Occluded Left Circumflex Artery in a Patient With the de Winter Sign on Electrocardiography
© 2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
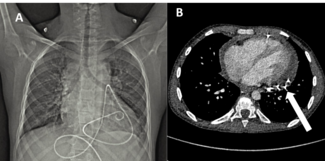
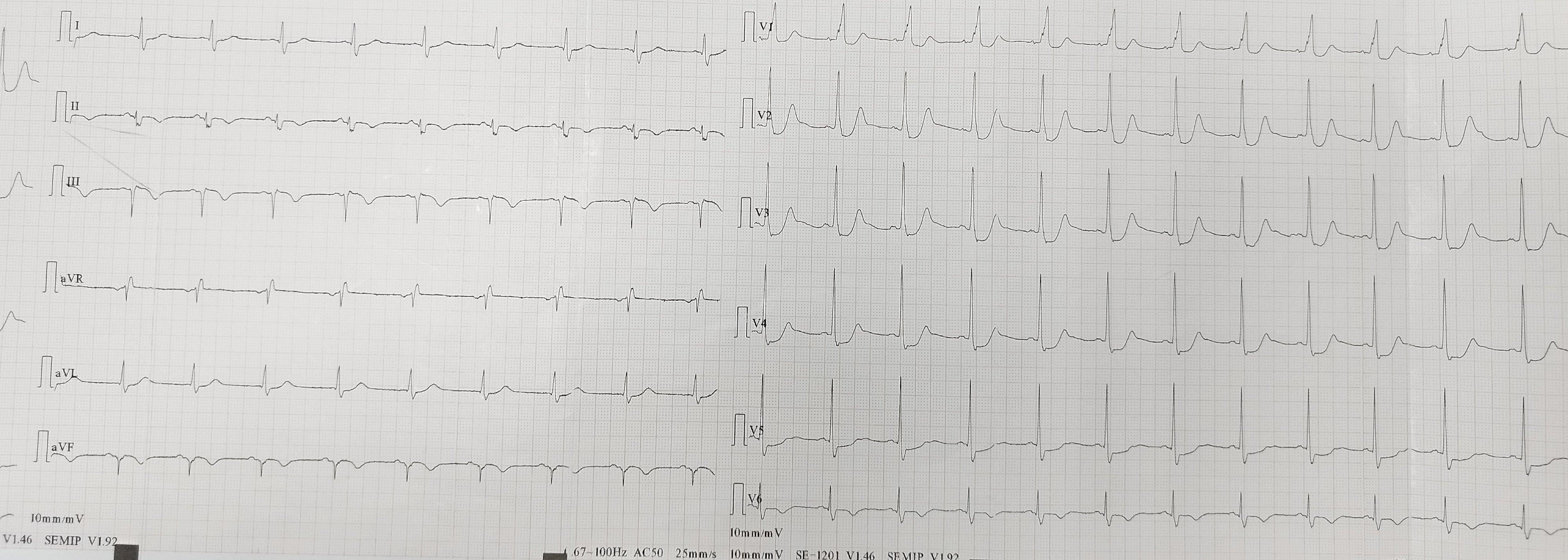
A 61-year-old man with hypertension and no other cardiovascular risk factors presented to a district hospital with chest pain. Electrocardiogram (ECG) demonstrated the de Winter sign (upsloping ST-segment depression at the J point in leads V1-V4, continuing into tall, positive, and symmetrical T waves) (Figure 1). Given the apparent high-risk non-ST-elevation myocardial infarction (NSTEMI), he was transferred to our institute for emergency coronary angiography. Transthoracic echocardiography showed mildly reduced left ventricular systolic function.
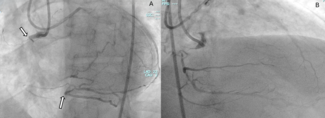
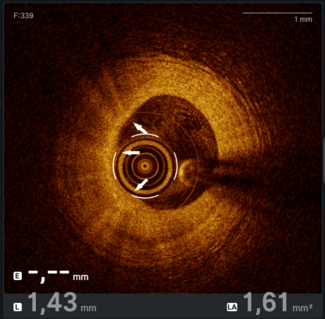
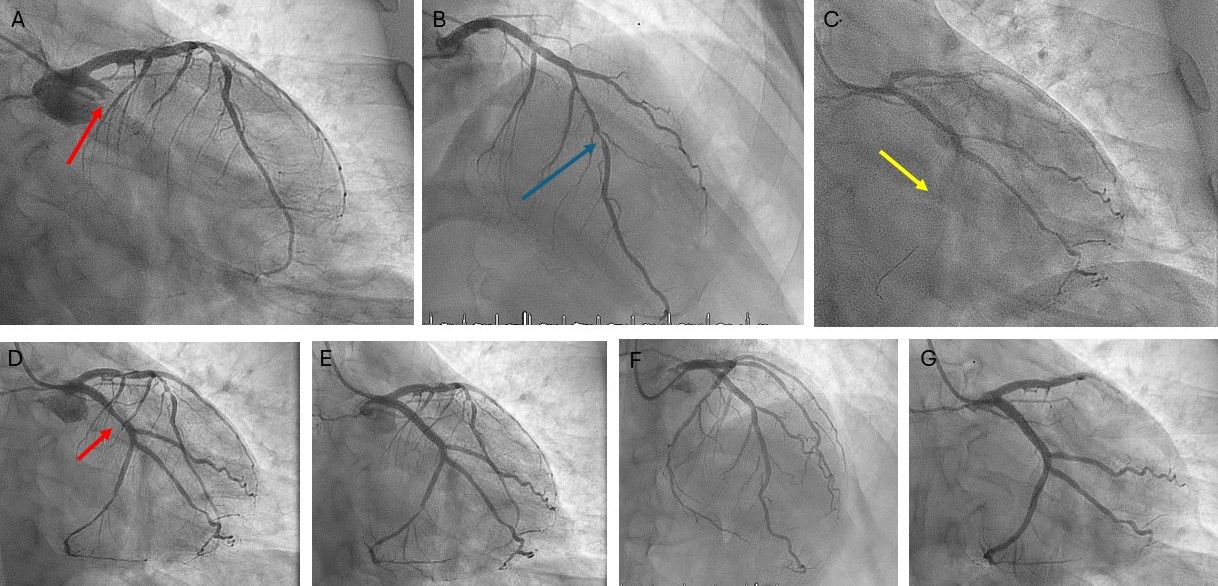
Emergency coronary angiography via the right radial artery revealed a fresh, proximal total occlusion of the left circumflex artery (LCx) with a very high thrombus burden (Figure 2A, Video 1). The left anterior descending artery (LAD) showed 90% to 95% stenosis in the mid-segment (Figure 2B), while the small-caliber, non-dominant right coronary artery was subtotally occluded.
A workhorse guidewire successfully crossed the occlusion and was advanced into an obtuse marginal (OM) branch. Predilatations with semicompliant balloons did not restore flow; therefore, 3 sessions of thrombus aspiration were performed followed by an intracoronary bolus infusion of tirofiban. Flow was restored in 2 large OM branches; however, because of high thrombus burden in the atrioventricular (AV) segment of the LCx (Figure 2C; Video 2), additional thrombus aspiration was performed in order to restore flow in the AV-LCx. Percutaneous coronary intervention with a drug-eluting stent (DES) was performed in the mid-LCx culprit lesion (Figure 2D and E; Videos 3 and 4), and the patient was transferred to the coronary care unit. Stenting of the severe lesion in the mid-segment of the AV-LCx was deferred. Four days later, the LAD was treated with 3 DES (Figure 2F); also, a DES was implanted in the AV-LCx lesion (Figure 2G). The patient had an uneventful recovery.
Since its first description by de Winter et al in 2008, the de Winter sign has been recognized as a STEMI equivalent, typically indicating acute proximal LAD occlusion. Approximately 2% of patients with acute myocardial infarction due to LAD occlusion exhibit this ECG pattern. To the best of our knowledge, only one other report in the literature has described a de Winter ECG pattern associated with LCx occlusion. Early recognition of this ECG pattern is crucial, as it mandates immediate reperfusion therapy similar to that for STEMI and can significantly improve clinical outcomes.
Affiliations and Disclosures
Konstantinos C. Theodoropoulos, MD, PhD; Spyridon-Filippos Papadopoulos, MD; George Samprokatsidis, MD; Alexandra Liakopoulou, MD, PhD; George Perdikos, MD; Matthaios Didagelos, MD, PhD; George Kassimis, MD, PhD; Antonios Ziakas, MD, PhD
From the 1st Cardiology Department, AHEPA University Hospital, Aristotle University of Thessaloniki, Thessaloniki, Greece.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Consent statement: The authors confirm that informed consent was obtained from the patient for the interventions described in the manuscript and to the publication of their data.
Address for correspondence: Konstantinos C. Theodoropoulos, MD, PhD, 1st Cardiology Department, AHEPA University Hospital, Aristotle University of Thessaloniki, Thessaloniki, Greece. Email: ktheod2005@hotmail.com; X: @ktheod2005