News
Angel Medical Systems Announces ALERTS Pivotal Study For Implantable Cardiac Monitor & Alert System
11/03/2008
For Immediate Release:
Media Contact:
Jonathan Harwood
Chief Operating Officer
Angel Medical Systems
1163 Shrewsbury Avenue
Shrewsbury, NJ 07702
(732) 542-3111
jharwood@angel-med.com
New study will evaluate efficacy of an implantable device, designed to reduce time-to-door and improve survival rates from myocardial infarction (MI) in high-risk patients.
Washington, DC, October 13, 2008: AngelMed announced today at the Transcatheter Cardiovascular Therapeutics symposium its initiation of a Phase II Pivotal US trial for the AngelMed Guardian implantable cardiac monitor and alert system. Designed to reduce time to presentation by detecting significant changes in the heart's electrical signal and then alerting patients to seek medical attention, the AngelMed Guardian System is commercially available in Brazil and available for investigational use only in the US.
AngelMed is actively seeking up to 50 clinical sites nationwide to participate in the trial.
"We have worked closely with some of the world's leading experts in heart attacks, like C. Michael Gibson, M.S., M.D., from Harvard University; David Holmes, MD, of the Mayo Clinic; and Mitchell Krucoff, MD of Duke University to design a study for evaluating the potential of the AngelMed Guardian System," says Jill Schweiger, AngelMed's Vice President of Clinical and Regulatory Affairs. "The study design is meant to test the system's ability to alert patients of significant ST shift events in order to reduce the incidence of death and new Q-wave MI and decrease the lapse of greater than 2 hours before presentation for life-threatening thrombotic coronary occlusions, such as those caused by vulnerable plaque ruptures."
According to the American Heart Association, one of every five deaths in the US is attributable to coronary heart disease. Further, fifty percent of heart-attack fatalities occur within 1 hour of symptom onset and occur before the patient even reaches the hospital.
"Experimental and clinical studies have shown that most of the damage to the yocardium occurs during the first two hours after coronary occlusion," says AngelMed's Chief Operating Officer Jonathan Harwood. "We've designed the device to warn patients of this and other cardiac events hours --- perhaps days -- before they occur."
The AngelMed Guardian System is comprised of an internal implantable device about the size of a standard pacemaker with a lead into the heart, an external telemetry device, and a programmer that aids physicians in evaluating heart signals.
"When someone has a heart attack, it is typically the result of a blood clot closing one of the three major coronary arteries. When this happens, there is a shift in the ST segment of the heart signal caused by the electrical difference between the portion of the heart muscle fed by the closed artery and the rest of the heart that is still receiving oxygen," adds AngelMed's CEO, David Fischell, Ph.D. "The ALERTS trial is designed to test the Guardian's ability to detect this electrical shift in a clinical setting and then provide an early warning to patients that could potentially save their lives."
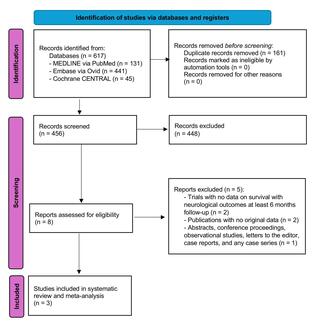
The ALERTS trial is currently underway and will be conducted in up to 50 centers across the United States. To participate in the ALERTS study, patients must meet various inclusion criteria. For more information on the AngelMed Guardian system or the LERTS study protocol, go to https://www.angel-med.com