Clinical Outcomes After Transcatheter Aortic Valve Replacement by Sex and Subtype of Low-Flow Aortic Stenosis
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/26.00096. Epub May 4, 2026.
Abstract
Objectives. Prior studies suggest sex differences in clinical outcomes following transcatheter aortic valve replacement (TAVR) in patients with severe aortic stenosis (AS). The authors hypothesized that among patients with low-flow aortic stenosis (LFAS), outcomes differ based on sex and subtype of LFAS: classical (cLFLG), paradoxical (pLFLG), and high gradient (LFHG).
Methods. This single-center, observational, longitudinal cohort study included adults with LFAS who underwent TAVR. Differences in clinical outcomes (all-cause mortality, heart failure hospitalization [HFH], and myocardial infarction [MI]) by subtype of LFAS within each sex were examined via Kaplan Meier curves. Log rank P-values were reported.
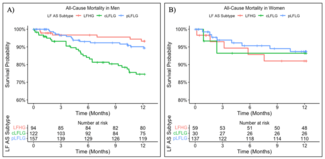
Results. A total of 599 patients with LFAS were followed for 12 months after TAVR. Of the male patients (n = 373), 25.2% had LFHG, 32.7% cLFLG, and 42.1% pLFLG AS. Of females (n = 226), 26.1% had LFHG, 13.3% cLFLG, and 60.6% pLFLG AS. Event rates in males were 13.1% all-cause mortality, 6.2% HFH, and 0.8% MI. Event rates in females included 6.6% all-cause mortality, 8.0% HFH, and 0 MI. In males, the LFHG vs cLFLG vs pLFLG subtypes demonstrated significant differences in time to all-cause mortality (P < .001), HFH (P = .014), and MI (P = .045).
Conclusions. In this cohort, clinical outcomes in males significantly differed by subtype of LFAS, whereas these differences were not found in females.
Introduction
Low-flow aortic stenosis (LFAS) represents a heterogeneous subset of severe AS characterized by reduced stroke volume (SV) in the presence of severe valve disease. While transcatheter aortic valve replacement (TAVR) has augmented the treatment of AS, outcomes in LFAS patients remain less well-defined, with emerging evidence suggesting that clinical trajectories may vary by both hemodynamic subtype1 and sex.2 Sex-based differences in valvular pathophysiology and cardiac remodeling are increasingly recognized, yet their interaction with LFAS subtypes and post-TAVR outcomes has not been comprehensively characterized. Understanding whether sex modifies the prognostic significance of hemodynamic subtypes is important for risk stratification, surveillance, and prognostication in this patient population.
Methods
This single-center observational analysis included adult patients with LFAS (aortic valve area [AVA] ≤1.0 cm2 and stroke volume index [SVi] ≤35 cm/m2) who underwent TAVR. LFAS subtypes were defined by left ventricular ejection fraction (LVEF) and mean gradient (MG): classical low-flow low-gradient (cLFLG) included an LVEF of less than 50% and an MG of less than 40 mm Hg, paradoxical low-flow low-gradient (pLFLG) included an LVEF greater than or equal to 50% and an MG of less than 40 mm Hg, and low-flow high-gradient (LFHG) included an MG of greater than or equal to 40 mm Hg irrespective of LVEF. Baseline demographic and clinical variables were collected and compared by sex. Continuous variables were compared via Wilcoxon rank sum test and categorical variables were compared via chi-square test. Outcomes at 12 months post-TAVR were collected for all-cause mortality, heart failure hospitalization (HFH), and myocardial infarction (MI). These were assessed by sex and subtype of LFAS via Kaplan-Meier curves, and log-rank P-values were reported.
The authors confirm this study was approved by the Mount Sinai Hospital institutional review board, with a waiver of informed consent.
Results
A total of 599 patients (373 males and 226 females) with LFAS were followed for 12 months after TAVR. Baseline demographic, clinical, and procedural characteristics are displayed in the Table. The baseline differences between sexes align with established patterns in AS. In our study, males presented with higher rates of comorbidities (coronary artery disease, prior revascularization, and diabetes), consistent with their generally higher cardiovascular risk profile. Distribution of LFAS subtype was significantly different by sex (P < .001), with pLFLG more common in females (60.6% [n = 137] vs 42.1% [n = 157]) and cLFLG more common in males (32.7% [n = 122] vs 13.3% [n = 30]). Event rates in males were 13.1% (n = 49) all-cause mortality, 6.2% (n = 23) HFH, and 0.8% (n = 3) MI. Event rates in females included 6.6% (n = 15) all-cause mortality, 8.0% (n = 18) HFH, and 0% (n = 0) MI. In males, LFAS subtypes demonstrated significant differences in time to all-cause mortality (P < .001), HFH (P = .001), and MI (P = .045) (Figure A). All events were highest in the cLFLG group for males; however, in females, no significant differences were observed in any outcome when compared by LFAS subtypes (Figure B).
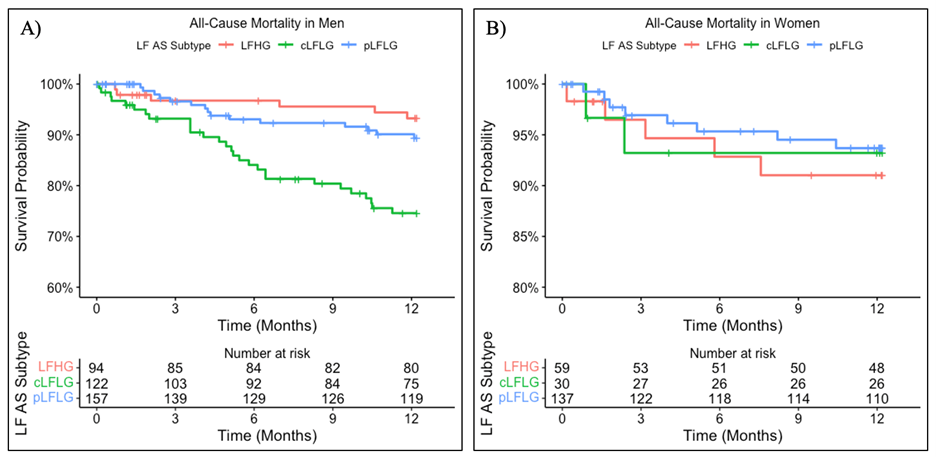
Discussion
Our significant findings of the impact of LFAS subtype on outcomes in males, but not females, may reflect differences in remodeling in response to AS. Males are more likely to undergo concentric hypertrophy in AS, while females tend towards concentric remodeling.3 It is possible that without compensatory, adaptive remodeling mechanisms, males may progress earlier into progressive LV dysfunction associated with poorer outcomes. This is supported by our analysis, which showed that cLFLG was the highest-risk subtype in males across all measured outcomes. The higher event rates in male patients with cLFLG suggest that systolic dysfunction portends worse outcomes regardless of potential reverse remodeling after TAVR. It is also important to note the significant presence of coronary artery disease in males compared with females. The inability to generate a gradient in males may be more related to infarcted myocardium rather than progressive myocardial fibrosis. Further studies may evaluate how the etiology of LV dysfunction in conjunction with remodeling in response to AS impacts outcomes.
Conversely, remodeling in response to severe AS in females is characterized by diffuse fibrosis, more progressive course, and intact systolic function.4 The low-flow state in the population of interest is more often reflective of diastolic dysfunction due to changes in ventricular geometry, filling, and afterload. Because LFAS subtype is dependent on systolic parameters (ie, ejection fraction and mean gradient), these are less likely to capture the nuanced spectrum of diastolic disease.
Limitations to this study include the inherent shortcomings of a retrospective study without corelab adjudications of echocardiographic parameters and short-term (12 months) follow-up. Low event rates further limit the study. Future studies can examine how the studied factors might shape longer-term outcomes.
Conclusions
Overall, our study demonstrates that for males, LFAS subtype may be a useful prognostic tool for outcomes following TAVR. However, for females, future research is needed to understand the prognostic implications of low-flow state.
Affiliations and Disclosures
Eileen Galvani, MD1; Oludamilola Akinmolayemi, MD, MPH2; Srivatsan Swaminathan, BS3; Carlo Mannina, MD2; Sahil Khera, MD, MPH2; Lucy Safi, DO2; Gilbert H.L. Tang, MD, MSc, MBA2; Samin K. Sharma, MD2; Annapoorna Kini, MD2; Stamatios Lerakis, MD, PhD2
From the 1Department of Medicine, Mount Sinai Hospital, New York, New York; 2Mount Sinai Fuster Heart Hospital, Icahn School of Medicine at Mount Sinai, New York, New York; 3Icahn School of Medicine at Mount Sinai, New York, New York.
Preliminary results were published as an abstract: https://www.jacc.org/doi/10.1016/S0735-1097(25)02801-3
Disclosures: Dr Tang has received speaker's honoraria and served as a physician proctor, consultant, advisory board member, TAVR publications committee member, RESTORE study steering and screening committee member, APOLLO trial screening committee member, and IMPACT MR steering committee member for Medtronic; has received speaker's honoraria and served as a physician proctor, consultant, advisory board member, ENVISION trial screening committee member, and TRILUMINATE trial anatomic eligibility and publications committee member for Abbott Structural Heart; and has served as an advisory board member for Boston Scientific, Anteris, Philips, Edwards Lifesciences, Peija Medical, and Shenqi Medical Technology. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Address for correspondence: Stamatios Lerakis, MD, PhD, Mount Sinai Fuster Heart Hospital, Icahn School of Medicine at Mount Sinai, 1 Gustave L. Levy Pl, Box 1030, New York, NY 10029, USA. Email: stamatios.lerakis@mountsinai.org
References
1. Wagener M, Reuthebuch O, Heg D, et al. Clinical outcomes in high-gradient, classical low-flow, low-gradient, and paradoxical low-flow, low-gradient aortic stenosis after transcatheter aortic valve implantation: a report from the SwissTAVI registry. J Am Heart Assoc. 2023;12(12):e029489. doi: 0.1161/JAHA.123.029489
2. Dismorr M, Granbom-Koski M, Ellfors E, et al. Sex differences and long-term clinical outcomes after transcatheter aortic valve replacement: a SWEDEHEART study. Am Heart J. 2024;277:27-38. doi:10.1016/j.ahj.2024.07.018
3. Treibel TA, Badiani S, Lloyd G, Moon JC. Multimodality imaging markers of adverse myocardial remodeling in aortic stenosis. JACC Cardiovasc Imaging. 2019;12(8 Pt 1):1532-1548. doi:10.1016/j.jcmg.2019.02.034
4. Hariri EH, El Halabi J, Kassis N, et al. Sex differences in the progression and long-term outcomes of native mild to moderate aortic stenosis. JACC Cardiovasc Imaging. 2024;17(1):1-12. doi:10.1016/j.jcmg.2023.06.006