Real-World Analysis of Outcomes of Patients Undergoing Coronary Intravascular Lithotripsy Versus Atherectomy for Calcified Coronary Artery Disease: Findings From the TriNetX Database
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/25.00351. Epub May 4, 2026.
Abstract
Objectives. Coronary intravascular lithotripsy (IVL) and coronary atherectomy are increasingly used for calcified coronary lesions, yet comparative data on their cardiovascular benefits remain limited. This real-world study compared outcomes of coronary IVL vs atherectomy using the TriNetX Global Collaborative Network.
Methods. Adult patients (≥18 years) undergoing PCI with either IVL or atherectomy between 2021 and 2024 were identified using procedural and diagnosis codes. Propensity score matching (1:1) balanced baseline demographics, comorbidities, laboratory values, and concomitant medications. The primary endpoint was all-cause mortality (ACM), while secondary endpoints included major adverse cardiovascular events (MACE), heart failure exacerbation (HFE), and procedural safety outcomes.
Results. Among 51 856 patients, 18 686 underwent IVL and 33 170 underwent atherectomy. After matching, 13 617 patients were retained in each group. At 30 days, IVL was associated with significantly lower risks of ACM (risk ratio [RR]: 0.67; 95% CI, 0.60-0.75; P < .001), MACE (RR: 0.42, 95% CI, 0.36-0.48; P < .001), and HFE (RR: 0.64; 95% CI, 0.57-0.72; P < .001) compared with atherectomy. These benefits persisted at 1 year for ACM (RR: 0.78; 95% CI, 0.73-0.84; P < .001), MACE (RR: 0.66; 95% CI, 0.60-0.72; P < .001), and HFE (RR: 0.79; 95% CI, 0.73-0.85; P < .001). Safety outcomes were comparable between groups.
Conclusions. In this propensity-matched, real-world cohort, IVL was associated with lower mortality and fewer adverse cardiovascular events than atherectomy, with a comparable safety profile. Given the likelihood of residual selection bias, these observational findings require confirmation in randomized trials.
Introduction
Calcific coronary lesions provide a major challenge to percutaneous coronary intervention (PCI), as they are associated with impaired stent delivery, suboptimal expansion, and worse outcomes.1 Atherectomy has been used traditionally for calcium modification but carries the risk of perforation in the vessels along with periprocedural complications.2 In 2021, coronary intravascular lithotripsy (IVL) (Shockwave) was approved by the US Food and Drug Administration (FDA) for managing heavily calcified coronary lesions after promising findings from the Disrupt CAD I-IV trials with a higher procedural success rate.3 However, head-to-head comparisons between coronary IVL and atherectomy to date remain limited, and often involve small sample sizes.4 Therefore, we investigated the comparative effectiveness of coronary IVL vs atherectomy in patients undergoing PCI using the TriNetX multicenter database.
Methods
Data source
The data in this study were collected from the TriNetX database, a global health collaborative clinical and research platform that collects real-time electronic health records (EHRs) from a network of healthcare organizations (HCOs) worldwide. TriNetX provides access to comprehensive electronic medical records, including demographics; diagnoses coded using the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM); procedures coded using the International Classification of Diseases, Tenth Revision, Procedure Coding System (ICD-10-PCS), or Current Procedural Terminology (CPT); laboratory values standardized with Logical Observation Identifiers Names and Codes (LOINC); and genomic data. The research network's data is sourced from academic and non-academic medical centers, community hospitals, and specialty units, encompassing data from both insured and non-insured patients. The HCOs are in 30 countries across the United States, Latin America, Europe, and the Asia-Pacific region. More details of the platform can be found elsewhere.5
This study was exempt from institutional review board review under US federal regulation 45 CFR 46.104(d)(4) because it involved the analysis of deidentified data obtained from the TriNetX research network.6 This study was conducted in accordance with the TriNetX Data Use Agreement and institutional policies governing the use of deidentified electronic health record data.
Patient population and study design
A retrospective cohort study was conducted in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines.7 Patients 18 years or older undergoing PCI with IVL or coronary atherectomy between January 2021 to December 2024 were identified (Supplemental Figure), as defined by Current Procedural Terminology, International Classification of Disease, Tenth Revision, Clinical Modification (ICD-10-CM) codes, Systematized Nomenclature of Medicine—Clinical Terms (SNOMED CT), and Healthcare Common Procedure Coding System (HCPCS). Details of code are mentioned in Supplemental Table 1.
The index event was defined as occurring 1 day after PCI with either lesion modification therapy, using an intention-to-treat framework. The study end date was the earliest of the last recorded patient encounter date, date of death, or the end of the follow-up period, on or before December 31, 2024.
Outcomes
The primary outcome was the risk of all-cause mortality (ACM). Secondary outcomes were major adverse cardiovascular events (MACE) (defined as a composite of acute myocardial infarction [MI], ischemic stroke, new onset heart failure [HF], and cardiac arrest), and heart failure exacerbation (HFE). Safety outcomes include perforation, bleeding, acute kidney injury (AKI), and stent thrombosis (ST) (Supplemental Table 2). Outcomes were assessed from the day of the index procedure through 30 days and 1 year; patients prior to the index event were excluded. Patients with outcomes of interest before the window period were excluded from the main analysis. Outcomes were identified and validated using data from the TriNetX database, a large, structured healthcare database that contains detailed diagnostic, procedural, and demographic information. Study outcomes definitions were based on ICD-10 diagnosis codes. Validated coding algorithms were applied to ensure accurate classification of events. Outcomes definitions were based on established standards and have been used in prior literature using the same source.
Statistical analysis
The study population was divided into 2 groups based on exposure to lesion modification therapy. Continuous variables are presented as mean ± standard deviation (SD), and categorical variables are reported as n (%). To ensure comparability of the cohorts, propensity score-matched analysis (PSM) (1:1) was performed for demographics, comorbidities, laboratory values, and concomitant medications use (Table 1). Patients having the outcome of interest prior to the window period were excluded from the final analysis. ICD-10-CM codes were used for all the outcomes of interest. PSM was performed using logistic regression based on the previously mentioned covariates and 1:1 greedy nearest-neighbor matching without replacement, and a caliper of 0.1 of the SD of the propensity score was conducted. Covariate balance was assessed using standardized mean differences, all of which were less than 0.1.
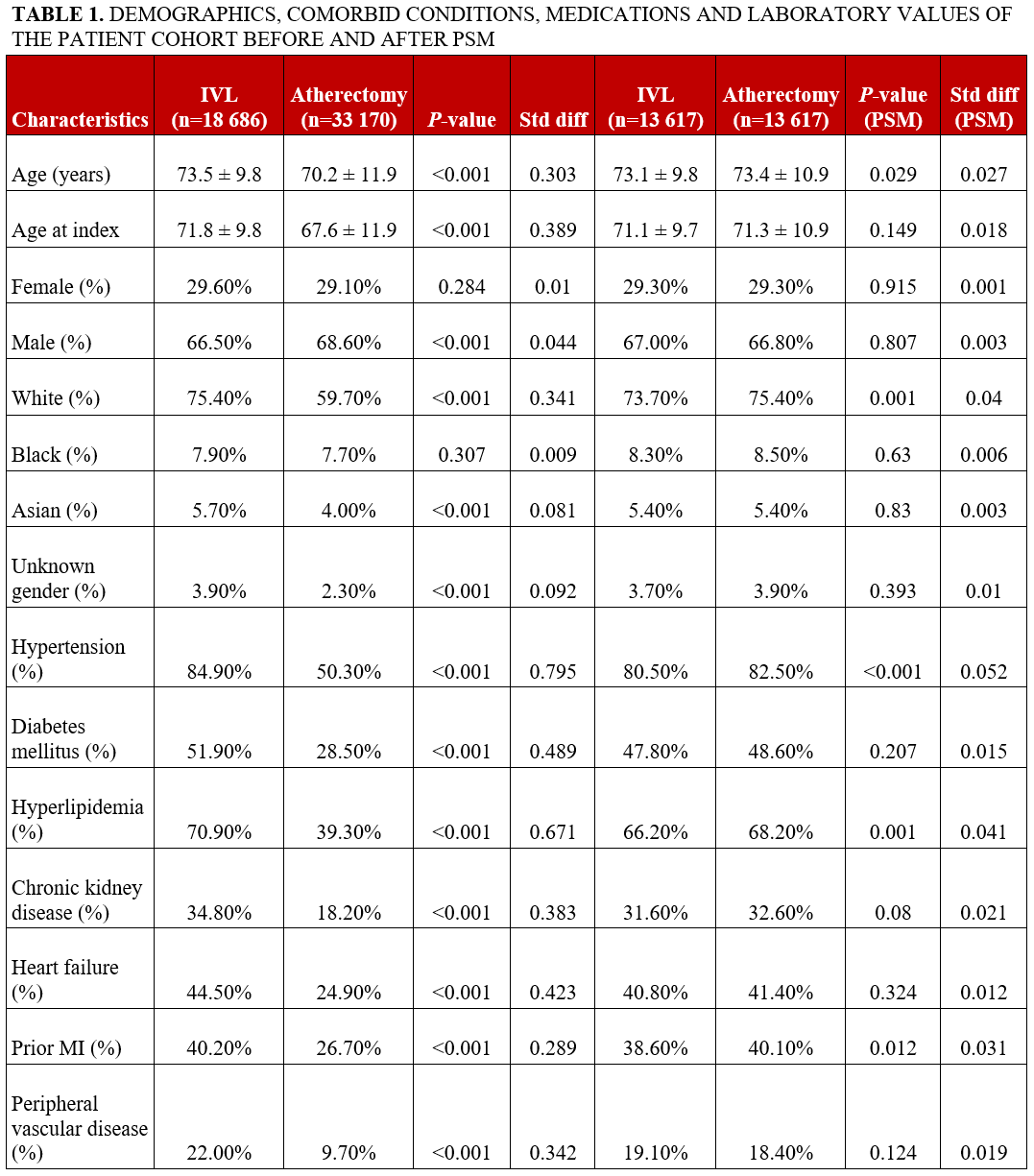
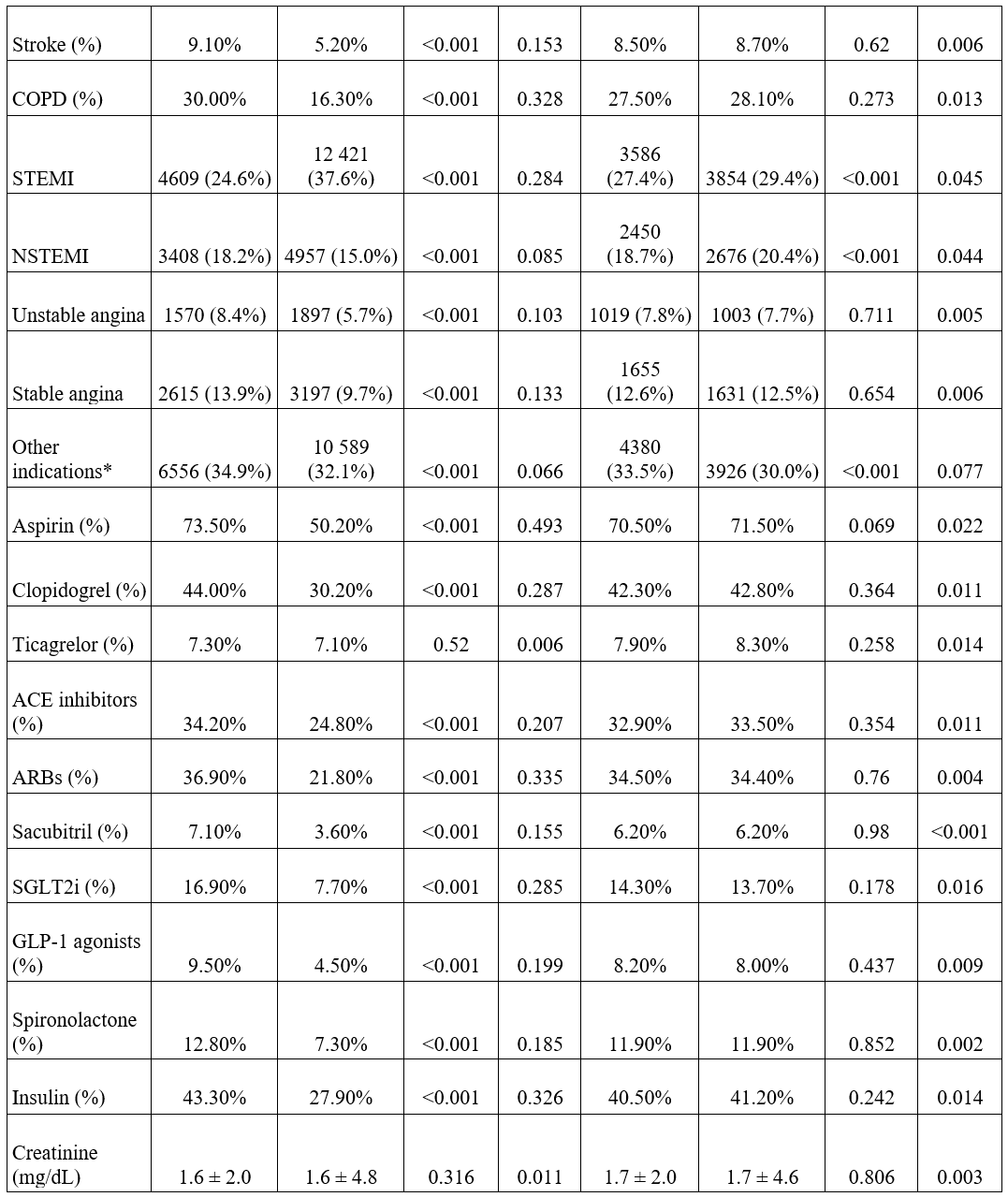
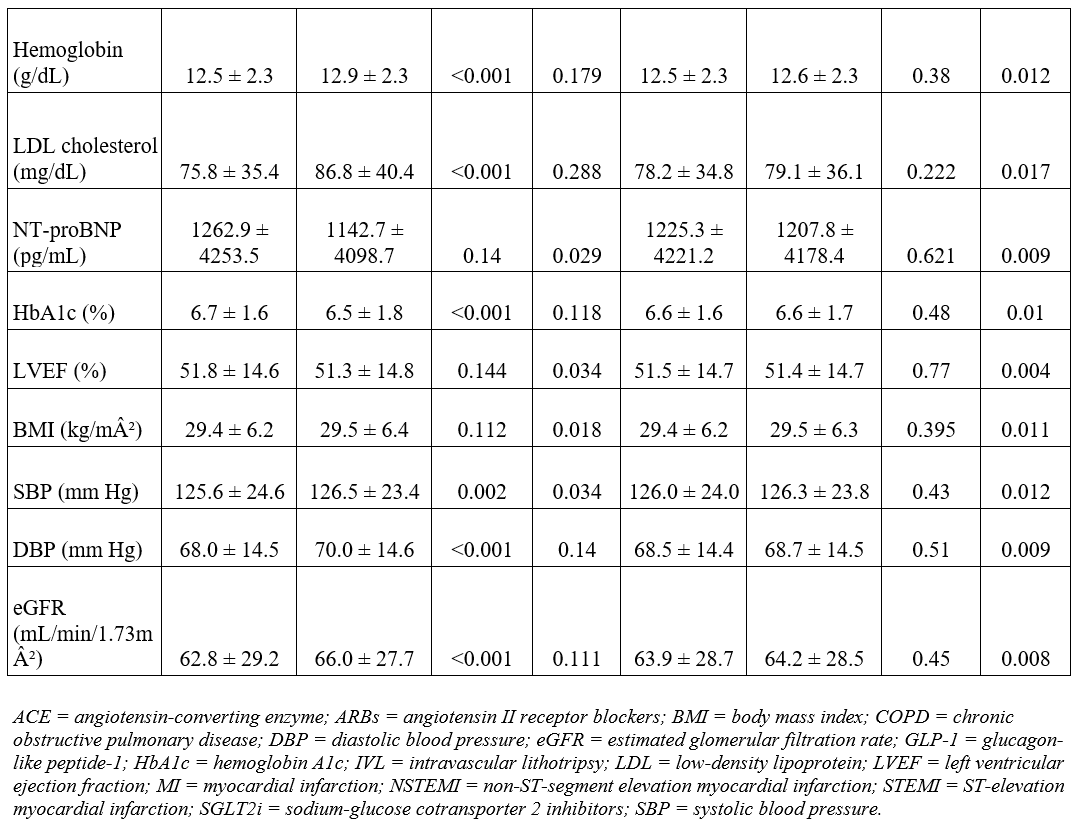
All baseline characteristics were assessed within the 12 months preceding the index date, defined as the date of the first PCI with lesion modification therapy. For laboratory variables, the most recent available values within that 12-month period were used. For comparison between 2 groups, we used chi-square tests for categorical variables and t-tests for continuous variables. Multivariable models were adjusted for relevant covariates based on clinical relevance and prior literature. All hypothesis tests were 2-tailed, with statistical significance defined as a P-value of less than 0.05. Two-tailed tests were chosen to account for the possibility of differences in either direction, reflecting an unbiased approach to hypothesis testing. Confidence intervals (95%) were reported to indicate the precision of the estimates. The database version utilized in this study included data available up to June 2025. Cohort construction, PSM, and outcome analyses were conducted using the integrated analytic tools within TriNetX. All statistical analyses were performed using the TriNetX real-world data analytics platform.5
Sensitivity analysis
E-values were calculated during the sensitivity analysis to evaluate the robustness of the findings to potential biases from unmeasured confounding or omitted covariates in both primary and secondary outcomes. E-value was calculated using the following formula8:
A higher E-value indicated that a more potent unmeasured cofounder would be necessary to invalidate the observed relationship and suggest a robust, causal link between the exposure and outcome. Given variability in definitions of MACE across the literature, a prespecified sensitivity analysis was performed using an expanded MACE definition that included cardiovascular mortality, new-onset MI, new-onset ischemic stroke, and new-onset HF. Outcomes were assessed at 1 month and 1 year following the index procedure using the same PSM cohort and analytic framework as the primary analysis.
Results
A total of 51 856 patients who underwent PCI with either coronary IVL or atherectomy were identified, including 18 686 (36.0%) in the coronary IVL group and 33 170 (64.0%) in the atherectomy group. Following PSM, 13 617 patients were retained in each cohort for the final analysis. The matched population had a mean age of 73.2 years, and 29.3% (3991 IVL, 3983 coronary atherectomy) of the patients were female. Baseline characteristics before and after PSM are summarized in Table 1.
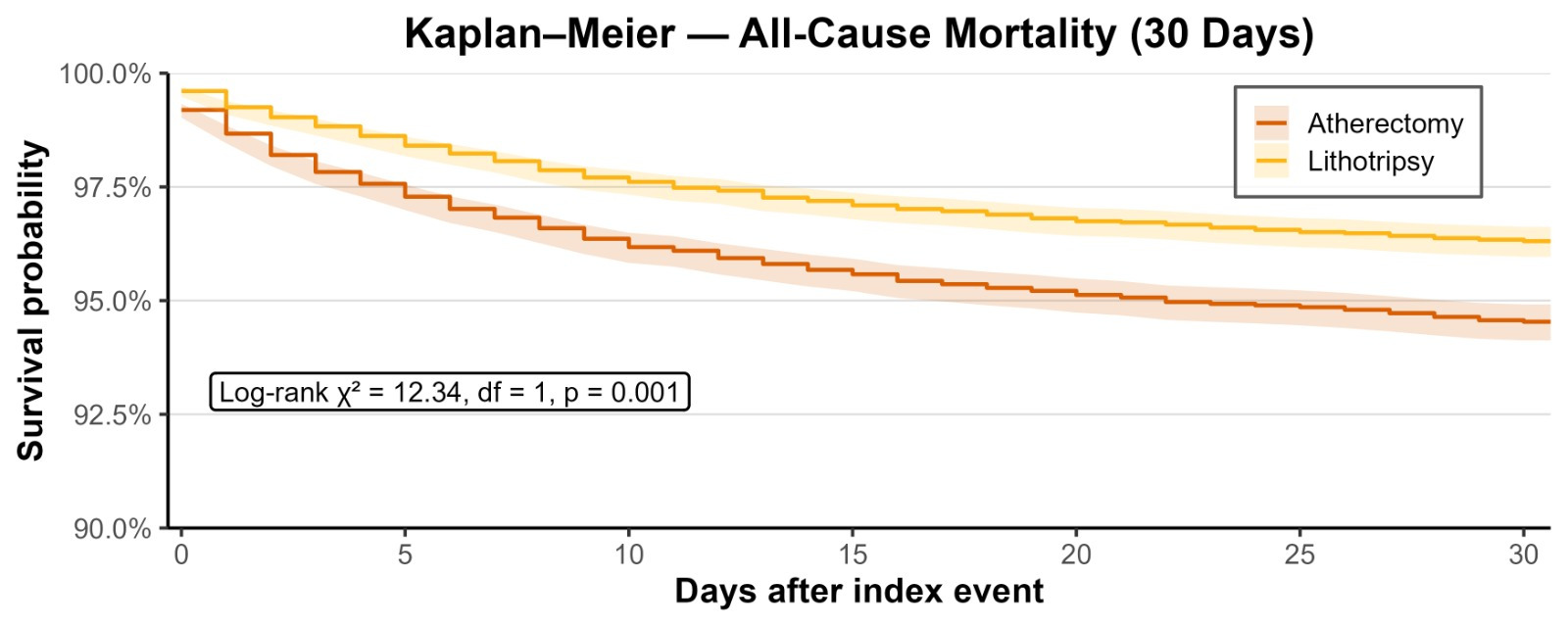
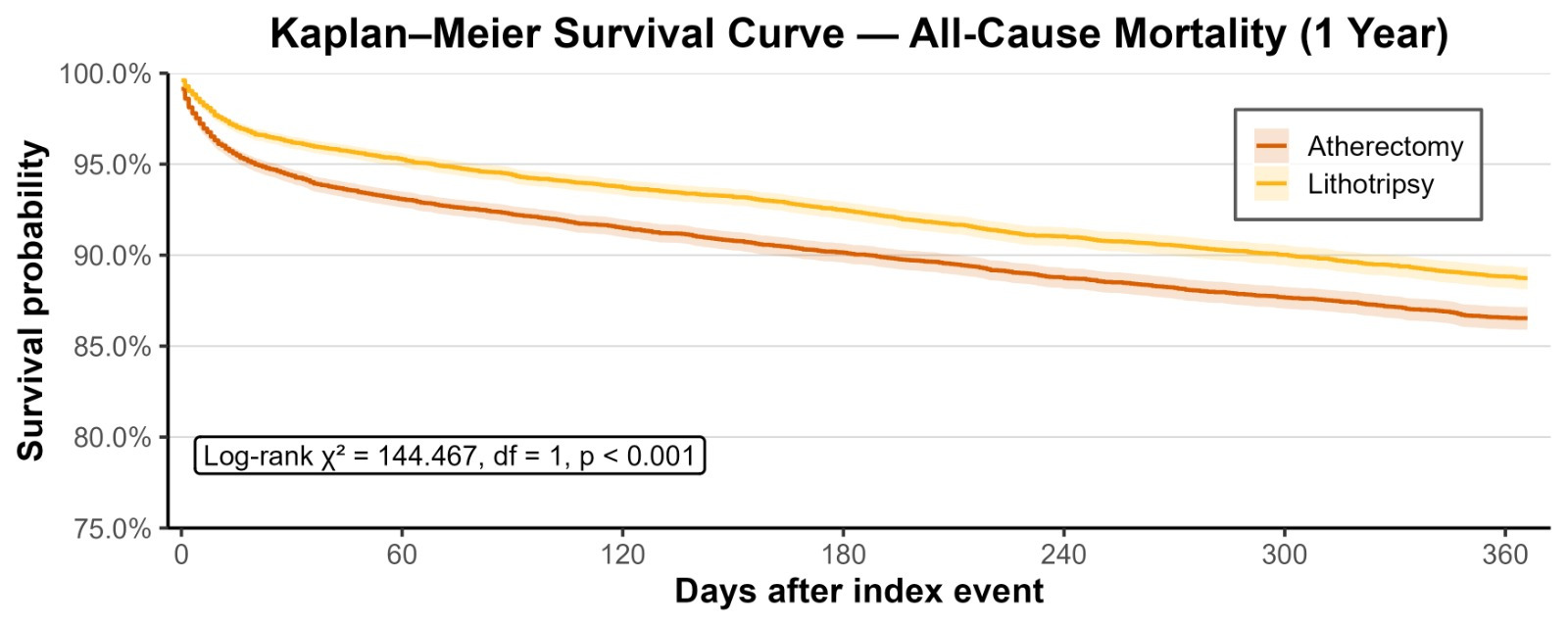
At 1-month follow-up, patients who underwent coronary IVL demonstrated a significantly lower risk of ACM (RR: 0.67; 95% CI, 0.60-0.75; P < .001), MACE (RR: 0.42; 95% CI, 0.36-0.48; P < .001), HFE (RR: 0.64; 95% CI, 0.57-0.72; P < .001) compared with those who underwent atherectomy. At the 1-year follow-up, this trend persisted, with coronary IVL group showing a significantly reduced risk of ACM (RR: 0.78; 95% CI, 0.73-0.84; P < .001), MACE (RR: 0.66; 95% CI, 0.60-0.72; P < .001), and HFE (RR: 0.79; 95% CI, 0.73-0.85; P < .001) compared with the control cohort who underwent atherectomy (Table 1). Figures 1 and 2 display Kaplan-Meier curves showing survival probability in the IVL group of patients at 1-month and 1-year follow-up, respectively, as well as a significant difference between the groups.
Safety outcomes
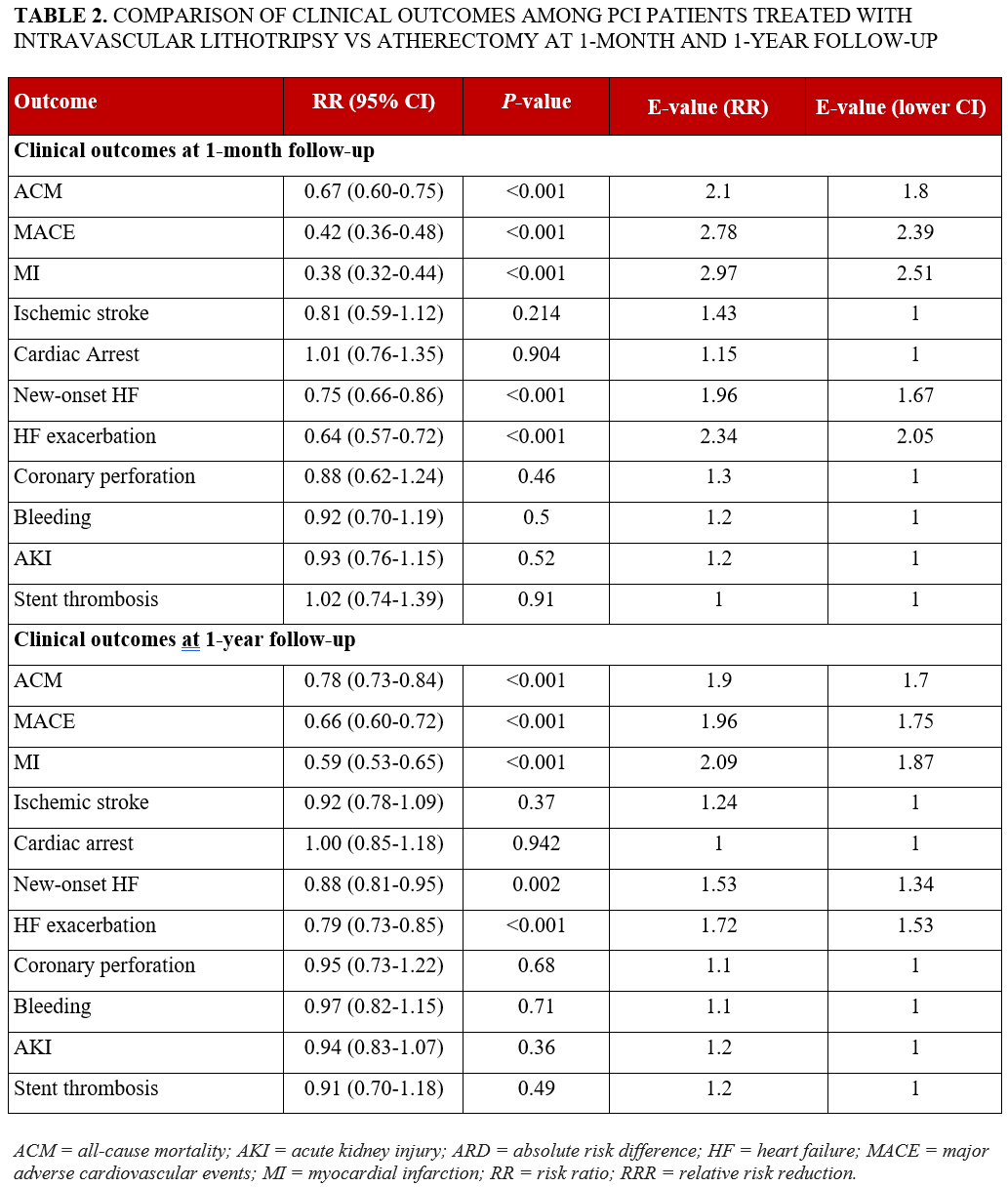
The risk of coronary perforation at 1 month (RR: 0.88; 95% CI, 0.62-1.24; P = .46) and at 1 year (RR: 0.95; 95% CI, 0.73-1.22; P = .68), risk of bleeding at 1 month (RR: 0.92; 95% CI, 0.70-1.19; P = .50) and at 1 year (RR: 0.97; 95% CI, 0.82-1.15; P = .71), risk of AKI at 1 month (RR: 0.93; 95% CI, 0.76-1.15; P = .52) and at 1 year (RR: 0.94; 95% CI, 0.83-1.07; P = .36), and risk ST at 1 month (RR: 1.02; 95% CI, 0.74-1.39; P = .91) and at 1 year (RR: 0.91; 95% CI, 0.70-1.18; P = .49) remained comparable between the groups (Table 2).
Sensitivity analyses
The E-values for 30-day outcomes were 2.1 (lower CI, 1.8) for ACM, 2.78 (2.39) for MACE, and 2.97 (2.51) for MI, indicating that an unmeasured confounder would need to be associated with both the exposure and each respective outcome by RRs of at least these magnitudes to fully explain away the observed associations. At 1 year, the E-values were 1.9 (1.7) for ACM, 1.96 (1.75) for MACE, and 2.09 (1.87) for MI, suggesting that moderately strong unmeasured confounding would be required to invalidate these associations.
A sensitivity analysis using the revised MACE definition inclusive of cardiovascular mortality was performed. It showed that IVL was associated with a significantly lower risk of MACE compared with atherectomy at both 1 month (RR: 0.39; 95% CI, 0.33-0.45; P < .01) and 1 year (RR: 0.71; 95% CI, 0.64-0.77; P < .01), consistent with the primary findings.
Discussion
This propensity-matched analysis of more than 27 000 patients constitutes the largest head-to-head comparison of IVL and atherectomy for calcified coronary artery disease. IVL was associated with significantly lower risks of ACM, MACE, and HFE at both 30 days and 1 year, without evidence of excess procedural risk. These findings extend earlier evidence by suggesting that the procedural and short-term safety advantages of IVL documented in single-arm trials and smaller comparative registries may translate into clinically meaningful outcomes at intermediate follow-up in a broadly representative real-world population.9
Context within existing literature
The evidence base for calcium modification in PCI has expanded substantially since regulatory approval for IVL, yet the comparative clinical effectiveness of IVL and coronary atherectomy has remained incompletely characterized.9 The Disrupt CAD program established the safety and single-arm efficacy of IVL as a device class, culminating in the pooled analysis of 628 patients that reported a 92.4% procedural success rate and a 30-day MACE rate of 7.3%.3 One-year data from Disrupt CAD III also confirmed the durability of this safety profile, with composite MACE of 13.8% and ischemia-driven target lesion revascularization of 6.0%.10 However, as these trials lacked an active comparator arm, they could not address whether IVL confers superiority or equivalence relative to atherectomy—a critical evidence gap that the present study directly addresses by providing the largest head-to-head outcome comparison to date.9
To date, the ROTA.shock and ROLLER COASTR-EPIC22 trials provide the only randomized comparative data in this domain.11,12 ROTA. shock, comprising 70 patients, demonstrated IVL noninferiority to rotational atherectomy (RA) for minimum stent area by optical coherence tomography (OCT) with comparable procedural metrics.11 An OCT substudy from the same trial revealed distinct calcium modification signatures: RA generated greater acute luminal gain while IVL produced a larger number of longer calcium fractures per cross-section—mechanistic differences that may govern longer-term outcomes.13 The ROLLER COASTR-EPIC22 trial, which enrolled 171 patients across 3 arms (RA, IVL, and excimer laser coronary angioplasty), similarly found no significant differences in stent expansion or 1-year clinical outcomes, reinforcing procedural equivalence among these modalities in selected anatomies.12
Although atherectomy has a long track record, these trials were limited by small sample sizes and surrogate imaging endpoints, and none demonstrated a definitive clinical outcome advantage for either modality.11-13 Our study extends these findings by showing that procedural advantages previously reported for IVL—including better stent expansion, fewer embolic events, and less vascular trauma—appear to translate into lower mortality and fewer adverse cardiovascular events over time.
Furthermore, real-world registry data from the Blue Cross Blue Shield of Michigan Cardiovascular Consortium (BMC2) offered a broader real-world lens: among 42 440 consecutive PCI cases at 48 Michigan hospitals, IVL use increased from under 0.04% to 4.28% between January 2021 and June 2022, eventually surpassing atherectomy statewide.14 In-hospital MACE and procedural success rates were comparable in unadjusted analyses, though the absence of propensity adjustment limited causal inference.15 Our findings add to this growing body of real-world evidence by applying rigorous PSM across a substantially larger multicenter cohort, enabling a more robust comparison of clinical outcomes between IVL and atherectomy than has previously been possible.
Interestingly, 2 independent systematic reviews and meta-analyses published in 2025 have provided the most rigorous comparative synthesis to date.15,16 Suruagy-Motta et al demonstrated shorter procedure times, lower contrast volumes, higher stent deployment success, and reduced target lesion revascularization with IVL, without significant MACE differences.15 Moghadam et al similarly found comparable MACE rates but a significantly lower risk of coronary perforation (OR, 0.43; 95% CI, 0.32-0.57) and slow-flow (OR, 0.34; 95% CI, 0.14-0.79) with IVL.16 The mortality and MACE advantages observed in the present cohort is substantially larger than any prior comparative analysis and extend these pooled procedural findings into the clinical outcomes domain, suggesting that the safety benefits of IVL reported in smaller studies translate into meaningful reductions in mortality and adverse events over time. This signal is especially pertinent in older patients and those with pre-existing chronic kidney disease or complex coronary anatomy, populations well represented in this cohort.
The ECLIPSE trial deserves particular attention in this context.17 This multicenter US randomized trial (n = 2005) found that routine orbital atherectomy before drug-eluting stent implantation did not reduce target vessel failure at 1 year vs conventional balloon angioplasty (11.5% vs 10.0%; HR, 1.16; 95% CI, 0.87-1.54; P = .28), while slow-flow rates were significantly higher and 30-day cardiac mortality was numerically elevated in the atherectomy arm.17 Notably, the trial excluded lesions deemed anatomically unsuitable for balloon preparation—precluding generalization to the most complex substrates—and was conducted in a setting of unusually high intravascular imaging use (62%), which may have optimized outcomes in both arms.17 Nonetheless, these findings challenge the assumption that atherectomy-mediated plaque debulking reliably translates into clinical benefit and underscore the importance of evaluating IVL as a potentially safer alternative in a broad patient population.
Mechanistic considerations
The differential outcome patterns observed in this study may plausibly reflect fundamental mechanistic distinctions between the 2 strategies. IVL transmits pulsatile acoustic pressure waves circumferentially through tissue planes without generating frictional heat or requiring high rotational speeds, selectively fracturing calcified plaque at both intimal and medial depths while preserving the endothelium and adventitia.9,18,19 Operating over any coronary guidewire, IVL avoids the eccentric ablation pattern that can arise from guidewire deflection during atherectomy, a recognized contributor to asymmetric calcium modification and incomplete lesion preparation.9,18,19
Atherectomy, by contrast, ablates superficial intimal calcium through mechanical abrasion. This process generates microemboli capable of occluding distal microvessels, precipitating slow flow or no-reflow-phenomena that occur more frequently with RA than IVL across pooled studies and that may disproportionately affect patients with impaired ventricular function, advanced renal insufficiency, or diffuse multivessel disease.9,18,19 The convergent safety findings in the present cohort—similar perforation, AKI, and ST rates despite the substantially higher pre-match comorbidity burden in IVL-treated patients—are consistent with the favorable safety signals reported across the Disrupt CAD program and comparative meta-analyses.3,15,16
Clinical implications
Current ACC/AHA/SCAI (American College of Cardiology/American Heart Association/Society for Cardiovascular Angiography & Interventions) 2021 Coronary Artery Revascularization Guidelines and 2018 ESC/EACTS (European Society of Cardiology/European Association for Cardio-Thoracic Surgery) Myocardial Revascularization Guidelines emphasize the importance of adequate lesion preparation in heavily calcified coronary disease to optimize stent delivery and expansion but do not endorse 1 device over another because of limited comparative evidence.20,21 The findings of this study help address this evidence gap and, if confirmed in randomized trials, may inform future updates to revascularization guidelines by providing comparative outcome data between coronary IVL and atherectomy at both early and late follow-up.
Limitations
Nevertheless, this study has certain limitations. First and most critically, residual selection bias is likely present: unmeasured confounders—including coronary calcium burden, lesion morphology, SYNTAX score, surgical turndown status, intravascular imaging use, and operator experience—are not captured by TriNetX and cannot be incorporated into the propensity model, irrespective of the E-values reported. Second, atherectomy modalities were analyzed as a composite because reliable stratification by rotational vs orbital technique was not feasible using ICD-10-CM codes alone. Third, granular angiographic, procedural, and anatomical data (reference vessel diameter, lesion length, calcium arc) were unavailable. Fourth, cause-specific mortality could not be ascertained. Fifth, the database is predominantly derived from insured populations in acute care settings, potentially limiting generalizability. Longer follow-up is required to establish the durability of the observed benefits. Lastly, we acknowledge that the study relies on billing codes, which may be subject to local hospital coding variation.
Conclusions
Among propensity-matched patients with calcified coronary artery disease undergoing PCI, IVL was associated with significantly lower rates of ACM, MACE, and HFE compared with atherectomy at both 30-day and 1-year follow-up, with comparable procedural safety. Given the inherent risk of residual selection bias in this observational analysis, these results are hypothesis-generating rather than definitive. Adequately powered randomized controlled trials directly comparing IVL and atherectomy are required before IVL can be formally endorsed as the preferred calcium modification strategy.
Affiliations and Disclosures
Vikash Jaiswal, MD1; Muhammad Hanif, MD2; Yusra Mashkoor, MBBS3; Aymen Ahmed, MBBS1; Gregory Mishkel, MD1; Jonathan Rosenberg, MD1; Mark J. Ricciardi, MD1; Arman Qamar, MD, MPH1
From the 1Endeavor Center for Cardiovascular Intervention Outcomes Research and Evaluation (ECCORE), Section of Interventional Cardiology, Endeavor Health Cardiovascular Institute, Glenview, Illinois; 2Department of Internal Medicine, SUNY Upstate Medical University, Syracuse, New York; 3Department of Internal Medicine, Dow University of Health Sciences, Karachi, Pakistan.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Ethical approval: Institutional review board approval was exempted by SUNY Upstate
Institutional Review Board, given that aggregate deidentified data was used from a research network database. These study findings are reported per the Strengthening the Reporting of Observational Studies in Epidemiology guideline for cohort studies. To ensure accuracy and completeness, TriNetX performs an internal check on data quality during every refresh.
Data availability statement: Data is provided within the article and supplementary material.
Address for correspondence: Arman Qamar, MD, MPH, Endeavor Center for Cardiovascular Intervention Outcomes Research and Evaluation (ECCORE), Section of Interventional Cardiology, Endeavor Health Cardiovascular Institute, Glenview, IL 60201, USA. Email: arman.qamar@endeavorhealth.org; aqamar@alumni.harvard.edu; X: @AqamarMD
References
- Madhavan MV, Tarigopula M, Mintz GS, Maehara A, Stone GW, Généreux P. Coronary artery calcification: pathogenesis and prognostic implications. J Am Coll Cardiol. 2014;63(17):1703-14. doi:10.1016/j.jacc.2014.01.017
- Hemetsberger R, Abdelghani M, Toelg R, et al. Impact of coronary calcification on clinical outcomes after implantation of newer-generation drug-eluting stents. J Am Heart Assoc. 2021;10(12):e019815. doi:10.1161/JAHA.120.019815
- Kereiakes DJ, Di Mario C, Riley RF, et al. Intravascular lithotripsy for treatment of calcified coronary lesions: patient-level pooled analysis of the Disrupt CAD studies. JACC Cardiovasc Interv. 2021;14(12):1337-1348. doi:10.1016/j.jcin.2021.04.015
- Kaul A, Dhalla PS, Bapatla A, et al. Current treatment modalities for calcified coronary artery disease: a review article comparing novel intravascular lithotripsy and traditional rotational atherectomy. Cureus. 2020;12(10):e10922. doi:10.7759/cureus.10922
- TriNetX. Accessed June 22, 2024. https://trinetx.com/
- US Department of Health and Human Services. Federal policy for the protection of human subjects. 45 CFR §46. Revised January 19, 2017. Accessed June 06, 2025. https://www.hhs.gov/ohrp/regulations-and-policy/regulations/45-cfr-46/index.html
- Ghaferi AA, Schwartz TA, Pawlik TM. STROBE reporting guidelines for observational studies. JAMA Surg. 2021;156(6):577-578. doi:10.1001/jamasurg.2021.0528
- Chung WT, Chung KC. The use of the E-value for sensitivity analysis. J Clin Epidemiol. 2023;163:92-94. doi:10.1016/j.jclinepi.2023.09.014
- Riley RF, Patel MP, Abbott JD, et al. SCAI expert consensus statement on the management of calcified coronary lesions. J Soc Cardiovasc Angiogr Interv. 2024;3(2):101259. doi:10.1016/j.jscai.2023.101259
- Kereiakes DJ, Hill JM, Shlofmitz RA, et al; Disrupt CAD III Investigators. Intravascular lithotripsy for treatment of severely calcified coronary lesions: 1-year results from the Disrupt CAD III study. J Soc Cardiovasc Angiogr Interv. 2022;1(1):100001. doi:10.1016/j.jscai.2021.100001
- Blachutzik F, Meier S, Weissner M, et al; ROTA.shock Investigators. Coronary intravascular lithotripsy and rotational atherectomy for severely calcified stenosis: results from the ROTA.shock trial. Catheter Cardiovasc Interv. 2023;102(5):823-833. doi:10.1002/ccd.30815
- Jurado-Román A, Gómez-Menchero A, Rivero-Santana B, et al. Rotational atherectomy, lithotripsy, or laser for calcified coronary stenosis: the ROLLER COASTR-EPIC22 trial. JACC Cardiovasc Interv. 2025;18(5):606-618. doi:10.1016/j.jcin.2024.11.012
- Blachutzik F, Meier S, Weissner M, et al. Comparison of coronary intravascular lithotripsy and rotational atherectomy in the modification of severely calcified stenoses. Am J Cardiol. 2023;197:93-100. doi:10.1016/j.amjcard.2023.02.028
- Sukul D, Seth M, Madder RD, et al. Contemporary trends and outcomes of intravascular lithotripsy in percutaneous coronary intervention: insights from BMC2. JACC Cardiovasc Interv. 2024;17(15):1811-1821. doi:10.1016/j.jcin.2024.04.039
- Suruagy-Motta RFO, Cabeça LS, Da Silva AMP, et al. Intravascular lithotripsy versus rotational atherectomy for calcified coronary lesions: a systematic review and an updated meta-analysis of clinical outcomes. Catheter Cardiovasc Interv. 2025;106(1):563-572. doi:10.1002/ccd.31591
- Moghadam AS, Kakavand N, Shirmard FO, et al. Intravascular lithotripsy versus rotational atherectomy in the management of calcific coronary lesions: a systematic review and meta-analysis. Catheter Cardiovasc Interv. 2025;106(2):1142-1152. doi:10.1002/ccd.31664
- Kirtane AJ, Généreux P, Lewis B, et al; ECLIPSE Investigators. Orbital atherectomy versus balloon angioplasty before drug-eluting stent implantation in severely calcified lesions eligible for both treatment strategies (ECLIPSE): a multicentre, open-label, randomised trial. Lancet. 2025;405(10486):1240-1251. doi:10.1016/S0140-6736(25)00450-7
- Gupta A, Shrivastava A, Dugal JS, et al. Coronary intravascular lithotripsy in contemporary practice: challenges and opportunities in coronary intervention. Ther Adv Cardiovasc Dis. 2024;18:17539447241263444. doi:10.1177/17539447241263444
- Gruslova AB, Inanc IH, Cilingiroglu M, Katta N, Milner TE, Feldman MD. Review of intravascular lithotripsy for treating coronary, peripheral artery, and valve calcifications. Catheter Cardiovasc Interv. 2024;103(2):295-307. doi:10.1002/ccd.30933
- Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: executive summary: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation. 2022;145(3):e4-e17. doi:10.1161/CIR.0000000000001039
- Neumann FJ, Sousa-Uva M, Ahlsson A, et al; ESC Scientific Document Group. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87-165. doi: 10.1093/eurheartj/ehy394