Modified Deployment of a Self-Expanding Aortic Endoprosthesis for Congenital Aortic and Conduit Repair Applications
© 2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2025. doi:10.25270/jic/25.00368. Epub December 22, 2025.
Endovascular grafts developed for adult aortic disease are now being applied in congenital heart interventions. The GORE TAG Thoracic Branch Endoprosthesis Extender (W.L. Gore & Associates) provides a self-expanding expanded polytetrafluoroethylene-covered platform with excellent conformability, radiopacity, and controlled deployment. Its successful use in right ventricular outflow tract preparation prior to transcatheter pulmonary valve replacement highlights its versatility and growing value in complex congenital applications.
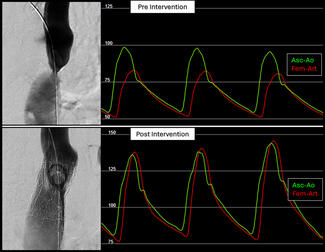
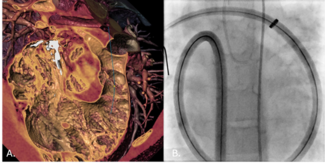
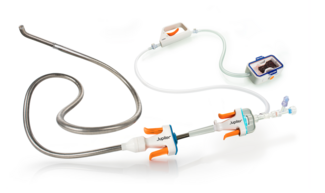
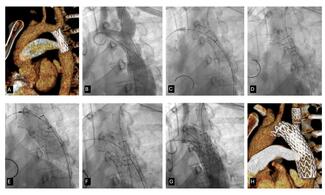
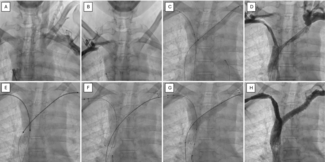
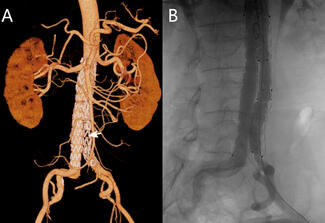
The device’s design reflects its original purpose in the aortic arch, where maintaining flow to the brachiocephalic vessels is critical. To achieve this, the graft includes a retention sleeve sutured to the delivery handle, allowing removal of the sleeve after deployment and ensuring no material from the delivery system extends across the branch vessels. In congenital applications—such as conduit rehabilitation or coarctation repair distal to the left subclavian artery—this feature is unnecessary and may introduce technical challenges. When deployed without an external sheath, traction on the fixed sleeve during withdrawal can transmit force to the implant, risking subtle displacement or distortion (Video). A modification eliminates this issue: cutting the suture connection allows the sleeve to release with graft deployment (Figure). This adjustment prevents transmitted tension during removal of the delivery catheter, maintaining stable graft position and contour. The modification enables safe, sheath-independent removal while preserving full graft support and integrity during delivery. This refinement broadens the versatility of the GORE TAG system for pediatric and congenital endovascular applications.

Endovascular grafts developed for adult aortic disease are now being applied in congenital heart interventions. The GORE TAG Thoracic Branch Endoprosthesis Extender (W.L. Gore & Associates) provides a self-expanding expanded polytetrafluoroethylene-covered platform with excellent conformability, radiopacity, and controlled deployment. Its successful use in right ventricular outflow tract preparation prior to transcatheter pulmonary valve replacement highlights its versatility and growing value in complex congenital applications.
The device’s design reflects its original purpose in the aortic arch, where maintaining flow to the brachiocephalic vessels is critical. To achieve this, the graft includes a retention sleeve sutured to the delivery handle, allowing removal of the sleeve after deployment and ensuring no material from the delivery system extends across the branch vessels. In congenital applications—such as conduit rehabilitation or coarctation repair distal to the left subclavian artery—this feature is unnecessary and may introduce technical challenges. When deployed without an external sheath, traction on the fixed sleeve during withdrawal can transmit force to the implant, risking subtle displacement or distortion (Video). A modification eliminates this issue: cutting the suture connection allows the sleeve to release with graft deployment (Figure). This adjustment prevents transmitted tension during removal of the delivery catheter, maintaining stable graft position and contour. The modification enables safe, sheath-independent removal while preserving full graft support and integrity during delivery. This refinement broadens the versatility of the GORE TAG system for pediatric and congenital endovascular applications.
Affiliations and Disclosures
Jason H. Anderson, MD1,2; Kaitlyn Krebushevski, MD1,2; Allison K. Cabalka, MD1,2; Abigail M. Sutter, PA-C1; Nathaniel W. Taggart, MD1,2; Pradyumna Agasthi, MD3
From the 1Department of Pediatric and Adolescent Medicine, Division of Pediatric Cardiology, Mayo Clinic, Rochester, Minnesota; 2Department of Cardiovascular Medicine, Division of Structural Heart Diseases, Mayo Clinic, Rochester, Minnesota; 3Department of Cardiology, Saint Francis Heart and Vascular Institute, Tulsa, Oklahoma.
Disclosures: Dr Anderson is on the cardiac advisory board for W.L. Gore & Associates and is a consultant for Edwards Lifesciences and Medtronic. Dr Cabalka is a consultant for Edwards Lifesciences and B. Braun Medical, Inc. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Consent statement: Informed consent was obtained for all procedures and images, with patient approval for publication.
Address for correspondence: Jason H. Anderson, MD, Mayo Clinic, Rochester, MN 55905, USA. Email: Anderson.Jason@mayo.edu; X: @Dr_JHAnderson