome at 6-month follow-up. Count data were pooled across trials through random-effects models with inverse variance weighting to estimate risk ratios (RRs) and 95% confidence intervals (CIs).
Results. A total of 3 RCTs (n = 420) were included. Most patients (85%) were male, and the mean/median age ranged from 54 to 59 years. At 6 months, 28.4% of the patients randomized to ECPR survived with favorable neurological outcomes compared with 18.6% of the patients randomized to conventional CPR (RR, 1.47; 95% CI, 0.43-5.09). Similar results were obtained for survival at hospital discharge (RR, 1.35; 95% CI, 0.35-5.19). Risk of Bias 2 analysis indicated low risk in 1 trial and some concerns in others.
Conclusions. ECPR may be associated with improved survival with favorable neurological outcomes at 6 months compared to conventional CPR for the treatment of OHCA. However, the available evidence remains inconclusive, and additional trials are needed to definitively assess the efficacy of ECPR compared to conventional CPR for OHCA.
Introduction
Over 350 000 out-of-hospital cardiac arrests (OHCA) occur annually in the United States, and only 9.1% of patients survive until hospital discharge.1,2 Extracorporeal cardiopulmonary resuscitation (ECPR) is a novel salvage procedure in which extracorporeal membrane oxygenation (ECMO) is initiated to bypass native cardiopulmonary circulation and sustain end-organ perfusion through recovery from cardiogenic shock. The use of this procedure may increase survival with favorable neurological outcomes in patients who have experienced an OHCA.3
There have been several randomized controlled trials (RCTs) that have assessed the efficacy of ECPR vs conventional cardiopulmonary resuscitation (CPR) among patients who have experienced an OHCA without return of spontaneous circulation (ROSC).4-7 However, the RCTs were inconclusive, and it remains unclear whether ECPR improves the rate of survival with favorable neurological status compared with conventional CPR among adults with refractory OHCA. Thus, our objective was to determine the efficacy of ECPR vs conventional CPR for the treatment of OHCA with unsustained ROSC in the pre-hospital setting.
Methods
Our systematic review and meta-analysis of RCTs was conducted following a pre-specified protocol, registered on the Open Science Framework (https://osf.io/tpv5x/), and in accordance with the Peer Review of Electronic Search Strategies (PRESS) 2015 guidelines.8 Our study is reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and checklist (Supplemental Material).9 Ethical approval was not required as per the Tri-Council Policy Statement (2022), article 2.2, which exempts research that relies exclusively on publicly available information.
We systematically searched MEDLINE (via PubMed [National Institutes of Health]), EMBASE (Elsevier) (via Ovid [Wolters Kluwer]), and the Cochrane Library (John Wiley & Sons) from inception to July 5, 2023, for relevant studies. We used the RCT search hedge from the Cochrane Handbook for Systematic Reviews of Interventions to limit our search to RCTs.10 Our search strategy used MeSH terms (for PubMed), EMTREE terms (for EMBASE), and keywords for the following concepts: ECPR, advanced cardiac life support, and out-of-hospital cardiac arrest. Our database-specific search strategies are described in detail in Supplemental Tables 1 through 3. In addition, reference lists of relevant articles and clinicaltrial.gov were hand-searched to identify additional trials not included in the electronic search. The publications identified in our search were imported into the Covidence systematic review software (Veritas Health Innovation Ltd), where duplicate citations were automatically removed by the software with human supervision and verification.
Two independent investigators (T.S. and A.D.A.) screened the titles and abstracts of identified publications, adhering to predetermined inclusion and exclusion criteria. The full text of any citation considered potentially eligible by either reviewer was obtained and assessed in duplicate, with disagreements resolved by consensus or a third reviewer. Included trials involved individuals with OHCA who had not achieved sustained ROSC in the pre-hospital setting and were randomized to receive either early ECPR or any form of conventional advanced cardiovascular life support. RCTs that reported survival and neurological outcomes at 6-month follow-up or greater were included in our meta-analysis. Crossover trials, abstracts, conference proceedings, observational studies, case reports, and case series were all excluded from our study. Trials with no data on survival and neurologic outcomes at 6 months were excluded. Finally, trials conducted in individuals with OHCA of a presumed non-cardiac etiology or with sustained ROSC within 5 minutes (or 3 rounds of defibrillation) of advanced cardiac life support performed by EMS were excluded. These inclusion and exclusion criteria are documented within our protocol on the Open Science Framework (https://osf.io/tpv5x/).
Data extraction
Two independent investigators (T.S. and A.D.A.) extracted data into Covidence, with data extracted according to an intention-to-treat approach. Thus, data were extracted and analyzed based on the original treatment assignments of participants in the RCT, regardless of treatment received. Disagreements were resolved through consensus or by third review (TZ). Data extraction was subsequently verified and cross-checked by a third investigator (A.M.)
Data extracted from the RCTs included baseline demographic and clinical characteristics, such as age, sex, and comorbidities. In addition, we extracted data on arrest characteristics, including the rate of arrests that were treated with bystander CPR, number of defibrillations/shocks received pre-hospitalization, initial rhythm, and time intervals between events, such as start of arrest to emergency medical service (EMS) arrival, the onset of arrest to start of EMS transport to the hospital, and beginning of the arrest to ROSC. Finally, data on any serious adverse events (SAE) pertaining to the neurological and cardiovascular systems were extracted.
Outcomes
The pre-specified primary outcome was 6-month survival with a favorable neurological outcome. Included RCTs assessed neurological outcomes using the Cerebral Performance Categories (CPC) 5-point scale system.11 In this scale, a CPC 1 describes a good cerebral performance, CPC 2 describes moderate cerebral disability (but independent), CPC 3 describes a severe cerebral disability, CPC 4 describes a coma or vegetative state, and, finally, a CPC 5 describes brain death.11 A favorable neurological outcome was defined as a CPC score of 1 or 2. Pre-established secondary outcomes were survival to hospital discharge, 30-day survival, and 3-month survival following OHCA. The safety outcomes assessed were any cardiovascular SAEs, neurological SAEs, and any other SAEs at discharge, 30 days, 3 months, and 6 months.
Quality assessment
The quality of RCTs was assessed independently by 2 reviewers (T.S. and A.D.A.) using the second version of the Cochrane Collaboration’s tool for assessing the risk of bias in randomized trials (RoB 2). Disagreements were resolved through consensus or by third review (TZ). Quality assessment results are reported in the results section. All eligible studies were included in the manuscript, regardless of study quality.
Statistical analysis
We used DerSimonian and Laird random-effects meta-analytic models with inverse variance weighing and Jackson and modified Hartung-Knapp method extensions to pool crude count data across all included RCTs and obtain risk ratios (RRs) and corresponding 95% confidence intervals (CIs).
The primary comparison was 6-month survival with favorable neurological outcomes in patients with OHCA randomized to early ECPR vs conventional CPR. The amount of heterogeneity that was present was assessed using the I2 and Tau2 statistics. All analyses were performed using Revman (The Cochrane Collaboration).
Results
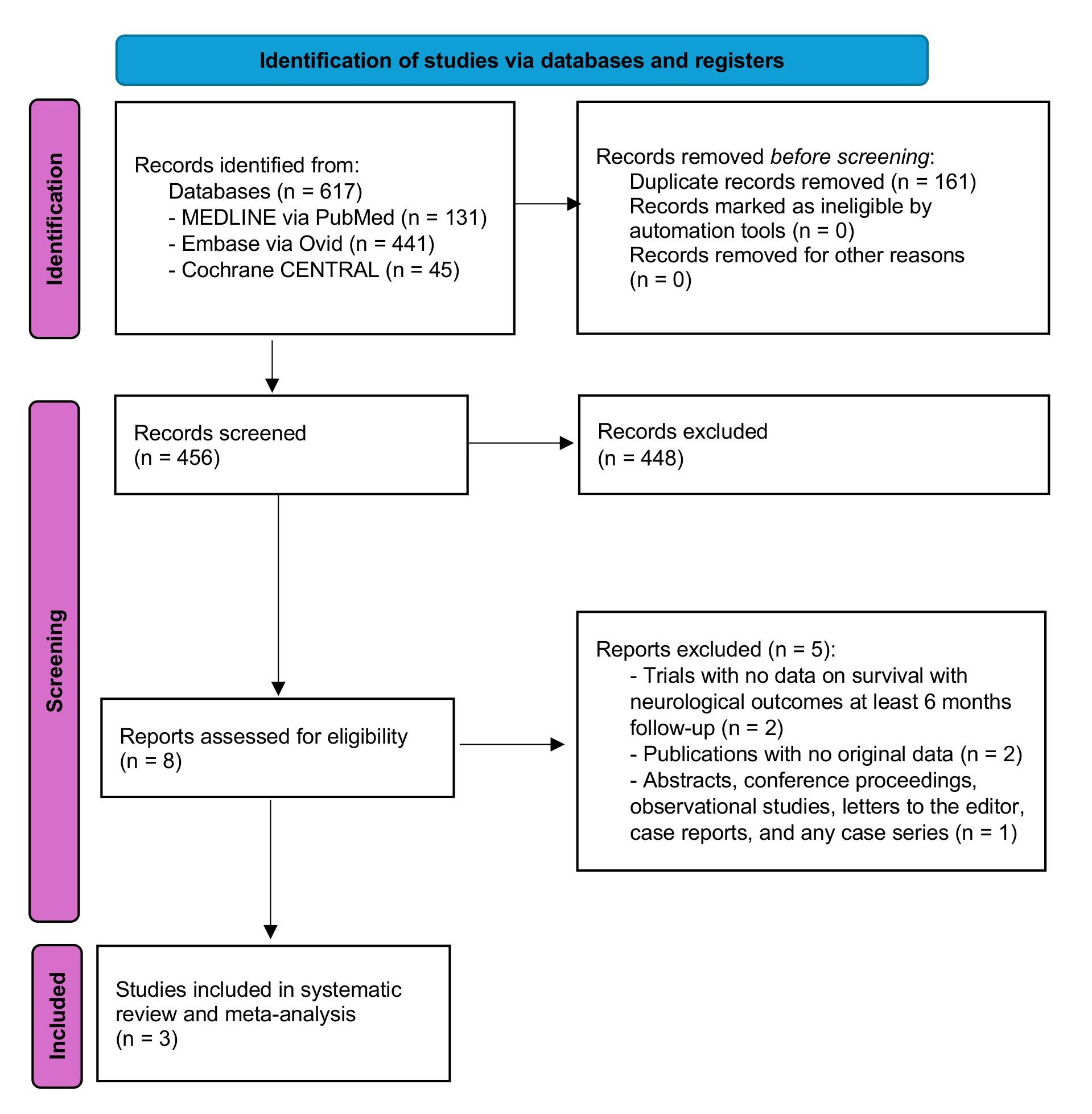
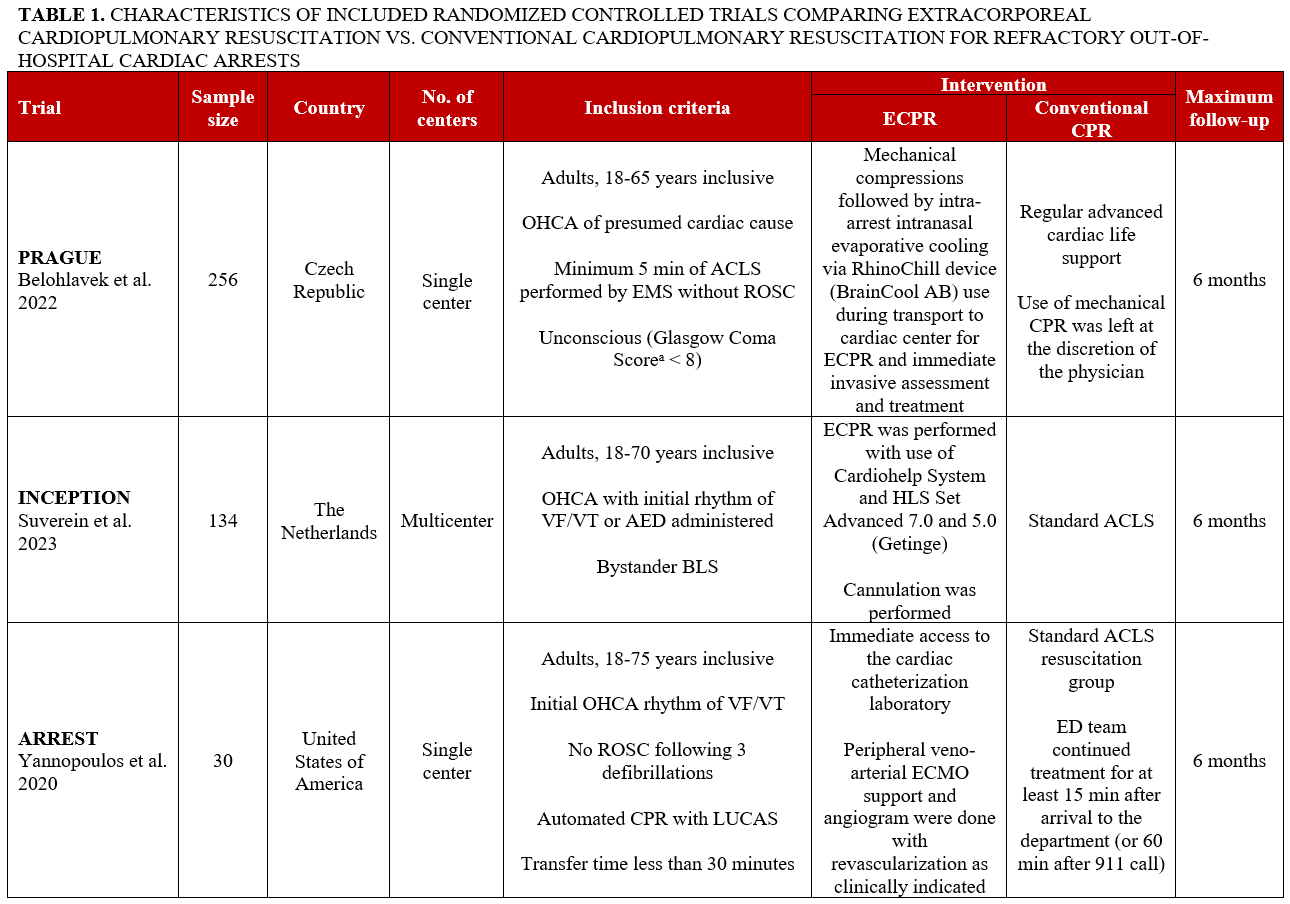
Our electronic search identified 617 potentially eligible records (Figure 1). After removing duplicates, 456 records were screened. A total of 3 RCTs published between 2020 and 2023 met our inclusion criteria and were thus included in the systematic review and meta-analysis (Figure 1).5-7 All trials enrolled adults who experienced a witnessed OHCA with no sustained ROSC within 5 to 15 minutes (or after 3 defibrillations) of advanced cardiac life support (ACLS) performed by EMS (Table 1). The maximum follow-up period was 6 months in all included trials.

Study characteristics
We presented the baseline clinical characteristics of the 420 participants included in the 3 studies in Table 2. Patient characteristics were broadly comparable, although some variability was observed across studies. The participants were mainly male (n = 357, 85%), and their mean/median ages ranged from 54 to 59 years. The prevalence of hypertension varied across the 3 studies, ranging from 13% to 55%. The prevalence of chronic heart failure ranged from 0% to 10%. Baseline chronic kidney disease was not reported in the INCEPTION trial, and its prevalence was low in the other studies. Cerebrovascular accident was reported in the INCEPTION and ARREST trials but not in the PRAGUE trial. In INCEPTION, prevalence was 5% in the ECPR arm and 17% in the conventional CPR arm, while in ARREST it was 0% in the ECPR arm and 7% in the conventional CPR arm.
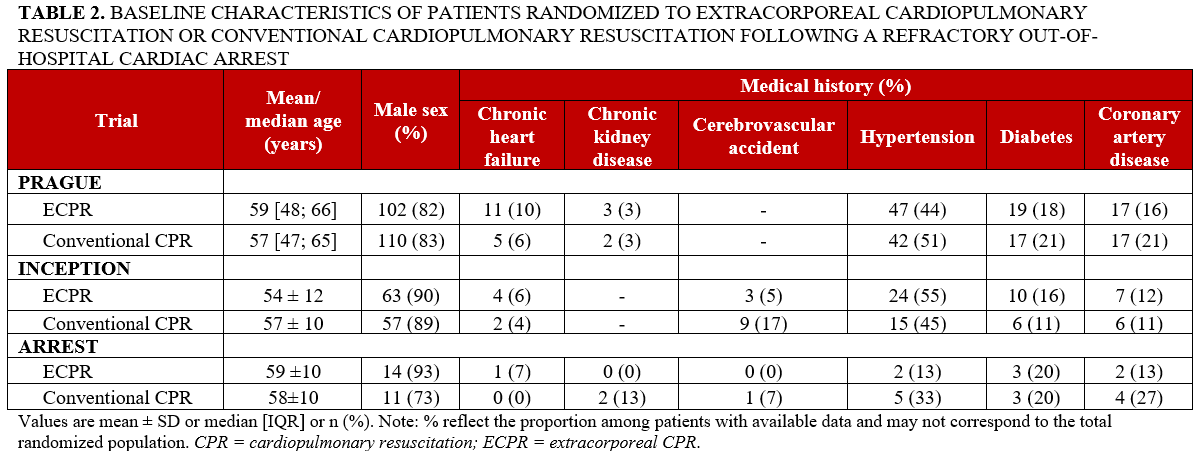
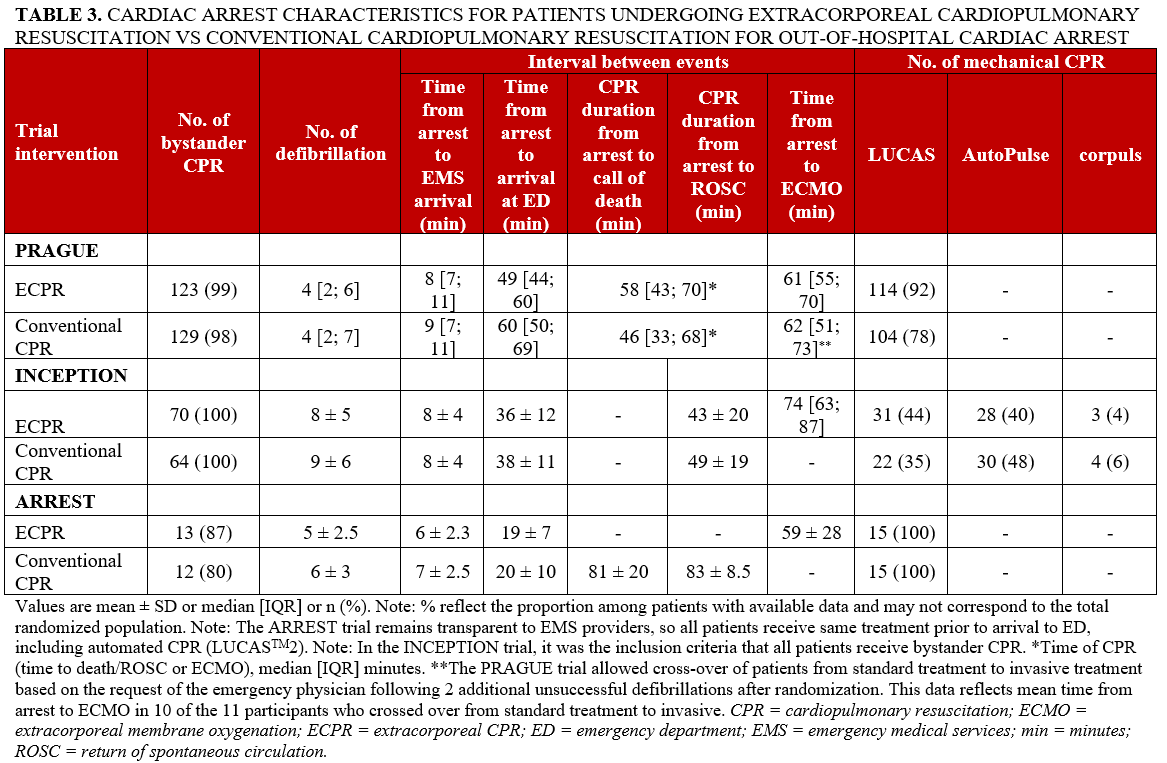
The clinical characteristics related to the cardiac arrest are presented in Table 3. The time from arrest to the initiation of ECMO varied, with durations of 59 ± 28 minutes in the ARREST trial, 61 minutes in the PRAGUE trial, and 74 minutes in the INCEPTION trial. All participants in the INCEPTION trial were required to receive bystander CPR. Eighty-three percent of participants in the ARREST trial and 98% of participants in the PRAGUE trial received bystander CPR. Across all 3 trials, the LUCAS mechanical chest compression system (Stryker Medical) was used during transport to hospital and in the ER; the INCEPTION trial—a multicenter RCT—employed other mechanical compression devices as well (eg, the AutoPulse [ZOLL Medical Corporation] and corpuls [GS Elektromedizinische Geräte G. Stemple] devices). In the PRAGUE trial, 11 participants (8%) of the non-invasive arm received ECPR, while the rate of crossover in the INCEPTION trial was 5%. The ARREST trial had no crossovers. All trials implemented post-arrest temperature management as part of standard care. In the PRAGUE trial, pre-hospital intranasal evaporative cooling was used in a subset of participants in the invasive strategy group (17%) and in some participants in the standard strategy group who crossed over to the invasive approach (9%).
Quality assessment
We describe the risk of bias of included trials, assessed using the RoB 2 Cochrane tool, in Supplemental Figure 1. The ARREST trial was deemed to have a low risk of bias, while the PRAGUE and INCEPTION trials were deemed to have some concerns for bias because of deviations from the intended intervention, primarily related to crossover between treatment groups.
Efficacy assessment
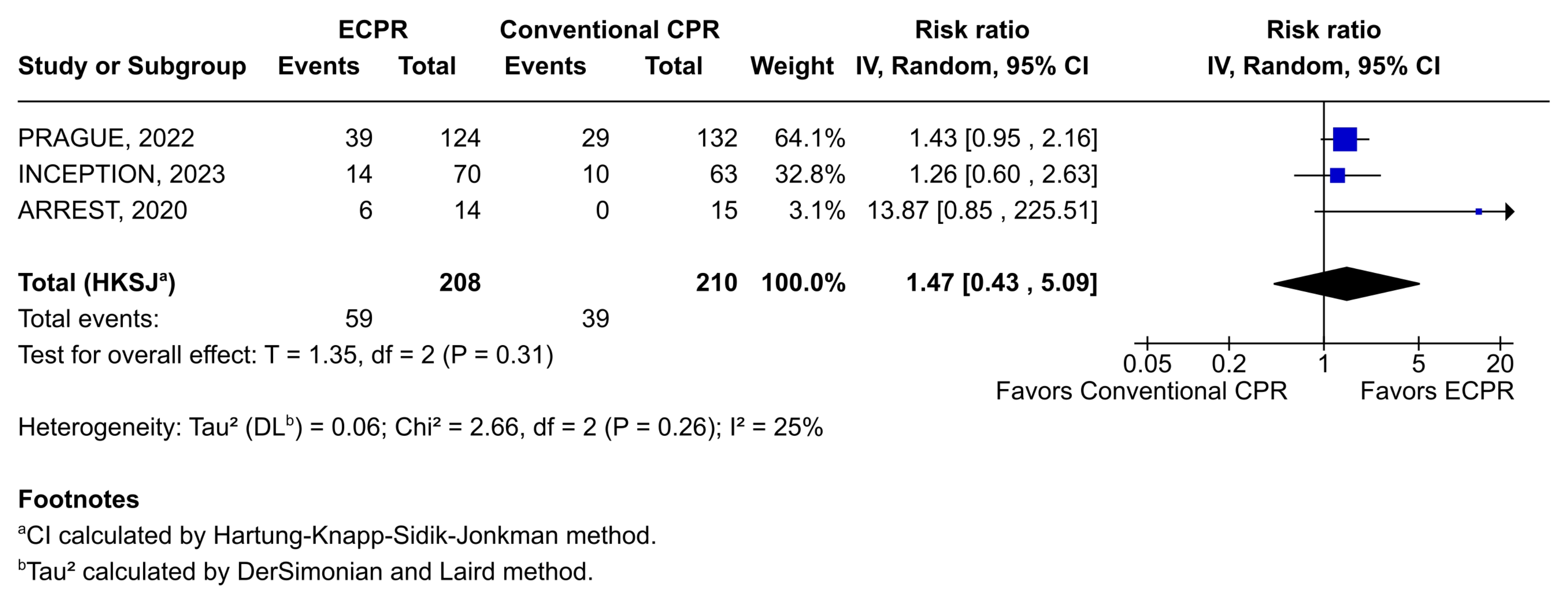
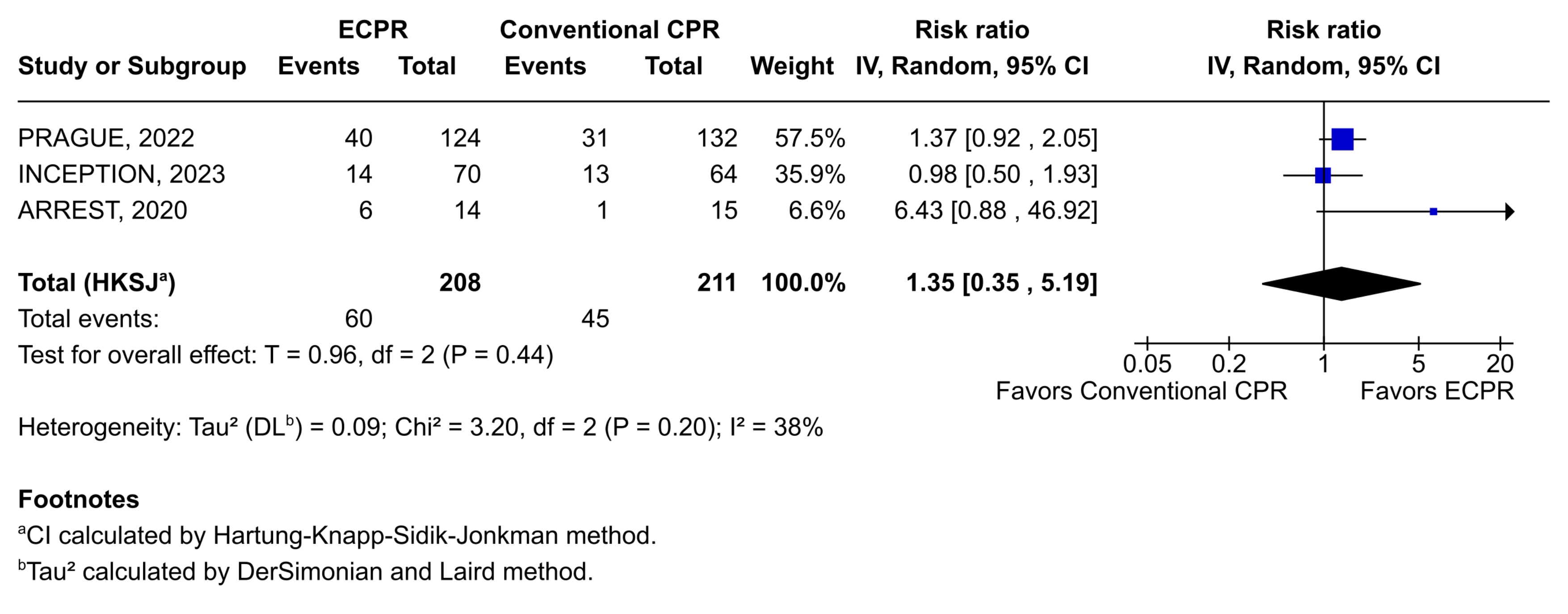
The results of the meta-analysis of survival with favorable neurological outcome at 6 months following an OHCA are provided in Figure 2. Early initiation of ECPR resulted in numerically higher survival to 6 months with favorable neurological outcomes compared with conventional CPR; however, it did not reach statistical significance. Combining data across trials yielded a pooled RR of 1.47 (95% CI, 0.43-5.09). Similar results were obtained for survival with favorable neurological outcome at hospital discharge (RR, 1.35; 95% CI, 0.35-5.19) (Figure 3; Supplemental Table 4).
Safety assessment
Safety reporting varied considerably across studies, with differences in outcome definitions and data availability (Table 4). Bleeding events were more frequently reported in the ECPR group, although direct comparisons were limited by heterogeneity in reporting. In the PRAGUE trial, overall bleeding events were reported in 36 of 116 participants (31.0%) in the ECPR group compared with 10 of 69 (15.5%) in the conventional CPR group. In the INCEPTION trial, rates were 11 of 70 (15.7%) and 2 of 64 (3.1%), respectively. In the ARREST trial, bleeding outcomes were reported by subtype, with high proportions observed in the ECPR group across several categories, including retrosternal or intrathoracic bleeding (4/13 [30.7%]) and bleeding requiring transfusion (5/12 [41.7%]). Technical complications, including ECMO circulation failure and tubing or cannulation-related issues, were reported exclusively in participants receiving ECPR.
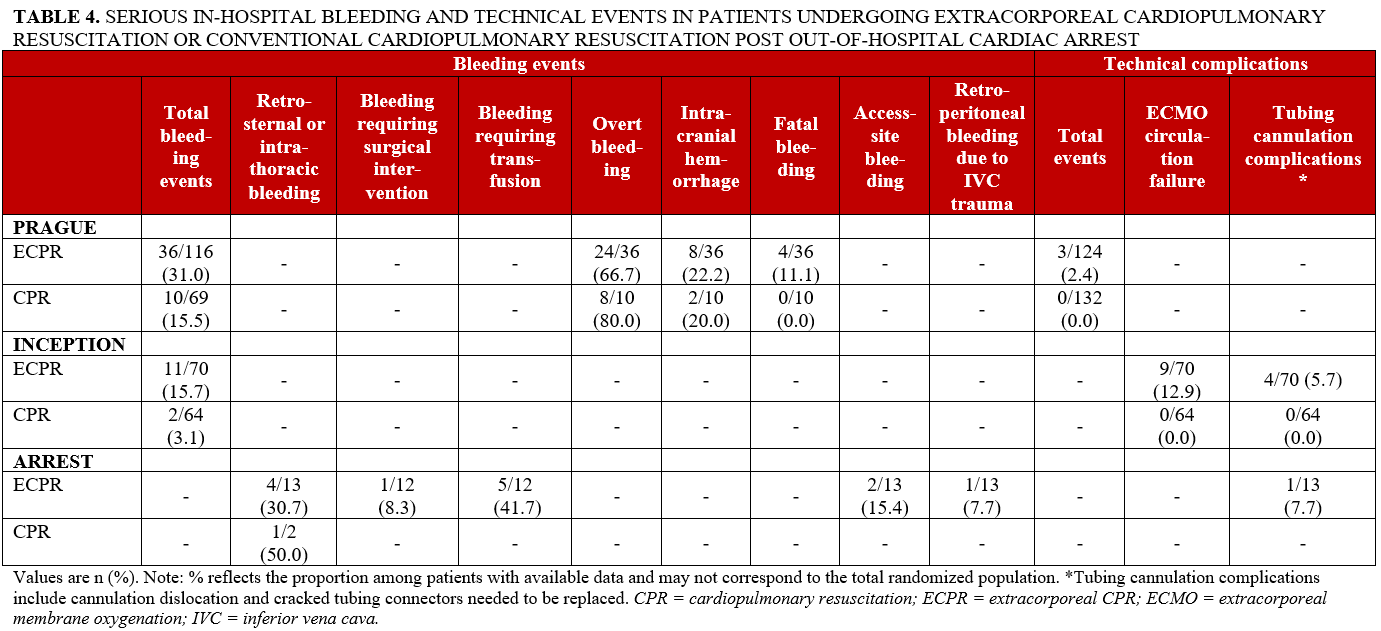
Cardiovascular and neurological adverse events were inconsistently reported (Table 5). In the INCEPTION trial, cardiogenic shock and limb ischemia were more frequent in the ECPR group compared with conventional CPR (15/70 [21.4%] vs 5/64 [7.8%] and 4/70 [5.7%] vs 0/64 [0%], respectively). In the ARREST trial, cardiogenic shock was common in both groups, although estimates in the conventional CPR group were based on limited available data. Post-anoxic encephalopathy was more frequently observed in the ECPR group in the INCEPTION trial (24/70 [34.3%] vs 3/64 [4.7%]). In the ARREST trial, neurological event estimates in the conventional CPR group were based on only 2 patients with available data, limiting interpretability.
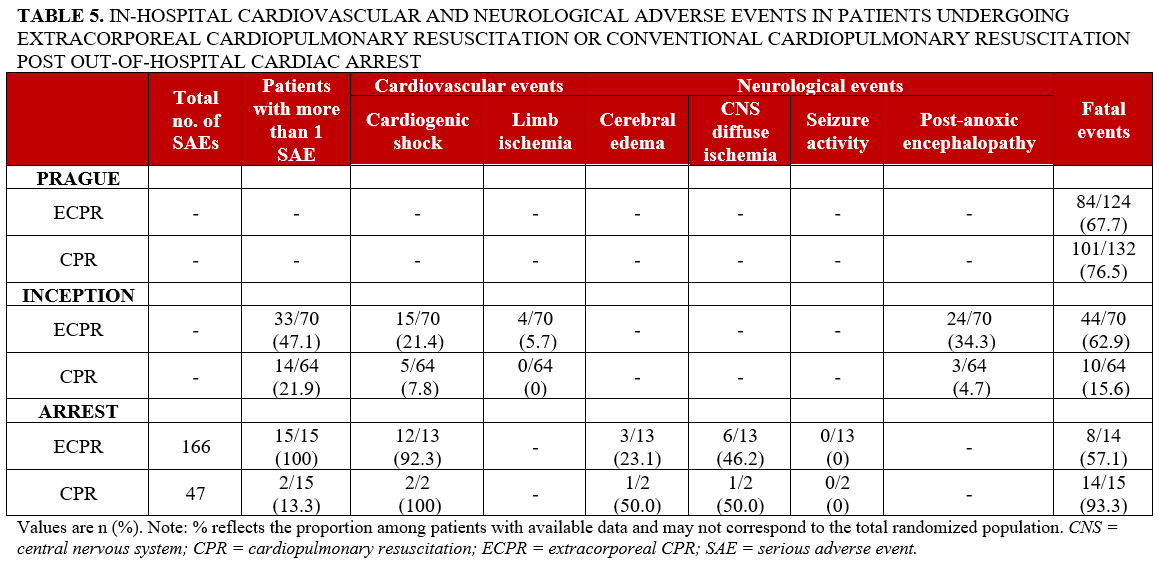
A more consistent pattern was observed for SAEs, with higher rates of multiple SAEs in the ECPR group in trials reporting this outcome. In the INCEPTION trial, 33 of 70 participants (47.1%) in the ECPR group experienced more than 1 SAE compared with 14 of 64 (21.9%) in the conventional CPR group. Similarly, in the ARREST trial, all participants in the ECPR group (15/15 [100%]) experienced more than 1 SAE compared with 2 of 15 (13.3%) in the conventional CPR group. However, rates of fatal adverse events varied across studies without a consistent pattern.
Discussion
Our study aimed to assess the efficacy and safety of early ECPR vs conventional CPR in patients with refractory OHCA. Our results suggest that ECPR may be associated with improved survival with favorable neurological outcome at 6 months compared with conventional CPR in the management of OHCA. However, the available evidence remains inconclusive, and our meta-analytic estimates were accompanied by wide 95% CIs. Nonetheless, with available data suggesting potential benefits associated with the use of early ECPR, the present study suggests that additional, large, multicenter trials are needed to definitively assess the efficacy of ECPR vs conventional CPR for OHCA.
The trials included in this meta-analysis had mixed results. Only 30 patients were enrolled in the ARREST trial, which was the only trial that suggested the superiority of early ECPR over conventional CPR.5 However, the trial was terminated early by the National Heart, Lung and Blood Institute because the Data Safety Monitoring Board assessed ECMO-facilitated CPR to be superior to conventional CPR. Of note, the ARREST trial focused specifically on patients with ventricular fibrillation (VF), a shockable rhythm associated with higher survival and favorable neurologic outcomes.12 This may have contributed to the favorable outcomes observed in the ECPR group in the ARREST trial. However, differences in initial rhythm alone are unlikely to fully explain the observed findings. The INCEPTION trial also exclusively enrolled patients with shockable rhythms (VF or ventricular tachycardia [VT]) yet did not demonstrate a benefit of ECPR over conventional CPR. In contrast, while most patients in the PRAGUE trial had VF (61%), others presented with non-shockable rhythms, including pulseless electrical activity (18%) or asystole (21%). Consistent with findings from ARREST, a post-hoc analysis of PRAGUE demonstrated that an initial shockable rhythm was associated with a better prognosis compared with non-shockable rhythms in refractory OHCA.13 Taken together, these findings suggest that while initial rhythm is an important determinant of outcome, it does not fully account for the favorable results observed in the ARREST trial. Finally, given the small sample size and early termination of the ARREST trial, its findings should be interpreted with caution.
In contrast, data from larger trials, such as the PRAGUE and INCEPTION trials, demonstrated no difference in the efficacy of ECPR vs conventional CPR.6,7 The INCEPTION trial, being the only multicenter trial of the 3 trials included in our study, provides a more realistic insight into the general applicability of ECPR. Multicenter trials, such as the INCEPTION trial, effectively represent a broader, more heterogeneous application of ECPR by reflecting real-world challenges such as team availability, various hospital protocols, and diverse patient profiles; this underscores the need for additional multicenter trials to fully understand the benefits of ECPR.
Furthermore, changes in best practices for pre-hospital resuscitation methods during the trials may have affected survival outcomes. In the PRAGUE trial, mechanical chest compression devices were originally reserved for those randomized to ECPR.7 However, almost 10 months after the PRAGUE trial had begun enrollment, a major publication on mechanical chest compressions demonstrated the efficacy of mechanical chest compression, regardless of resuscitation strategy.7 In the INCEPTION trial, most, but not all, patients received mechanical CPR.6 In contrast, investigators in the ARREST trial required the use of mechanical CPR across both arms of the trial.5 With automated pistons or band-like mechanisms, mechanical CPR has been developed to provide continuous, effective chest compressions to restore blood flow while avoiding human error or exhaustion.14
All 3 trials also incorporated post-arrest temperature management as part of standard care, although the PRAGUE trial additionally included intra-arrest intranasal evaporative cooling (RhinoChill [BrainCool AB]) in a subset of patients in the invasive strategy group and in patients in the standard strategy group who crossed over to the invasive approach. While this may represent a source of clinical heterogeneity, the proportion exposed was small and is unlikely to have meaningfully affected the overall findings. The included trials did not consistently report the use of additional mechanical circulatory support devices (eg, intra-aortic balloon pump, Impella [Abiomed], or other ventricular assist devices) in a way that allowed comparison between treatment groups, which may represent an additional source of unmeasured heterogeneity.
The potential benefits and harms seen with ECPR may vary with patient characteristics, such as age, underlying neurologic status, cardiac rhythm, and baseline comorbidity.15 Across the included trials, participants were relatively young, predominantly male, and had a low prevalence of chronic heart failure and other major baseline comorbidities. These trial populations may therefore not fully reflect the higher-risk patients encountered in routine clinical practice.
The success of ECPR in OHCA is also highly dependent on pre-hospital and arrest-related factors, including whether the arrest is witnessed, the provision of bystander CPR, the estimated downtime and low-flow time, the quality of CPR, whether there is a shockable rhythm with intermittent ROSC throughout resuscitative efforts, and ultimately the arrest-to-cannulation time. In addition, center experience and ECPR volume may also influence outcomes. Current guidelines suggest that patients with witnessed cardiac arrests of presumed cardiac etiology with a shockable rhythm are more likely to benefit from ECPR, especially in cases where the duration from start of conventional CPR until initiation of ECPR is less than 60 minutes.16 Across the included trials, the time from arrest to ECMO initiation was broadly similar (approximately 60-75 minutes), suggesting that differences in cannulation timing are unlikely to account for the observed variability in outcomes. However, the duration between arrest and ECMO initiation is a well-established determinant of survival,17 and the available trial data did not allow for a detailed assessment of its impact in the present analysis.
Ongoing studies, such as the ON-SCENE study (NCT04620070), are evaluating the feasibility and potential impact of earlier or on-scene initiation of ECPR in patients with OHCA, which may help clarify whether reducing low-flow time translates into improved clinical outcomes.18 Similarly, the EVIDENCE trial (ACTRN12621000668808) compared expedited intra-arrest transport with more extended on-scene resuscitation, including access to ECPR when available, but did not demonstrate improved outcomes; however, relatively few patients ultimately received ECPR.19,20
In a recent meta-analysis,21 investigators examined the survival rates with favorable neurological outcomes in patients who underwent ECPR after OHCA and provided similar results to those of the present study. However, this previous study included trials that did not specifically evaluate outcomes at 6 months, focusing on both “mid-term” and “long-term” outcomes. Consequently, they combined results from 3 and 6 months as 1 endpoint, and survival to hospital discharge and 30 days as another endpoint. This approach may have introduced additional heterogeneity into their results.
The potential benefits of ECPR are supported by previous studies, one of which concluded that ECPR was associated with an increased rate of ROSC compared with conventional CPR.22 Anoxic brain injury due to a prolonged lack of cerebral perfusion is often the cause of death in patients with prolonged cardiac arrest.18 ECMO, coupled with high-quality pre-hospital ACLS, allows for the near-normal restoration of blood flow in patients and may benefit OHCA patients with an underlying cardiac or otherwise reversible etiology.23 By providing hemodynamic and oxygenation support,24 ECPR may potentially reduce anoxic injury, improve end-organ perfusion, and encourage favorable neurological outcome.
A long-term follow-up of the PRAGUE trial provides additional insight into outcomes beyond the 6-month time frame evaluated in the present analysis. At a median follow-up of approximately 5 years, the ECPR-based approach was associated with improved long-term survival, although no differences in neurological outcomes, major cardiovascular events, or quality of life were observed between groups. These findings are broadly consistent with the uncertainty observed at earlier time points and suggest that any survival benefit may not be accompanied by corresponding improvements in other clinically relevant outcomes, although the trial may have been underpowered to detect such differences.
The extensive resource requirements associated with ECPR pose substantial challenges to the accessibility and availability of this approach to OHCA. On average, the cost of ECPR is $73,122 per patient, with total hospital costs amounting to $210,142.25 ECMO care is labor-intensive, requiring an intensive care unit bed with individual nursing, an on-site perfusionist continuously present, and operative time for device maintenance and eventual decannulation. Thus, the implementation of ECMO programs in hospitals requires an investment in the training and development of personnel such as critical care physicians, nurses, and paramedics.26 A successful ECPR system requires a multidisciplinary team where collaboration and communication are essential for success.26 As a result, some centers may not have the necessary resources to become high-volume centers of excellence in ECMO care and offer continuous coverage, which may further contribute to adverse outcomes.
Across the 3 trials, ECPR was associated with more frequent reporting of certain complications, particularly bleeding and ECMO-related technical events, although adverse event definitions varied substantially across studies.5-7 For several outcomes, estimates were based on limited data availability with small denominators, particularly in the conventional CPR arm, restricting the interpretability of between-group comparisons (eg, neurological outcomes in the ARREST trial). Venoarterial ECMO inherently increases retrograde arterial flow, which can lead to left ventricular overload, pulmonary edema, blood stagnation, and intracardiac thrombi.27 ECMO also involves large-bore vascular access, typically via the common femoral artery and vein, leading to increased risk of access-site bleeding complications. This risk is further heightened by the systemic use of anticoagulation.28 These complications are not unique to the trials included in our study; the use of ECMO, irrespective of the underlying condition, such as cardiac events or respiratory failure, is frequently linked to complications that may be serious and affect several organ systems, including the vasculature and the brain.29
Neurological adverse events are a recognized complication of ECMO support. In the included trials, neurological adverse events appeared more frequently reported in the ECPR group in absolute terms. This may reflect several factors, including prolonged low-flow time prior to cannulation, thromboembolic complications, hemodynamic instability, and risks related to anticoagulation and vascular access.29-31 Thus, while ECMO may improve survival in selected patients, these findings highlight the importance of careful patient selection, timely initiation, and close neurologic monitoring.
There is a survivorship bias associated with ECMO, as patients who survive long enough to receive and remain on ECMO are also more likely to experience complications. This can lead to an apparent increase in the adverse event rates in the ECPR group, independent of treatment effect. It is therefore essential that these adverse events are carefully monitored and addressed through quality improvement initiatives. There may be ways to mitigate complications, such as hypoxic-ischemic brain injury via targeted temperature management, neurologic monitoring, and early reversal of hypoxia and ischemia.32 Tackling bleeding events that occur in the ECPR group may be managed through cautious anticoagulation management, but this approach can be associated with higher in-hospital mortality.32 Thus, while ECMO is intended to sustain life, it can also introduce serious complications. In addition, the nature of the procedure, which requires initiation of anticoagulation, may carry additional risks in patients that are of high-bleeding risk.33 However, there is evidence that ECMO may alter the pharmacokinetics and pharmacodynamics of administered medications.21
Limitations
There are several potential limitations in our study. First, only one of the included trials was classified as having a low risk of bias, and the quality of the included primary studies must be considered when considering the results of the meta-analysis. Second, the risk of publication bias could not be quantitatively assessed because of the limited number of included trials and their modest sample sizes; this emphasizes the need for larger, more adequately powered trials in this area. Third, patients randomized to the ECMO arm are subject to a survivorship bias because they are artificially supported, which may affect shorter-term outcomes and adverse events. We therefore defined our primary endpoint accordingly and selected for trials that presented data on 6-month survival with favorable neurological outcomes.
Conclusions
ECPR may be associated with improved survival with favorable neurological outcomes at 6 months compared with conventional CPR for the treatment of OHCA. However, the available evidence is derived from small trials, and pooled estimates are inconclusive due to sparse data. Given the small sample size and heterogeneity within the protocols of the included trials, these findings should be interpreted with caution. There remains a need for additional large, adequately powered multicenter trials to conclusively evaluate the efficacy of ECPR for OHCA.
Affiliations and Disclosures
Tara Seirafi1,2; Jeremy Y. Levett, MD2,3; Areesha Moiz, MSc1; Tetiana Zolotarova, MD1; Kristian B. Filion, PhD1,2,4,5; Adelina Dobromir Angheluta1,2; Pauline Reynier, MSc1; Mark J. Eisenberg, MD, MPH1,2,4,5,6
From the 1Centre for Clinical Epidemiology, Lady Davis Institute, Jewish General Hospital, Montreal, Canada; 2Faculty of Medicine and Health Sciences, McGill University, Montreal, Canada; 3Division of Cardiac Surgery, Department of Surgery, McGill University, Montreal, Canada; 4Department of Epidemiology, Biostatistics and Occupational Health, McGill University, Montreal, Canada; 5Department of Medicine, McGill University, Montreal, Canada; 6Division of Cardiology, Jewish General Hospital/McGill University, Montreal, Canada.
A version of the study’s abstract was presented at the Transcatheter Cardiovascular Therapeutics (TCT) conference in October 2024 and has been published: Seirafi T, Levett J, Zolotarova T, et al. TCT-584 extracorporeal versus conventional cardiopulmonary resuscitation for out-of-hospital cardiac arrests: a systematic review and meta-analysis. JACC. 2024;84(18_Supplement). doi:10.1016/j.jacc.2024.09.693
Acknowledgments: The authors would like to thank Genevieve Gore, liaison librarian at McGill University, for her assistance with the development of the search strategy.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Funding: Ms Seirafi is supported by the Ivan Racheff Scholarship, funded through the McGill University Summer 2023 Research Bursary Program. Dr Filion is supported by a career award from the Fonds de recherche du Québec – santé and a William Dawson Scholar award from McGill University. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Data availability statement: This meta-analysis is based on aggregate data derived from publicly available, published studies. All relevant data used in the analysis have been included within the manuscript or provided in the corresponding appendices. No individual-level data were used or accessed in the course of this research.
Address for correspondence: Mark J. Eisenberg, MD, MPH, James McGill Professor of Medicine, Divisions of Cardiology and Clinical Epidemiology, Jewish General Hospital/McGill University, 3755 Côte Ste-Catherine Road, Suite H-421.1, Montreal, Quebec H3T 1E2, Canada. Email: mark.eisenberg@ladydavis.ca
References
1. Tsao CW, Aday AW, Almarzooq ZI, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics-2023 update: a report from the American Heart Association. Circulation. 2023;147(8):e93-e621. doi:10.1161/CIR.0000000000001123
2. Armour R, Wall N, Deakin J, Helmer J, Roberts L, Lee S. Cardiac Arrest Annual Report 2019/2020. British Columbia Emergency Health Services; 2021. Accessed August 7, 2023. https://www.bcehs.ca/our-research-site/Documents/annual_report_cardiac_arrest_2019-2020.pdf
3. Kumar KM. ECPR-extracorporeal cardiopulmonary resuscitation. Indian J Thorac Cardiovasc Surg. 2021;37(Suppl 2):294-302. doi:10.1007/s12055-020-01072-2
4. Hsu CH, Meurer WJ, Domeier R, et al. Extracorporeal cardiopulmonary resuscitation for refractory out-of-hospital cardiac arrest (EROCA): results of a randomized feasibility trial of expedited out-of-hospital transport. Ann Emerg Med. 2021;78(1):92-101. doi:10.1016/j.annemergmed.2020.11.011
5. Yannopoulos D, Bartos J, Raveendran G, et al. Advanced reperfusion strategies for patients with out-of-hospital cardiac arrest and refractory ventricular fibrillation (ARREST): a phase 2, single centre, open-label, randomised controlled trial. Lancet. 2020;396(10265):1807-1816. doi:10.1016/S0140-6736(20)32338-2
6. Suverein MM, Delnoij TSR, Lorusso R, et al. Early extracorporeal CPR for refractory out-of-hospital cardiac arrest. N Engl J Med. 2023;388(4):299-309. doi:10.1056/NEJMoa2204511
7. Belohlavek J, Smalcova J, Rob D, et al; Prague OHCA Study Group. Effect of intra-arrest transport, extracorporeal cardiopulmonary resuscitation, and immediate invasive assessment and treatment on functional neurologic outcome in refractory out-of-hospital cardiac arrest: a randomized clinical trial. JAMA. 2022;327(8):737-747. doi:10.1001/jama.2022.1025
8. McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of electronic search strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. doi:10.1016/j.jclinepi.2016.01.021
9. Shamseer L, Moher D, Clarke M, et al; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015;350:g7647. doi:10.1136/bmj.g7647
10. McKenzie JE, Brennan SE, Ryan RE, Thomson HJ, Johnston RV, Thomas J. Chapter 3: Defining the criteria for including studies and how they will be grouped for the synthesis [last updated August 2023]. In: Higgins JP, Thomas J, Chandler J, et al, eds. Cochrane Handbook for Systematic Reviews of Interventions version 6.5. Cochrane, 2024. https://www.cochrane.org/authors/handbooks-and-manuals/handbook/current/chapter-03
11. Ajam K, Gold LS, Beck SS, Damon S, Phelps R, Rea TD. Reliability of the Cerebral Performance Category to classify neurological status among survivors of ventricular fibrillation arrest: a cohort study. Scand J Trauma Resusc Emerg Med. 2011;19:38. doi:10.1186/1757-7241-19-38
12. Foszcz P, Springer A, Dreher A, et al. Impact of initial rhythm, rhythm at hospital admission and cause of arrest on the outcome of extracorporeal cardiopulmonary resuscitation in out-of-hospital cardiac arrest. Am J Emerg Med. 2026;99:90-96. doi:10.1016/j.ajem.2025.09.032
13. Havranek S, Fingrova Z, Rob D, et al. Initial rhythm and survival in refractory out-of-hospital cardiac arrest. Post-hoc analysis of the Prague OHCA randomized trial. Resuscitation. 2022;181:289-296. doi:10.1016/j.resuscitation.2022.10.006
14. Wang PL, Brooks SC. Mechanical versus manual chest compressions for cardiac arrest. Cochrane Database Syst Rev. 2018;8(8):CD007260. doi:10.1002/14651858.CD007260.pub4
15. Woszczyk D, Zasada W, Cholerzyńska H, Kłosiewicz T, Puślecki M. Neurological outcome predictors after extracorporeal cardiopulmonary resuscitation: a systematic review. Syst Rev. 2025;14(1):67. doi:10.1186/s13643-025-02818-y
16. Lorusso R, Whitman G, Milojevic M, et al. 2020 EACTS/ELSO/STS/AATS expert consensus on post-cardiotomy extracorporeal life support in adult patients. J Thorac Cardiovasc Surg. 2021;161(4):1287-1331. doi:10.1016/j.jtcvs.2020.09.045
17. Yukawa T, Kashiura M, Sugiyama K, Tanabe T, Hamabe Y. Neurological outcomes and duration from cardiac arrest to the initiation of extracorporeal membrane oxygenation in patients with out-of-hospital cardiac arrest: a retrospective study. Scand J Trauma Resusc Emerg Med. 2017;25(1):95. doi:10.1186/s13049-017-0440-7
18. Ali S, Moors X, van Schuppen H, et al. A national multi centre pre-hospital ECPR stepped wedge study; design and rationale of the ON-SCENE study. Scand J Trauma Resusc Emerg Med. 2024;32(1):31. doi:10.1186/s13049-024-01198-x
19. Burns B, Marschner I, Eggins R, et al; EVIDENCE Investigators. A randomized trial of expedited intra-arrest transfer versus more extended on-scene resuscitation for refractory out of hospital cardiac arrest: rationale and design of the EVIDENCE trial. Am Heart J. 2024;267:22-32. doi:10.1016/j.ahj.2023.10.003
20. Dennis M, Burns B, Marschner I, Keech A. Abstract 4135335: A randomized trial of expedited intra-arrest transfer versus more extended on-scene resuscitation for refractory out of hospital cardiac arrest: the EVIDENCE Study. Circulation. 2024;150(Suppl_1):A4135335-A. doi:10.1161/circ.150.suppl_1.4135335
21. Cheema HA, Shafiee A, Jafarabady K, et al. Extracorporeal cardiopulmonary resuscitation for out-of-hospital cardiac arrest: a meta-analysis of randomized controlled trials. Pacing Clin Electrophysiol. 2023;46(10):1246-1250. doi:10.1111/pace.14820
22. Dennis M, Lal S, Forrest P, et al. In-Depth Extracorporeal Cardiopulmonary Resuscitation in Adult Out-of-Hospital Cardiac Arrest. J Am Heart Assoc. 2020;9(10):e016521. doi:10.1161/JAHA.120.016521
23. Boudoulas KD, Whitson BA, Keseg DP, et al. Extracorporeal cardiopulmonary resuscitation (ECPR) for out-of-hospital cardiac arrest due to pulseless ventricular tachycardia/fibrillation. J Interv Cardiol. 2020;2020:6939315. doi:10.1155/2020/6939315
24. Bartos JA, Carlson K, Carlson C, et al. Surviving refractory out-of-hospital ventricular fibrillation cardiac arrest: critical care and extracorporeal membrane oxygenation management. Resuscitation. 2018;132:47-55. doi:10.1016/j.resuscitation.2018.08.030
25. Mishra V, Svennevig JL, Bugge JF, et al. Cost of extracorporeal membrane oxygenation: evidence from the Rikshospitalet University Hospital, Oslo, Norway. Eur J Cardiothorac Surg. 2010;37(2):339-342. doi:10.1016/j.ejcts.2009.06.059
26. Bertini P, Sangalli F, Meani P, et al; ITACTAIC. Establishing an extracorporeal cardiopulmonary resuscitation program. Medicina (Kaunas). 2024;60(12):1979. doi:10.3390/medicina60121979
27. Lim Y, Kim MC, Jeong IS. Left ventricle unloading during veno-arterial extracorporeal membrane oxygenation: review with updated evidence. Acute Crit Care. 2024;39(4):473-487. doi:10.4266/acc.2024.00801
28. Kalra R, Kosmopoulos M, Goslar T, Raveendran G, Bartos JA, Yannopoulos D. Extracorporeal cardiopulmonary resuscitation for cardiac arrest. Curr Opin Crit Care. 2020;26(3):228-235. doi:10.1097/MCC.0000000000000717
29. Cai J, Abudou H, Chen Y, et al. The effects of ECMO on neurological function recovery of critical patients: a double-edged sword. Front Med (Lausanne). 2023;10:1117214. doi: 10.3389/fmed.2023.1117214
30. Chiarini G, Cho SM, Whitman G, Rasulo F, Lorusso R. Brain injury in extracorporeal membrane oxygenation: a multidisciplinary approach. Semin Neurol. 2021;41(4):422-436. doi:10.1055/s-0041-1726284
31. Shi X, Zhang L, Zeng X, Li Y, Hu W, Xi S. Neurologic impairment in patients with extracorporeal cardiopulmonary resuscitation support: clinical features and long-term outcomes. Shock. 2023;59(1):41-48. doi:10.1097/SHK.0000000000002041
32. Kang JK, Darby Z, Bleck TP, Whitman GJR, Kim BS, Cho SM. Post-cardiac arrest care in adult patients after extracorporeal cardiopulmonary resuscitation. Crit Care Med. 2024;52(3):483-494. doi:10.1097/CCM.0000000000006102
33. Cheng V, Abdul-Aziz MH, Roberts JA, Shekar K. Optimising drug dosing in patients receiving extracorporeal membrane oxygenation. J Thorac Dis. 2018;10(Suppl 5):S629-S641. doi:10.21037/jtd.2017.09.154