Case Presentation: BRAF-V600E Mutant Pediatric Low-Grade Glioma

Patient Case
A 2-year-old girl presented to her PCP with a complaint of 4 months of mild right eye outward deviation. Additionally, parents reported that their daughter sometimes “misses people” when trying to give them a hug, suggesting to them that her vision is impaired. She has not had any headaches, vomiting, ataxia, or other neurologic changes. This child has otherwise been healthy throughout her life with normal growth and development.
The child was quickly referred to an ophthalmologist whose findings included right optic nerve hypoplasia with pallor, and a right-sided afferent pupillary defect with decreased visual acuity. There was no papilledema. Physical exam findings were otherwise unremarkable, including there were no findings suggestive of NF1.
An MRI of the brain with and without contrast demonstrated an extensive optic pathway glioma involving the right optic nerve, optic chiasm/hypothalamus, bilateral optic tracts, and bilateral thalami.
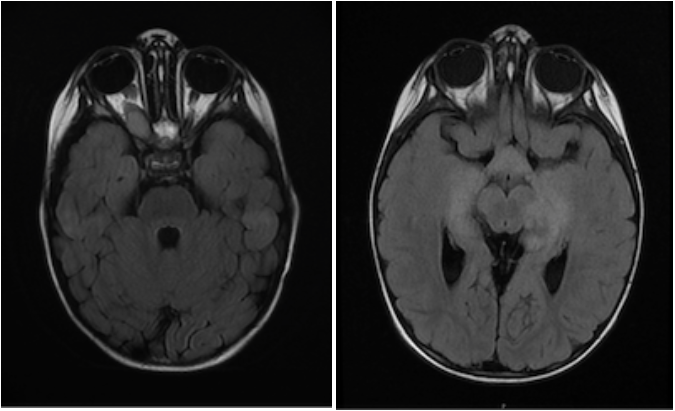
Treatment was initiated with monthly carboplatin/vincristine. Following 3 months of this therapy, MRI brain/orbits demonstrated a mixed response with increased size of the pre-chiasmatic right optic nerve tumor and stable findings elsewhere. Ophthalmologic evaluation showed stability of prior findings. A biopsy was offered, but parents declined due to concern for possible risk of harm to vision. Carboplatin/vincristine therapy was discontinued, and treatment with daily trametinib was begun. However, while still on trametinib monotherapy 12 months later, MRI brain/orbits showed progressive disease with a new left temporal lobe lesion. A biopsy of this lesion was safely performed which revealed a pilocytic astrocytoma, WHO grade 1, with a BRAF-V600E mutation. Dabrafenib was added to the existing trametinib therapy, which resulted in disease stabilization immediately, followed by disease response with tumor shrinkage in all areas.
Combination dabrafenib/trametinib therapy was given for 2.5 years, after which the medical team and family mutually determined to discontinue treatment in favor of disease surveillance with MRI and ophthalmologic examination, which have both been stable since that time.











